Abstract
Omega-3 fatty acids, and specifically docosahexaenoic acid (DHA), are important and essential nutrients for human health. Thraustochytrids are recognised as commercial strains for nutraceuticals production, they are group of marine oleaginous microorganisms capable of co-synthesis of DHA and other valuable carotenoids in their cellular compartment. The present study sought to optimize DHA and squalene production by the thraustochytrid Schizochytrium limacinum SR21. The highest biomass yield (0.46 g/gsubstrate) and lipid productivity (0.239 g/gsubstrate) were observed with 60 g/L of glucose, following cultivation in a bioreactor, with the DHA content to be 67.76% w/wtotal lipids. To reduce costs, cheaper feedstocks and simultaneous production of various value-added products for pharmaceutical or energy use should be attempted. To this end, we replaced pure glucose with organosolv-pretreated spruce hydrolysate and assessed the simultaneous production of DHA and squalene from S. limacinum SR21. After the 72 h of cultivation period in bioreactor, the maximum DHA content was observed to 66.72% w/wtotal lipids that was corresponded to 10.15 g/L of DHA concentration. While the highest DHA productivity was 3.38 ± 0.27 g/L/d and squalene reached a total of 933.72 ± 6.53 mg/L (16.34 ± 1.81 mg/gCDW). In summary, we show that the co-production of DHA and squalene makes S. limacinum SR21 appropriate strain for commercial-scale production of nutraceuticals.
Similar content being viewed by others
Introduction
Omega-6 (n-6) and omega-3 (n-3) are polyunsaturated fatty acids (PUFAs), whose precursors include linoleic acid and alpha-linolenic acid. They are considered essential because humans cannot synthesize them due to insufficient levels of elongases and delta-6-desaturases1,2. Although conversion of alpha-linolenic acid into omega-3 fatty acids, such as eicosapentaenoic acid (C20:5n−3), docosapentaenoic acid (C22:5n−3), and docosahexaenoic acid (C22:6n−3, DHA) occurs in humans, it happens at a very slow rate3 and so these fatty acids must be provided by the diet. These PUFAs have several health benefits and have been found to be very effective in the preventions and treatment of fetal diseases4. DHA plays a major role in cell signalling and is found mainly in the brain and retina tissues5. Additionally, it acts as an anti-inflammatory agent, a precursor of several metabolites and potent lipid mediators, and could be used to treat several cardiovascular or neurologic disorders, such as hypertension and Alzheimer’s6. Fish of the Salmonidae, Scombridae, and Clupeidae families have been the sole commercial source of DHA as their oil contains approximately 20 to 30% DHA3. However, DHA purification and concentration from fish oil is costly and the resulting oil quality depends on species, location, and pollution7. Furthermore, fish-derived DHA is unsuitable for vegetarians, and diminishing fish stocks together with marine pollution limit the increasing demand for DHA7. Oils from genetically engineered plant oilseeds, such as Brassica juncea, Arabidopsis thaliana, and Camelina sativa can, to some extent, fulfil the demand for DHA; however, omega-3 PUFAs content in vegetable oils is very low and there is no method to concentrate them for commercial purposes8. The production of plant oil is totally dependent on the environmental factors and the accessibility of arable land. In comparison, oleaginous microorganisms achieve better productivity of omega-3 fatty acids than plants9; therefore, microalgae, fungi, and bacteria could serve as a renewable and sustainable source of DHA.
Microalgae are natural producers of omega-3 PUFAs and can be grown under autotrophic, mixotrophic, and heterotrophic conditions10. The unicellular structure of microalgae facilitates the cultivation at large scale contrast to fungi where they form mycelial structure11. Thraustochytrids represent a marine group of oleaginous eukaryotic protists (class Labyrinthula of the Chromista Kingdom) often referred to as microalgae, even though they lack photosynthetic capability12. Many species of the genera Schizochytrium, Thraustochytrium, and Ulkeniain grow exclusively heterotrophically and accumulate considerable amounts of triacylglycerols with a high proportion of long-chain PUFAs, particularly DHA, which makes them suitable for commercial exploitation13.
Schizochytrium limacinum SR21, also known as Aurantiochytrium limacinum SR21, was selected for the present study. Isolation, identification, and optimization of cultivation conditions in flasks to enhance DHA production have been described before; however, few have offered insights into bioprocess development potential and the use of sources other than pure glucose or glycerol. Although crude glycerol has been tested on S. limacinum SR21, the presence of impurities (e.g., methanol and soap) can inhibit growth and lower DHA productivity. The commercial viability of microbial oils must be supported by inexpensive raw materials. Here, forest biomass consisting of non-edible lignocellulosic feedstocks was used. Forest-based industries contribute substantially to the Swedish economy and rely on vast swaths of forests, particularly those of Norway spruce (Picea abies), which accounts for 40.8% of the forest’s total standing volume14. Production of high-value nutraceutical compounds from side- and waste-streams of the forest sector could offer an important additional source of revenue to the forest-based industry. At the same time, nutraceuticals-based industries need to make upstream and downstream processing of DHA more sustainable and more economical13 by, for example, integrating it with simultaneous production of various value-added products for the pharmaceutical and petrochemical industries. Most thraustochytrids produce several antioxidants, such as β-carotene, squalene, and astaxanthin15. Aurantiochytrium/Schizochytrium species produce the polyunsaturated hydrocarbon triterpenoid squalene (2,6,10,15,19,23-hexamethyl-6,6,10,14,18,20-tetracosahexane)16. The most common source of squalene is liver oil of deep-sea sharks and whales; however, contamination with heavy metals and polychlorinated biphenyls poses a safety risk, while the oil’s often putrid odor and unpleasant taste diminish its appeal to consumers17. Furthermore, downstream processing of squalene from liver oils is hampered by the presence of chemically similar compounds such as cholesterol. Squalene can reduce serum cholesterol levels, enhance the immune response, and suppress tumor proliferation18,19, as well as increase immune responsiveness to vaccines20. In cosmetics, it is used as an antioxidant to quench singlet oxygen (1O2)21. Recently its value towards the production of high-grade aviation fuels has gained interest. Squalene is branched hydrocarbon that can be converted into smaller alkanes by catalytic conversion using ruthenium on ceria (Ru/CeO2) without skeletal isomerization and aromatization22. It has been already mentioned that Ru/CeO2-alcohol thermal catalysts are significantly superior to conventional methods using metal‐acid bifunctional catalyst23. Squalene obtained from Botryococcus braunii was transformed into bio jet fuel by using Ru/CeO2-alcohol thermal catalyst23. Due to increasing demand of squalene it cannot be fulfilled by mining from liver of marine mammals which is even not sustainable option for aquatic ecosystem24. Some plants can synthesize squalene but the quantities is not sufficient for commercial usage25. The only prominent source of squalene is thraustochytrids that can cultivate on industrial scale26.
In the present study, DHA and squalene co-production from S. limacinum SR21 were optimized in flask and bioreactor at batch conditions. Heterotrophic cultivation of S. limacinum SR21 was carried out on saccharified organosolv-pretreated forest biomass, to test a cost-effective and sustainable production of microbial DHA.
