Abstract
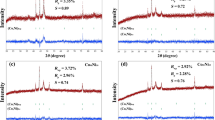
A method for the preparation of a SiO2–TiO2/Cr2O3(840) porous composite material in a block form with walls made of a silicate matrix and the interconnected channels of the matrix containing TiO2/Cr2O3(840) spherical hollow oxide composites was proposed. The specific surface area (Ssp) of the block composites was 212.2 m2/g, which is 20 times greater than the value of Ssp for spherical composites not fixed in the silicon(IV) oxide matrix. The composition of the TiO2/Cr2O3(840) and SiO2–TiO2/Cr2O3(840) samples was studied by X-ray diffractometry and X-ray microanalysis, which indicated the presence of chromium(III) oxide, titanium(IV) oxide, and an amorphous silicon(IV) oxide phase in the latter. The SiO2–TiO2/Cr2O3(840) block composite material exhibited catalytic activity in the deep oxidation reaction of para-xylene: a 100% conversion of the organic substance was achieved at a temperature of 350°C.





Similar content being viewed by others
REFERENCES
Porsin, A.V., Kulikov, A.V., Dalyuk, I.K., Rogozhnikov, V.N., and Kochergin, V.I., Chem. Eng. J., 2015, no. 15, p. 00226.
Zauresh, T., Zheksenbaeva, S.A., Tungatarova, T.S., and Baizhumanova, E.Sh., Chem. Eng. Trans., 2015, vol. 15, p. 1213
Goodman, E.D., Dai, Sh., Yang, An.-Ch., Wrasman, C., Gallo, A., Russell, S., Adam, B., Hoffman, S., Jaramillo, Th.F., Graham, G.W., Pan, X., and Cargnello, M., ACS Catal., 2017, vol. 7, p. 4272.
Lokhande, S., Doggali, P., Rayalu, S., Devotta, S., and Labhsetwar, N., Atmos. Pollut. Res., 2015, vol. 5, p. 589.
Tidahy, H.L., Siffert, S., Wyrwalski, F., Lamonier, J.-F., and Aboukais, A., Catal. Today, 2007, vol. 119, p. 317.
Barbato, P.S., Colussi, S., Benedetto, A.D., Landi, G., Lisi, L., Llorca, J., and Trovarelli, A., J. Phys. Chem. C, 2016, vol. 120, no. 24, p. 13039.
Baidya, T., Murayama, T., Bera, P., Safonova, O.V., Steiger, P., Katiyar, N.K., Biswas, K., and Haruta, M., J. Phys. Chem. C, 2017, vol. 121, p. 15256.
Albert, J.J., Johna, F., Ronald, M., Arorab, A., Sharmab, H., Hamilton, A., and Wessleya, J.J., Int. J. Comput. Sci. Eng., 2017, vol. 5, p. 19.
Yim, S.D. and Nam, I.-S., J. Catal., 2004, vol. 221, p. 601.
Yim, S.D., Chang, K.-H., Koh, D.J., Nam, I.-S., and Kim, Y.G., Catal. Today, 2000, vol. 63, p. 215.
Padilla, A.M., Corella, J., and Toledo, J.M., Appl. Catal., B, 1999, vol. 22, p. 107.
Boreskov, G.K., Geterogennyi kataliz (Heterogeneous Catalysis), Moscow: Nauka, 1986.
Berner, M.K., Zarko, V.E., and Talavar, M.B., Combust., Explos. Shock Waves, 2013, vol. 49, no. 6, p. 625.
Lopatin, S.A., Tsyrul’nikov, P.G., Kotolevich, Y.S., Mikenin, P.E., Pisarev, D.A., and Zagoruiko, A.N., Catal. Ind., 2015, no. 3, p. 329.
Kantserova, M.R., Orlik, S.N., and Shvets, A.V., Catal. Ind., 2014, vol. 6, no. 2, p. 88.
RF Patent 2608125, 2017.
Shamsutdinova, A.N., Brichkov, A.S., Paukshtis, E.A., Larina, T.V., Cherepanova, S.V., Glazneva, T.S., and Kozik, V.V., Catal. Commun., 2017, vol. 89, p. 1.
RF Patent 2505800, 2014.
Ignatenkova, V.V., Bespalov, A.V., and Grunskii, V.N., Usp. Khim. Khim. Tekhnol., 2010, vol. 24, no. 2, p. 13
Fukumoto, S., Nakanishi, K., and Kanamori, K., New J. Chem., 2015, vol. 39, p. 6761.
Trogadas, P., Nigra, M.M., and Coppens, M.-O., New J. Chem., 2016, vol. 40, p. 4016.
Tomas, Dzh. and Tomas, U., Geterogennyi kataliz (Heterogeneous Catalysis), Moscow: Mir, 1969
Amereh, M., Haghighi, M., and Estifaee, P., Arabian J. Chem., 2018, vol. 11, p. 81.
Huang, Sh., Zhang, Ch., and He, H., Catal. Today, 2008, vol. 139, p. 15.
Wu, Zh., Deng, J., Xie, Sh., Yang, H., Zhao, X., Zhang, K., Lin, H., Dai, H., and Guo, G., Microporous Mesoporous Mater., 2016, vol. 224, p. 311.
Nesterenko, S.S., Ovchinnikova, E.V., and Chumachenko, V.A., Catal. Ind., 2014, vol. 6, no. 4, p. 329.
Wang, Y., Zhang, Ch., Liu, F., and He, H., Appl. Catal., B, 2013, vol. 142, p. 72.
Jeong, M.-G., Park, E.J., Jeong, B., Kim, D.H., and Kim, Y.D., Chem. Eng. J., 2014, vol. 237, p. 62.
ACKNOWLEDGMENTS
We are grateful to the scientific group of Prof. V.I. Syryamkin from the National Research Tomsk State University for obtaining the results by 3D microtomography.
Funding
This work was performed within the framework of state contract no. 10.2281.2017/PCh.
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated by V. Makhlyarchuk
Rights and permissions
About this article
Cite this article
Rogacheva, A.O., Buzaev, A.A., Brichkov, A.S. et al. Catalytically Active Composite Material Based on TiO2/Cr2O3 Hollow Spherical Particles. Kinet Catal 60, 484–489 (2019). https://doi.org/10.1134/S002315841904013X
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S002315841904013X