Results and Discussion
Batch cultivation of S. limacinum SR21 in Erlenmeyer flasks
Initially, we aimed to enhance total lipid concentration and increase DHA content by first optimizing the concentration of artificial seawater in growth medium during batch cultivation of S. limacinum SR21 in Erlenmeyer flasks. This was done by varying the amount of artificial seawater from 25% to 100% (v/v) at a fixed concentration of glucose (30 g/L) along with a C/N ratio of 10 adjusted by yeast extract (Fig. 1A). While this strain can survive well at 0% artificial seawater, cell dry weight (6.43 ± 1.23 g/L) and lipid concentration (1.23 ± 0.21 g/L) were low under these conditions. The highest cell dry weight (13.1 ± 0.43 g/L) and total lipids (6.05 ± 0.28 g/L or 46.18 ± 0.92 w/w of lipid content) were observed with 50% artificial seawater (Fig. 1A). The DHA content was reported the lowest (23.12% of total lipid) at 0% artificial seawater, while 36.87% DHA was observed at 50% artificial seawater. The DHA content was not significantly increased with increasing concentration of artificial seawater from 50 to 100% (Fig. 1A). Kim et al. (2015) suggested that increasing the concentration of sea salt from 2 g/L to 20 g/L reduced cell rupturing of Aurantiochytrium sp. KRS101 and augmented biomass from 13.61 to 17.31 g/L27.
(A) Cultivation of S. limacinum SR21 at various concentrations of artificial seawater with fixed 30 g/L of glucose and C/N (g/g) ratio of 10. (B) Effect of various C/N (g/g) ratios on cell dry weight (g/L), total lipid concentration, lipid content and DHA content of S. limacinum SR21 cultivated in Erlenmeyer flasks. Growth medium contained 30 g/L of glucose and 50% of artificial seawater. (C) Cells of S. limacinum SR21 cultivated at a C/N ratio of 20 and with 30 g/L glucose were separated into two layers after centrifugation at 8000 rpm (7881*g) for 10 min. (D) Effect of various concentrations of glucose on cell dry weight (g/L), total lipid concentration (g/L), lipid content (%. w/w), DHA content (%, total lipids), biomass and lipid yield (g/g substrate), and biomass and lipid productivity (g/L/day) of S. limacinum SR21 cultivated in flasks. Data values represent average with standard deviation.
Limitation of key nutrients, such as nitrogen and phosphorus, causes stress to oleaginous microorganisms, retards growth, and leads to the conversion of excess carbon sources into lipids28. To study the effect of varying the C/N (g/g) ratio from 10 to 50, S. limacinum SR21 was cultivated at a fixed glucose concentration of 30 g/L and 50% artificial seawater (Fig. 1B). The highest cell dry weight (13.1 ± 0.43 g/L) and total lipid concentration (6.05 ± 0.28 g/L), corresponding to 46.18 ± 0.92% w/w lipid content, were obtained at C/N 10. At a C/N ratio of 20, lipid content remained similar (46.61 ± 0.81% w/w), but cell dry weight and lipid accumulation were only 11.05 ± 0.31 g/L and 5.15 ± 0.16 g/L, respectively. Increasing the C/N ratio to 30, 40, and 50 resulted in a further decrease of biomass and lipid formation (Fig. 1B). The DHA content was also decreased from 36.43% to 28.98% with increasing C/N ratio from 10 to 50 (Fig. 1B). Although higher C/N ratios are reportedly beneficial for high lipid accumulation in oleaginous microorganisms, this was not our case. The decreased biomass and lipid synthesis were likely due to lower levels of yeast extract, and consequently fewer vitamins and minerals, at increasing C/N ratios. Huang et al.29 showed that increasing the C/N ratio from 1.25 to 1.875 (with 75 g/L glycerol) promoted lipid accumulation; however, the accompanying reduction in biomass severely decreased total lipid and DHA concentrations29.
Interestingly, at a high C/N ratio, growth shifted from a vegetative to a reproductive phase, resulting in lower biomass and lipids. At a low C/N ratio, all glucose was consumed before reaching stationary phase; while at a high C/N ratio, the cells started to form zoosporangia and released zoospores into the medium (Fig. 1C), before glucose was consumed. A representative microscopic image of S. limacinum SR21 cultivated on 60 g/L of glucose with two different C/N ratio (10 and 50) is presented in Supplementary Fig. S1, where it is showing the formation of zoosporangia as a result of sexual reproduction at high C/N ratio and the emptied cells are clearly visualized after releasing of zoospores from the cells. Abad and Turan30 reported that discharge of zoospores from A. limacinum occurred only at the highest growth rate30. Aurantiochytrium mangrovei MP2 and Aurantiochytrium sp. KRS101 are susceptible to cell rupturing in freshwater medium, causing their cytoplasm to be released into the medium in a phenomenon that is highly dependent on nutrients’ availability27,31. Kim et al.27 suggested that cell rupture was more evident at lower C and N concentrations27. Whereas these authors observed a floating layer of small lipid bodies after centrifuging the cultures27,31, we could not dissolve the floating layer in chloroform and methanol (2:1 v/v) solution, ruling out the presence of lipid droplets. We believe that the floating layer could be explained by the shift from vegetative to zoosporic phase, whereby cells burst and release zoospores in the medium. Recently, evidence of sexual reproduction and morphological characterization of vegetative cells as well as zoosporangia of Aurantiochytrium acetophilum, A. limacinum, and Schizochytrium mangrovei was described32.
Once artificial seawater content and C/N ratio were optimized, different concentrations of glucose were tested at a range of 30–120 g/L (Fig. 1D). Cell dry weight increased from 13.1 ± 0.36 to 25.59 ± 0.32, 32.64 ± 0.47, and 36.78 ± 0.76 g/L as glucose increased from 30 to 60, 90, and 120 g/L of glucose. The corresponding lipid concentrations were 6.05 ± 0.24, 11.91 ± 0.43, 18.62 ± 0.72, and 19.91 ± 0.43 g/L. The increments in biomass and total lipid concentration were significant when shifting cultivation from 30 to 60 g/L but not when going from 90 to 120 g/L of glucose. The highest lipid content of 57.05 ± 0.89% w/w was recorded with 90 g/L of glucose, whereas the highest biomass productivity (0.21 g/L/h) and lipid productivity (0.09 g/L/h) were observed with 60 g/L of glucose (Fig. 1D). The DHA content in the total lipids was 36.43% with 30 g/L glucose and increased to 45.54% with 60 g/L glucose. Further increasing glucose concentration didn’t show any significant increment in DHA content (Fig. 1D). Chi et al.33 cultivated S. limacinum SR21 (ATCC MYA-1381) on glucose. They reported cell dry weight and biomass productivity of 18.47 g/L and 3.08 g/L/day (0.12 g/L/h), respectively when growing cells on 90 g/L of glucose33. Thus, biomass was higher in the present study, which could be attributed to complete glucose utilization. In a different study, whereby S. limacinum SR21 was cultivated on 30 to 120 g/L of glucose, biomass increased continuously up to 90 g/L of glucose and decreased thereafter; notably, cell dry weight (24.2 g/L) and lipid concentration (18.2 g/L) were also highest at 90 g/L of glucose34. These cell dry weight and biomass productivity values were still lower than those observed in the present study with 60 g/L of glucose, which may be explained by a different amount of yeast extract and cultivation conditions. Similar substrate inhibition at higher C source concentration was observed by Yokochi et al.35 when S. limacinum SR21 was cultivated with 30 to 120 g/L of glucose. Biomass synthesis became inhibited when the initial glucose concentration reached > 90 g/L35.
Optimization of aeration rate and glucose concentration during batch cultivation in a bioreactor
Oxygen plays a crucial role in biomass, lipid, and DHA production36. Once the C/N ratio and concentration of artificial seawater were optimized, aeration rate was assessed by testing different flow rates (0.66, 1.33, 2, 2.66 vvm) in a 3-L bioreactor with 1 L working volume (Fig. 2A). Glucose utilization was severely affected by different aeration rates: at 0.66 vvm, glucose was fully consumed after 120 h of cultivation; whereas at 2 vvm, glucose was consumed in less than 72 h (Fig. 2A). Although biomass concentration was similar (~28 g/L) in all cases, the significantly different glucose consumption rates affected biomass productivity (g/L/h). At 2 vvm, cells grew faster than at 1.33 or 0.66 vvm, which caused utilization of all available oxygen in the medium. A high aeration rate in the initial phase of growth was responsible for high cell density; whereas later on, low oxygen promoted lipid synthesis (Fig. 2B). At high cell concentrations, oxygen consumption may exceed its supply, leading to oxygen limitation37. Surprisingly, at a high aeration rate (2 vvm), DO rapidly dropped to zero after 12 h and remained < 10% of saturation until 72 h of cultivation (Fig. 2B). Biomass synthesis reached 13.24 g/L at 24 h, which was almost 4 times higher than the value obtained with 1.33 vvm and at this stage lipid accumulation remained very low (only 1.11 g/L) (Fig. 2A,B). No significant changes were observed in biomass or lipid accumulation and glucose consumption when the aeration rate was shifted from 2 to 2.66 vvm (data not shown), hence 2 vvm was selected for further experiments. A high aeration rate has been reported to increase cell growth rate38,39, which supports our results. The DHA content (%, total lipids) was 47.76%, 58.65% and 66.72% at 0.33, 1.33 and 2 vvm of aeration rate, respectively (Fig. 2C). The abundance of DO in cultivation medium promotes cell respiration, energy metabolism, and carbon flux to the tricarboxylic acid cycle, eventually favoring high growth rate and production of valuable metabolites39. There are two contradictory reports: one suggests that molecular oxygen is required for desaturation during PUFA synthesis in most oleaginous microorganisms40; the other claims that DHA production can be enhanced by low DO saturation level41. A possible explanation for these two hypotheses is the existence of two distinct pathways for the biosynthesis of DHA in thraustochytrids42. The first occurs via an oxygen-dependent desaturation/elongation pathway (fatty acid synthesis pathway; FAS), in which molecular oxygen is required to start the synthesis of 18:4–6,9,12,15 from 18:3–9,12,15 by Δ6 desaturase, and elongation to 20:4–8,11,14,1743,44. The other, oxygen-independent polyketide synthase pathway (PKS) only occurs in bacteria and some eukaryotes including Schizochytrium42. Chang et al. (2013) showed that a continuous supply of oxygen to a culture of Schizochytrium sp. S31 enhanced growth and DHA synthesis, whereas a high oxygen transfer rate enhanced metabolic activity and promoted substrate assimilation to produce acetyl-CoA and NADPH for lipid synthesis45, confirming an earlier report40. Based on the genome annotation results of S. limacinum SR21, Ye et al., (2015) suggested that this microorganism doesn’t have delta-4 desaturase and some ORFs were considered to be as PKS proteins. These results confirm that the DHA could not be synthesized by FAS route but rather utilizes PKS pathway for DHA synthesis46. However, similar to fatty acids synthesis pathway, it requires a high amount of acetyl-CoA and NADPH for DHA synthesis via PKS pathway42,46,47,48.
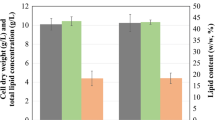
Effect of various aeration rates on (A) cell dry weight and glucose utilization, as well as (B) DO (%) and lipid concentration of S. limacinum SR21 cultivated on 60 g/L of glucose in a bioreactor. (C) Effect of aeration rate on DHA content and concentration of S. limacinum SR21 cultivated on 60 g/L of glucose in a bioreactor (D) Effect of glucose concentration on cell dry weight (g/L), total lipid concentration (g/L), lipid content (% w/w), biomass and lipid yield (g/g substrate), and biomass and lipid productivity (g/L/h) of S. limacinum SR21 cultivated in a bioreactor. Data values represent average with standard deviation.
After optimizing the aeration rate, we identified the highest applicable glucose concentration (Fig. 2D). Cell dry weight increased continuously from 13.69 ± 0.65 to 46.03 ± 0.69 g/L as glucose increased from 30 to 120 g/L. Similarly, lipid concentration increased from 6.65 ± 0.54 to 18.81 ± 0.73 g/L when glucose increased from 30 to 90 g/L; but dropped to 15.03 ± 0.58 at 120 g/L of glucose. The highest lipid content (50.91% w/w), lipid productivity (0.20 g/L/h), biomass yield (0.47 g/gsubstrate), and lipid yield (0.24 g/gsubstrate) were observed at 60 g/L of glucose. Similarly, when Chen and Yang (2018) cultivated Thraustochytrium sp. BM2 at glucose concentrations ranging from 15 to 90 g/L, the highest lipid content with maximum DHA productivity was observed at 60 g/L of glucose, however, the highest biomass productivity was reported at 75 g/L of glucose but dropped thereafter49. In flasks, both cell dry weight and lipids increased continuously with the consumption of glucose, irrespective of its concentration. Instead, in a bioreactor, biomass synthesis was rapid in the early log phase, after which glucose was consumed to support lipid synthesis, as demonstrated by lipid-free biomass. The different levels of biomass and lipid production exhibited by thraustochytrid strains in flask compared to bioreactor cultivation have not been explained yet. To investigate the effect of glucose utilization by S. limacinum SR21 on cell dry weight, lipid concentration, and lipid content, time-course experiments were performed in flasks and a bioreactor (Supplementary Fig. S2). Iida et al.50 suggested that Thraustochytrium roseum grew better in flasks because high mechanical stirring inhibited growth in the bioreactor50. In contrast, Nakahara et al. suggested the opposite was true for Schizochytrium sp. SR21 because the cells of this strain is extremely resistant to mechanical stirring51.
Fatty acid profile and DHA content in S. limacinum SR21 lipids
The fatty acid profile of S. limacinum SR21 cultivated in 60 g/L of glucose is presented in Fig. 3. All thraustochytrid strains have potential to synthesize mainly myristic acid (C14:0), palmitic acid (C16:0), stearic acid (C18:0), docosapentaenoic acid (C22:5) and docosahexaenoic acid C22:6 (DHA) whereas the composition and ratio usually varies with cultivation time52. After 24 h, the fatty acid profile from flask cultivation was C14:0 (3.9%), C15:0 (0.22%), C16:0 (51.77%), C17:0 (1.77%), C18:0 (4.59%), C22:5 (4.28%) and DHA (24.99%). In a bioreactor, the values doubled for DHA (53.63%). In flask cultivation, DHA content increased constantly from 24.99% at 24 h to 46.37% at 120 h (stationary phase); whereas, in a bioreactor, DHA content increased from 53.63% at 24 h to 66.72% at 72 h. The increment in DHA content in the course of cultivation might be related to declining DO in the medium. Due to increased utilization of carbon source with high aeration rate in the bioreactor, synthesis of biomass and lipid accumulation along with DHA content was significantly higher than those reported with flask cultivation. It has been reported previously that the high oxygen supply in the initial stage of cultivation leads to high substrate utilization capacity that further increase the lipid synthesis53.
Instead, Chang et al. (2013a) proposed that Schizochytrium sp. S3 produced more DHA in a baffled flask than in an unbaffled flask because the mass transfer coefficient (kLa) of oxygen was higher45. DHA synthesis in thraustochytrid species is thought to occur via two routes: the aerobic fatty acid synthase pathway, whereby oxygen is required in a series of desaturation and elongation steps54; and the oxygen-independent polyketide synthases pathway, whereby dehydration and isomerization reactions involving fatty acyl intermediates elongate the carbon chain without requiring dissolved oxygen (DO) during fermentation in later growth stages55. Chi et al. (2009) suggested that DHA production in S. limacinum SR21 could be improved by shifting DO levels during cultivation56.
Growth and lipid production by S. limacinum SR21 cultivated on OPSH
Although previous studies have optimized the bioprocessing of DHA production, supplementation with glucose and glycerol obtained from renewable substrates has not been established until recently. Here, commercial glucose has been replaced with glucose derived from spruce biomass, which represents a renewable substrate. Spruce was treated with a hybrid organosolv-steam explosion method, that resulted into pretreated solids with high-level-cellulose-content (72% w/w)57. The solids (10% w/w) were further hydrolyzed by commercial enzymes that resulted into 64.70 g/L of glucose57. The concentration of glucose for the fermentation experiments was adjusted to 60 g/L with an appropriate amount of OPSH and growth occurred in flask and bioreactor (Fig. 4A,B). After consumption 11.57 g/L and 14.60 g/L of glucose from flask and bioreactor in 24 h, the cell dry weight were 5.98 ± 0.36 g/L and 12.36 ± 0.67 g/L, respectively, whereas the respective total lipid concentration were 1.23 ± 0.45 g/L and 0.98 ± 0.53 g/L. In the flask, cell dry weight was relatively linear between 24 and 96 h, but increased thereafter, driven by elevated lipid synthesis until all glucose was consumed (Fig. 4A). In a bioreactor, cell dry weight and lipid synthesis augmented linearly from 24 h until stationary phase at 72 h (Fig. 4B). In the initial phase of bioreactor growth (24 to 48 h), increasing cell numbers and growth contributed to the cell dry weight; thereafter, the cells stopped growing and started synthesizing lipids between 48 and 72 h (stationary phase) until all glucose was consumed (Fig. 4B). Accordingly, lipid-free biomass was 11.38 g/L at 24 h, increased to 16.01 g/L at 48 h, and declined to 13.86 g/L at 72 h. For S. limacinum SR21 cultivated in flasks, the highest cell dry weight (26.87 ± 0.69 g/L) and lipid concentration (12.87 ± 0.95 g/L) was observed at 120 h of cultivation with complete glucose utilization (Fig. 4A). While in the bioreactor, all glucose was consumed at 72 h of cultivation whereas the cell dry weight and total lipid concentrations were 29.07 ± 0.84 g/L and 15.21 ± 0.72 g/L, respectively that was corresponded to 52.32% (w/w) of lipid content (Fig. 4B). The high biomass and lipid accumulation in bioreactor was likely due to efficient oxygen transfer and continuous pH adjustment during cultivation13. Once all the C source was depleted, the cells started to consume their own lipids and a decrease in lipid concentration was observed after 72 h in the bioreactor and 144 h in the flask, phenomenon that is often observed in oleaginous microorganisms. Similarly, Schizochytrium sp. S31 grown in flask on 20 g/L of glucose showed 6.01 g/L of cell dry weight with 2.38 g/L of total lipid concentration after all sugar utilization in five days58. In the present study, S. limacinum SR21 displayed a sudden change in morphology at this stage, shifting from vegetative to reproductive growth (Fig. 4C). The pattern of lipid synthesis in this microorganism is like the other oleaginous microalgae e.g. Crypthecodinium cohnii and Schizochytrium sp. S3159,60.
Time-course determination of cell dry weight, total lipid concentration, lipid content, residual glucose, DHA concentration, and DHA content of S. limacinum SR21 cultivated on OPSH containing 60 g/L of glucose in (A) Erlenmeyer flasks and (B) a bioreactor. (C) Morphological analysis of S. limacinum SR21 during cultivation on OPSH containing 60 g/L of glucose in a bioreactor. (D) Fatty acid profile (% w/wtotal lipid) of S. limacinum SR21 cultivated on OPSH containing 60 g/L of glucose in Erlenmeyer flasks and a bioreactor. Data values represent average with standard deviation.
In flask cultivation, the biomass and lipid productivity were 5.37 ± 0.23 g/L/d and 2.57 ± 0.25 g/L/d, respectively while with bioreactor cultivation, it was boosted to 9.69 ± 0.34 g/L/d and 5.07 ± 0.13 g/L/d, correspondingly (Table 1). Similarly, the biomass yield (0.48 ± 0.01 g/gsubstrate) and lipid yield (0.25 ± 0.01 g/gsubstrate) were observed higher in bioreactor cultivation at 72 h (Fig. 3A) than those obtained with flask cultivation at 120 h (0.44 ± 0.01 g/gsubstrate and 0.21 ± 0.01 g/gsubstrate, respectively).
Liang et al.61 reported 2.30 ± 0.06 g/L/d of biomass productivity and 1.15 ± 0.03 of lipid productivity, when S. limacinum SR21 was cultivated in flasks with 48 g/L of glucose61.
Overall, the fatty acid profile was similar to that of S. limacinum SR21 cultivated on pure glucose (Fig. 4D); the only difference was observed during extended cultivation after stationary phase. After 24 h, S. limacinum SR21 synthesized 24.99% DHA (w/wtotal lipids) when grown in a flask (Fig. 4A), but 53.63% w/wtotal lipids in a bioreactor (Fig. 4B). The highest DHA content in a flask (46.36% w/wtotal lipids), corresponding to 5.86 g/L DHA, was observed after 120 h; whereas in a bioreactor, maximal DHA content (66.72% w/wtotal lipids) and concentration (10.14 g/L) were even higher and were achieved sooner (72 h) (Fig. 4B and Table 1). A list of DHA producing thraustochytrids is presented in Table 2.
The light orange color of thraustochytrids cultures is given by the presence of carotenoids, in which squalene, astaxanthin, canthaxanthin, and zeaxanthin are prominent carotenoids13,62,63. In the present study, squalene was extracted along with lipids from S. limacinum SR21 cultivated on OPSH in a flask and bioreactor (Fig. 5). Squalene yield increased from 13.23 mg/gCDW (0.07 g/L) at 24 h to 38.45 mg/gCDW (0.56 g/L) at 72 h of cultivation in the flask but dropped to 9.12 mg/gCDW (0.21 g/L) at 144 h (Fig. 5A). This decline in squalene could be explained by the simultaneous formation of steryl ester (SE) from squalene, as identified by TLC (Fig. 5C, lanes 5, 6, 7). A similar trend was observed during bioreactor cultivation, whereby squalene yield increased from 29.32 mg/gCDW (0.36 g/L) at 24 h to 45.38 mg/gCDW (1.01 g/L) at 48 h, thus nearly doubling the values obtained with flask cultivation (Fig. 5B). The concentration then declined to 0.63 g/L at 96 h and a high amount of SE was observed at that point (Fig. 5D, lane 5). The bands for squalene from extracted lipid at 24 to 96 h was identified in Fig. 5D, lanes 2 to 5, where this squalene was purified as unsaponifiable fraction that was contaminated with some triacylglycerol and SE (Fig. 5E, lane 4) from saponified fraction of lipids with minor quantity of squalene (Fig. 5E, lane 3). The advanced purification of squalene can be performed by other chromatographic methods25,64. Nakazawa et al.65 used TLC to analyze almost 176 strains and identified 38 that were capable of synthesizing squalene65. Three different strains of S. mangrovei (FB1, FB2, FB3) were analyzed for squalene content, with FB1 reaching a maximum value of 0.162 mg/gCDW (8.53 g/L CDW)64. In another study, addition of 0.1 mM methyl jasmonate to the cultivation medium increased squalene by 60% to 1.17 ± 0.006 mg/gCDW after 48 h of cultivation66. In contrast to our results, Aurantiochytrium sp. 18W-13a synthesized high amounts of squalene (198 mg/gCDW) at 4 days of cultivation67.
Evolution of squalene yield (mg/gCDW) and concentration (g/L) during cultivation of S. limacinum SR21 on OPSH containing 60 g/L of glucose in (A) Erlenmeyer flasks and (B) a bioreactor. (C) TLC analysis of extracted lipids from S. limacinum SR21. Analysis of squalene in total lipid extracts from S. limacinum SR21cultivated on OPSH containing 60 g/L of glucose in Erlenmeyer flasks where lane 1, squalene standard; lanes 2–7, samples cultivated for 24–144 h; lane 8, squalene standard. (D) S. limacinum SR21cultivated on OPSH containing 60 g/L of glucose in bioreactor where lane 1, squalene standard; lanes 2–5, samples cultivated for 24–96 h. (E) Purification of squalene from total lipid extracts following saponification: lane 1, squalene standard; lane 2, unsaponifiable fraction; lane 3, saponified fraction; lane 4, purified product from the unsaponifiable fraction; lane 5, triacylglycerol (TAG) standard. FFA, free fatty acids; DAG, diacylglycerol; MAG, monoacylglycerol. Images presented in Fig. 5C–E were obtained after TLC of different samples in different time frame.
Conclusion
The present study optimized the cultivation parameters for attaining high cell dry weight and lipid accumulation by S. limacinum SR21 in flasks and a bioreactor. The highest biomass (0.39 g/gsubstrate) and lipid yields (0.20 g/gsubstrate) were obtained with 60 g/L glucose. The maximum DHA productivity with 60 g/L of pure glucose was 1.10 ± 0.16 g/L/d and 3.23 ± 0.31 g/L/d in flask and bioreactor cultivation, respectively. Pure glucose was replaced with organosolv-pretreated spruce hydrolysate (OPSH) to test the cost-effective production of nutraceuticals. In this case, biomass and lipid productivity were 9.69 ± 0.34 and 5.07 ± 0.13 g/L/d, respectively, when using a bioreactor. Similarly, bioreactor cultivation doubled the DHA content to 66.72 ± 0.31% w/w total lipids (10.15 g/L) and squalene content to 0.93 g/L, as compared to flask cultivation. The DHA productivity with OPSH containing 60 g/L glucose was 1.17 ± 0.12 g/L/d and 3.38 ± 0.27 g/L/d in flask and bioreactor cultivation, respectively. Importantly, we show that S. limacinum SR21 has great expertise to utilize as commercial strain for simultaneous production of DHA and squalene.
Methods
Microbial strain and cultivation conditions
The marine thraustochytrid S. limacinum SR21 (ATCC-MYA-1381) was procured from the American Type Culture Collection and was cultured in ATCC 790 By + medium containing yeast extract (1 g), peptone (1 g), and glucose (5 g) in artificial seawater (1000 mL). Medium pH was adjusted to 6.8 with 1 N HCl before sterilization. The content of artificial seawater was similar as reported in our previous studies68.
Seed culture preparation
The seed culture of S. limacinum was cultivated in the above medium in 250-mL Erlenmeyer flasks with 100 mL working solution. The flasks were incubated for 48 h in an incubator at 25 °C with 180 rpm.
Batch cultivation in Erlenmeyer flasks
Cultivation trials were carried out in Erlenmeyer flask (500-mL) with the 100 mL of working solution. To optimize the concentration of artificial seawater in the growth medium, the latter contained 30 g/L of glucose in 25%, 50%, 75%, 100% (v/v) artificial seawater adjusted with distilled water. Yeast extract was used as organic N source at a C/N (g/g) ratio of 10; this was based on the assumption that yeast extract represented 11.6% of total N with 6.2% amino N (Sigma-Aldrich, St. Louis, MO, USA). It has been already reported that the yeast extract (which contains 9–12% w/w of total N) obtained from brewery industries after processing with spent yeast biomass is utilized for cultivation of thraustochytrids69. To determine the effect of various C/N ratios (10, 20, 30, 40, 50), the appropriate amount of yeast extract was added together with 30 g/L of glucose. After selecting a suitable C/N ratio for maximum lipid production, various amounts of glucose (30, 60, 90, 120 g/L) were tested. To avoid Maillard’s reaction between amino acids in the yeast extract and reducing sugars, glucose was always autoclaved separately. Medium pH was adjusted to 6.8 with 1 N NaOH and 1 N HCl prior to autoclaving. After inoculation of medium with seed culture, the fermentation experiment was carried out in an incubator shaker with 180 rpm at 25 °C and sampling was done at every 24 h of cultivation for the analysis of growth and residual sugars.
Batch cultivation in a bioreactor
Batch cultivation was carried out in a 3-L BioBundle bioreactor equipped with the ez2 control bundle (Applikon Biotechnology, JG Delft, The Netherlands) with 1 L working volume. The bioreactor was maintained at 25 °C and pH was adjusted with 3 N NaOH and 3 N HCl. Different aeration rates (0.66, 1.33, 2, 2.66 vvm) with fixed agitation speed at 300 rpm were tried to optimize biomass growth and lipid production by S. limacinum. To avoid excess moisture loss from the medium during cultivations with high aeration rate, a 5-L humidifier bottle was connected to the inlet air between air hose and cultivation vessel, and the condenser of the reactor was connected with an external thermostatic bath circulating with 90% glycol at 5 °C. After optimization of the aeration rate, a time-course experiment tested various amounts of glucose (30, 60, 90, 120 g/L) to study, by harvesting 50-mL samples every 24 h.
Batch cultivation of S. limacinum with organosolv-pretreated spruce hydrolysate (OPSH)
Norway spruce chips obtained from mills in Northern Sweden was crushed into fine powder in a Retsch SM 300 knife mill (Retsch GmbH, Haan, Germany). The milled chips were subjected to pretreatment with hybrid organosolv-steam explosion method according to our optimized method70. The pretreated solids was obtained with high cellulose content (72%, w/w) including low hemicellulose (4%) and lignin (15.4%)70. The pretreated solids (10% of w/w) were hydrolyzed by commercial enzyme Cellic CTec2 (Novozymes A/S, Bagsværd, Denmark) at 20 FPU/g of solids in 50 mM citrate-phosphate buffer (pH 5) at 50 °C for 48 h.
The appropriate amount of hydrolysate was added to 50% v/v artificial seawater to adjust the glucose content at 60 g/L and finally, C/N ratio was maintained at 10 with yeast extract. The pH of medium was adjusted at 6.8. Cultivation experiments were carried out in Erlenmeyer flasks and bioreactor under optimized cultivation parameters as mentioned above.
Analytical methods
Samples from Erlenmeyer flasks (15 mL) and bioreactor (50 mL) were aspirated off every 24 h to determine the growth, biomass, lipid accumulation and utilization of carbon source. The harvested samples were centrifuged at 8000 rpm for 10 min, and the supernatant was used for residual sugars analysis by HPLC equipped with Aminex HPX-87H column (Bio-Rad, Hercules, CA, USA) according to our established protocol71. The cell pellets were dried in a hot air oven at 40 °C till constant weight and the dry cell weight (DCW; g/L) was measured gravimetrically by weighing balance.
The oven-dried pellets were then pulverized into fine powder by using mortar and pestle and blended with a solution of chloroform and methanol (2:1, v/v). The slurry was transferred in screwed capped glass tubes followed by incubation under mild shaking at room temperature for 2 h. After that the slurry was mixed with ½ volume of water and allowed still for phase separation. The clear bottom layer of chloroform was aspirated in preweighed glass vials, the solvent was evaporated and the total lipids (g/L) were determined gravimetrically.
For the determination of squalene, the extracted lipids were dissolved in absolute acetonitrile and analyzed by HPLC (PerkinElmer, Waltham, MA, USA) equipped with C18 reverse-phase column (MACHEREY-NAGEL GmbH & Co. KG, Düren, Germany) using acetonitrile: water (9:1, v/v) as mobile phase with 1.5 mL/min flow rate at 30 °C. The quantification of squalene was done at UV detector at 210 nm with calibration curve prepared by standard solution of squalene with a range of 0.001 mg/mL to 10 mg/mL (Sigma-Aldrich, St. Louis, MO, USA). The extraction of squalene from total lipid was carried out by saponification method mentioned by Nakazawa et al.65. The squalene was separated as unsaponifiable fraction from the saponifiable lipid fraction. The purity of both fractions was analyzed by thin-layer chromatography (TLC). The fractions were spotted on silica gel 60 F254 normal phase plates (Merk, Dermstadt, Germany) and chromatogram was developed by using of n-hexane: diethyl ether: acetic acid (85:15:1; v/v/v) as mobile phase. The after spraying the methanolic MnCl2 solution (MnCl2.4H2O, 0.32 g; water, 30 mL; methanol, 30 mL, and H2SO4, 4 mL). After drying the plates in air, spots were visualized by charring of plates in hot air oven at 125 °C for 5 min. Triolein and pure squalene were used as standard.
The total lipids were transesterified for the estimation of fatty acids profile by using acid catalysts as described previously68. The fatty acid methyl esters (FAMEs) were estimated by GC-FID (Agilent, Santa Clara, CA, USA) equipped with capillary column Select FAME; dimensions 50 m × 0.25 mm ID and 0.25 μm film thickness. The quantification of individual peaks was carried out by using standard mixture of FAME, Supelco 37 Component FAME Mix (47885-U, Sigma-Aldrich).
Statistical analysis
All experiments were carried out in triplicates to achieve the average value and errors bars by ± standard deviation. One-way analysis of variance (ANOVA) with p < 0.05 for data acceptance was performed with Microsoft Office Excel 2016 (Microsoft, USA).
References
Cho, H. P., Nakamura, M. & Clarke, S. D. Cloning, Expression, and Fatty Acid Regulation of the Human ∆ -5 Desaturase Cloning, Expression, and Fatty Acid Regulation of the Human ⌬-5 Desaturase. J. Biol. Chem. 274, 37335–37339 (1999).
Lands, B. Historical perspectives on the impact of n-3 and n-6 nutrients on health. Prog. Lipid Res. 55, 17–29 (2014).
Shahidi, F. & Wanasundara, U. N. Omega-3 fatty acid concentrates: Nutritional aspects and production technologies. Trends Food Sci. Technol. 9, 230–240 (1998).
Zárate, R., el Jaber-Vazdekis, N., Tejera, N., Pérez, J. A. & Rodríguez, C. Significance of long chain polyunsaturated fatty acids in human health. Clin. Transl. Med. 6, 25 (2017).
Ling, X. et al. Impact of carbon and nitrogen feeding strategy on high production of biomass and docosahexaenoic acid (DHA) by Schizochytrium sp. LU310. Bioresour. Technol. 184, 139–147 (2015).
Berliner, D. et al. The omega-3 index in patients with heart failure: A prospective cohort study. Prostaglandins Leukot. Essent. Fat. Acids 140, 34–41 (2019).
Rubio-Rodríguez, N. et al. Production of omega-3 polyunsaturated fatty acid concentrates: A review. Innov. Food Sci. Emerg. Technol. 11, 1–12 (2010).
Ward, O. P. & Singh, A. Omega-3/6 fatty acids: Alternative sources of production. Process Biochem. 40, 3627–3652 (2005).
Scheben, A. & Edwards, D. Bottlenecks for genome-edited crops on the road from lab to farm. Genome Biol. 19, 178 (2018).
Chung, Y. S., Lee, J. W. & Chung, C. H. Molecular challenges in microalgae towards cost-effective production of quality biodiesel. Renew. Sustain. Energy Rev. 74, 139–144 (2017).
Khan, M. I., Shin, J. H. & Kim, J. D. The promising future of microalgae: Current status, challenges, and optimization of a sustainable and renewable industry for biofuels, feed, and other products. Microb. Cell Fact. 17, 1–21 (2018).
Leyland, B., Leu, S. & Boussiba, S. Are Thraustochytrids algae? Fungal Biol. 121, 835–840 (2017).
Raghukumar, S. Thraustochytrid Marine Protists: Production of PUFAs and Other Emerging Technologies. 631–640, https://doi.org/10.1007/s10126-008-9135-4 (2008).
Skogsmarkens kolförråd. Forest statistics 2017, Aktuella uppgifter om de svenska skogarna från Riksskogstaxeringen (2017).
Selvaraj, M., Kumar, T. S. & Rao, M. V. Squalene, Biosynthesis and its role in production of bioactive compounds, a Proper Scientific Challenge – A Review. J. Emerg. Technol. Innov. Res. 6, 505–526 (2019).
Gohil, N., Bhattacharjee, G., Khambhati, K., Braddick, D. & Singh, V. Engineering Strategies in Microorganisms for the Enhanced Production of Squalene: Advances, Challenges and Opportunities. Front. Bioeng. Biotechnol. 7, 1–24 (2019).
Pollier, J. et al. A widespread alternative squalene epoxidase participates in eukaryote steroid biosynthesis. Nat. Microbiol. 4, 226–233 (2019).
Garcia-Bermudez, J. et al. Squalene accumulation in cholesterol auxotrophic lymphomas prevents oxidative cell death. Nature 567, 118–122 (2019).
Brown, A. J., Chua, N. K. & Yan, N. The shape of human squalene epoxidase expands the arsenal against cancer. Nat. Commun. 10, 2–5 (2019).
Reddy, L. H. & Couvreur, P. Squalene: A natural triterpene for use in disease management and therapy. Adv. Drug Deliv. Rev. 61, 1412–1426 (2009).
Güneş, F. E. Medical use of squalene as a natural antioxidant. J. Marmara Univ. Inst. Heal. Sci. 3, 220–228 (2013).
Oya, S. I. et al. Catalytic Production of Branched Small Alkanes from Biohydrocarbons. Chem. Sus. Chem. 8, 2472–2475 (2015).
Zhang, K., Zhang, X. & Tan, T. The production of bio-jet fuel from: Botryococcus braunii liquid over a Ru/CeO2 catalyst. RSC Adv. 6, 99842–99850 (2016).
Rosales-Garcia, T., Jimenez-Martinez, C. & Davila-Ortiz, G. Squalene Extraction: Biological Sources and Extraction Methods. Int. J. Environ. Agric. Biotechnol. 2, 1662–1670 (2017).
Popa, O., Bəbeanu, N. E., Popa, I., Niţə, S. & Dinu-Pârvu, C. E. Methods for obtaining and determination of squalene from natural sources. Biomed Res. Int. 2015 (2015).
Xie, Y., Sen, B. & Wang, G. Mining terpenoids production and biosynthetic pathway in thraustochytrids. Bioresour. Technol. 244, 1269–1280 (2017).
Kim, K. et al. A novel fed-batch process based on the biology of Aurantiochytrium sp. KRS101 for the production of biodiesel and docosahexaenoic acid. Bioresour. Technol. 135, 269–274 (2013).
Patel, A., Pruthi, V. & Pruthi, P. A. Synchronized nutrient stress conditions trigger the diversion of CDP-DG pathway of phospholipids synthesis towards de novo TAG synthesis in oleaginous yeast escalating biodiesel production. Energy 139, 962–974 (2017).
Huang, T. Y., Lu, W. C. & Chu, I. M. A fermentation strategy for producing docosahexaenoic acid in Aurantiochytrium limacinum SR21 and increasing C22:6 proportions in total fatty acid. Bioresour. Technol. 123, 8–14 (2012).
Abad, S. & Turon, X. Biotechnological production of docosahexaenoic acid using aurantiochytrium limacinum: Carbon sources comparison and growth characterization. Mar. Drugs 13, 7275–7284 (2015).
Wong, M. K. M., Tsui, C. K. M., Au, D. W. T. & Vrijmoed, L. L. P. Docosahexaenoic acid production and ultrastructure of the thraustochytrid Aurantiochytrium mangrovei MP2 under high glucose concentrations. Mycoscience 49, 266–270 (2008).
Ganuza, E., Yang, S., Amezquita, M., Giraldo-Silva, A. & Andersen, R. A. Genomics, Biology and Phylogeny Aurantiochytrium acetophilum sp. nov. (Thraustrochytriaceae), Including First Evidence of Sexual Reproduction. Protist 170, 209–232 (2019).
Chi, Z., Pyle, D., Wen, Z., Frear, C. & Chen, S. A laboratory study of producing docosahexaenoic acid from biodiesel-waste glycerol by microalgal fermentation. Process Biochem. 42, 1537–1545 (2007).
Patil, K. P. & Gogate, P. R. Improved synthesis of docosahexaenoic acid (DHA) using Schizochytrium limacinum SR21 and sustainable media. Chem. Eng. J. 268, 187–196 (2015).
Yokochi, T., Honda, D., Higashihara, T. & Nakahara, T. Optimization of docosahexaenoic acid production by Schizochytrium limacinum SR21. Appl. Microbiol. Biotechnol. 49, 72–76 (1998).
Li, J. et al. A strategy for the highly efficient production of docosahexaenoic acid by Aurantiochytrium limacinum SR21 using glucose and glycerol as the mixed carbon sources. Bioresour. Technol. 177, 51–57 (2015).
Shuler, M. L. & Kargi, F. Bioprocess Engineering: Basic Concepts. Prentice Hall 2, (2002).
Dosoretz, C. G., Chen, A. H. & Grethlein, H. E. Applied Microbiology Biotechnology Effect of oxygenation conditions on submerged cultures of Phanerochaete ¢ so porium. 131–137 (1990).
Alfenore, S. et al. Aeration strategy: A need for very high ethanol performance in Saccharomyces cerevisiae fed-batch process. Appl. Microbiol. Biotechnol. 63, 537–542 (2004).
Ratledge, C. Fatty acid biosynthesis in microorganisms being used for Single Cell Oil production. Biochimie 86, 807–15 (2004).
Jakobsen, A. N., Aasen, I. M., Josefsen, K. D. & Strøm, A. R. Accumulation of docosahexaenoic acid-rich lipid in thraustochytrid Aurantiochytrium sp. strain T66: effects of N and P starvation and O2 limitation. Appl. Microbiol. Biotechnol. 80, 297–306 (2008).
Qiu, X. Biosynthesis of docosahexaenoic acid (DHA, 22:6-4, 7,10,13,16,19): Two distinct pathways. Prostaglandins Leukot. Essent. Fat. Acids 68, 181–186 (2003).
Oboh, A. et al. Two alternative pathways for docosahexaenoic acid (DHA, 22:6n-3) biosynthesis are widespread among teleost fish. Sci. Rep. 7, 1–10 (2017).
Qiu, X., Hong, H. & MacKenzie, S. L. Identification of a Δ4 Fatty Acid Desaturase from Thraustochytrium sp. Involved in the Biosynthesis of Docosahexanoic Acid by Heterologous Expression in Saccharomyces cerevisiae and Brassica juncea. J. Biol. Chem. 276, 31561–31566 (2001).
Chang, G. et al. Improvement of docosahexaenoic acid production on glycerol by Schizochytrium sp. S31 with constantly high oxygen transfer coefficient. Bioresour. Technol. 142, 400–406 (2013).
Ye, C. et al. Reconstruction and analysis of the genome-scale metabolic model of schizochytrium limacinum SR21 for docosahexaenoic acid production. BMC Genomics 16, 1–11 (2015).
Wallis, J. G., Watts, J. L. & Browse, J. Polyunsaturated fatty acid synthesis: What will they think of next? Trends Biochem. Sci. 27, 467–473 (2002).
Metz, J. G. et al. Production of polyunsaturated fatty acids by potyketide synthases in both prokaryotes and eukaryotes. Science (80-.). 293, 290–293 (2001).
Chen, C. Y. & Yang, Y. T. Combining engineering strategies and fermentation technology to enhance docosahexaenoic acid (DHA) production from an indigenous Thraustochytrium sp. BM2 strain. Biochem. Eng. J. 133, 179–185 (2018).
Iida, I. et al. Improvement of docosahexaenoic acid production in a culture of Thraustochytrium aureum by medium optimization. J. Ferment. Bioeng. 81, 76–78 (1996).
Nakahara, T. et al. Production of docosahexaenoic and docosapentaenoic acids by Schizochytrium sp. isolated from yap islands. JAOCS, J. Am. Oil Chem. Soc. 73, 1421–1426 (1996).
Barclay, W. R., Meager, K. M. & Abril, J. R. Heterotrophic production of long chain omega-3 fatty acids utilizing algae and algae-like microorganisms. J. Appl. Phycol. 6, 123–129 (1994).
Yin, F.-W. et al. Development of a real-time bioprocess monitoring method for docosahexaenoic acid production by Schizochytrium sp. Bioresour. Technol. 216, 422–427 (2016).
Hauvermale, A. et al. Fatty acid production in Schizochytrium sp.: Involvement of a polyunsaturated fatty acid synthase and a type I fatty acid synthase. Lipids 41, 739–747 (2006).
Metz, J. G. Production of Polyunsaturated Fatty Acids by Polyketide Synthases in Both Prokaryotes and Eukaryotes. Science (80-.). 293, 290–293 (2001).
Chi, Z., Liu, Y., Frear, C. & Chen, S. Study of a two-stage growth of DHA-producing marine algae Schizochytrium limacinum SR21 with shifting dissolved oxygen level. Appl. Microbiol. Biotechnol. 81, 1141–1148 (2009).
Patel, A., Matsakas, L., Rova, U. & Christakopoulos, P. Heterotrophic cultivation of Auxenochlorella protothecoides using forest biomass as a feedstock for sustainable biodiesel production. Biotechnol. Biofuels 11, 169 (2018).
Wu, S. T., Yu, S. T. & Lin, L. P. Effect of culture conditions on docosahexaenoic acid production by Schizochytrium sp. S31. Process Biochem. 40, 3103–3108 (2005).
Safdar, W., Zan, X. & Song, Y. Synergistic Effect of Phosphorus and Nitrogen on Growth, Lipid Accumulation and Docosahexaenoic Acid Production in Crypthecodinium Cohnii. Int. J. Agric. Innov. Res. 5, 768–775 (2017).
Chang, G. et al. Fatty acid shifts and metabolic activity changes of Schizochytrium sp. S31 cultured on glycerol. Bioresour. Technol. 142, 255–260 (2013).
Liang, Y. et al. Use of sweet sorghum juice for lipid production by Schizochytrium limacinum SR21. Bioresour. Technol. 101, 3623–3627 (2010).
Burja, A. M., Radianingtyas, H., Windust, A. & Barrow, C. J. Isolation and characterization of polyunsaturated fatty acid producing Thraustochytrium species: Screening of strains and optimization of omega-3 production. Appl. Microbiol. Biotechnol. 72, 1161–1169 (2006).
Armenta, R. E., Burja, A., Radianingtyas, H. & Barrow, C. J. Critical assessment of various techniques for the extraction of carotenoids and co-enzyme Q10 from the thraustochytrid strain ONC-T18. J. Agric. Food Chem. 54, 9752–9758 (2006).
Jiang, Y., Fan, K.-W., Tsz-Yeung Wong, R. & Chen, F. Fatty Acid Composition and Squalene Content of the Marine Microalga Schizochytrium mangrovei. J. Agric. Food Chem. 52, 1196–1200 (2004).
Nakazawa, A. et al. TLC screening of thraustochytrid strains for squalene production. J. Appl. Phycol. 26, 29–41 (2014).
Yue, C. J. & Jiang, Y. Impact of methyl jasmonate on squalene biosynthesis in microalga Schizochytrium mangrovei. Process Biochem. 44, 923–927 (2009).
Kaya, K. et al. Thraustochytrid Aurantiochytrium sp. 18W-13a Accummulates High Amounts of Squalene. Biosci. Biotechnol. Biochem. 75, 2246–2248 (2011).
Patel, A., Rova, U., Christakopoulos, P. & Matsakas, L. Simultaneous production of DHA and squalene from Aurantiochytrium sp. grown on forest biomass hydrolysates. Biotechnol. Biofuels 12, 1–12 (2019).
Bumbak, F., Cook, S., Zachleder, V., Hauser, S. & Kovar, K. Best practices in heterotrophic high-cell-density microalgal processes: Achievements, potential and possible limitations. Appl. Microbiol. Biotechnol. 91, 31–46 (2011).
Matsakas, L. et al. Lignin-first biomass fractionation using a hybrid organosolv – Steam explosion pretreatment technology improves the saccharification and fermentability of spruce biomass. Bioresour. Technol. 273, 521–528 (2019).
Patel, A., Hrůzová, K., Rova, U., Christakopoulos, P. & Matsakas, L. Sustainable biorefinery concept for biofuel production through holistic volarization of food waste. Bioresour. Technol. 294 (2019).
Ren, L. J. et al. Development of a stepwise aeration control strategy for efficient docosahexaenoic acid production by Schizochytrium sp. Appl. Microbiol. Biotechnol. 87, 1649–1656 (2010).
Acknowledgements
Authors would like to thank Sveaskog, Sweden, for providing the spruce chips that were used in this study and Bio4 Energy, a strategic research environment appointed by the Swedish government, for supporting this work. We would like to acknowledge Kentaro Umeki and Albert Bach Oller from Energy Engineering, Division of Energy Science, Department of Engineering Sciences and Mathematics, Luleå University of Technology, Luleå, Sweden for providing the GC-FID apparatus. Open access funding provided by Lulea University of Technology.
Author information
Authors and Affiliations
Contributions
A.P.: Conceived the study, performed the experimental and analytical work, analyzed the data and drafted the manuscript. S.L.: performed experiments, U.R. and P.C.: conceived the study and participated in experimental design and data analysis. L.M.: Conceived the study, participated in the experimental design, analyzed the data and performed the organosolv pretreatment.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Patel, A., Liefeldt, S., Rova, U. et al. Co-production of DHA and squalene by thraustochytrid from forest biomass. Sci Rep 10, 1992 (2020). https://doi.org/10.1038/s41598-020-58728-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-020-58728-7
- Springer Nature Limited
This article is cited by
-
Transcriptomics aids in uncovering the metabolic shifts and molecular machinery of Schizochytrium limacinum during biotransformation of hydrophobic substrates to docosahexaenoic acid
Microbial Cell Factories (2024)
-
Exploring the potential of Thraustochytrids and other microorganisms for sustainable omega-3 production: a comprehensive review of patents, perspectives, and scale-up strategies
Systems Microbiology and Biomanufacturing (2024)
-
Potential and future perspectives of thraustochytrids in bioremediation
International Journal of Environmental Science and Technology (2023)
-
Bioprocess conditions and regulation factors to optimize squalene production in thraustochytrids
World Journal of Microbiology and Biotechnology (2023)
-
Recent Progress in Microalgal Squalene Production and Its Cosmetic Application
Biotechnology and Bioprocess Engineering (2022)