Abstract
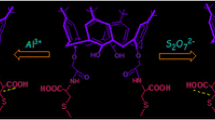
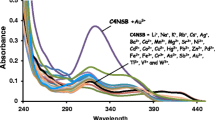
This article reports the selective sensing ability of a newly synthesized calix[4]arene Schiff base (C4TSB) derivative. C4TSB exhibited strong turn-off fluorescence affinity for Hg2+ and Au3+. The selective sensing ability of receptor was investigated in the presence of different co-existing competing ions. The limit of detection for Hg2+ and Au3+ was determined as 1.9 × 10−5 and 1.0 × 10−6 M, respectively. Receptor forms 1:1 stoichiometric complex with both metals and their binding constants were calculated as 7.9 × 103 M−1 for Hg2+ and 5.7 × 103 M−1 for Au3+. Complexes were also characterized through FT-IR spectroscopy.










Similar content being viewed by others
References
A.P. De Silva, H.Q.N. Gunaratne, T. Gunnlaugsson, A.J.M. Huxley, C.P. McCoy, J.T. Rademacher, T.E. Rice, Chem. Rev. 97, 1515 (1997)
J.M. Ilobet, G. Falco, A. Teixido, J.L. Domingo, J. Agric. Food Chem. 51, 838 (2003)
J.J.J. Gutknecht, Mem. Biol. 61, 61 (1981)
M. Tuzen, O.D. Uluozlu, I. Karaman, M. Soylak, J. Hazard. Mater. 169, 345 (2009)
M. Tuzen, A. Sari, D. Mendil, M. Soylak, J. Hazard. Mater. 169, 263 (2009)
T.W. Ckarckson, L. Magos, G.J. Myers, N. Engl, J. Med. 349, 1731 (2003)
C.M.L. Carvalho, E.-H. Chew, S.I. Hashemy, J. Lu, A. Holmgren, J. Biol. Chem. 283, 11913 (2008)
M. Harada, Crit. Rev. Toxicol. 25, 1 (1995)
X. Mao, Y. Ma, A. Zhang, L. Zhang, G. Liu, Anal. Chem. 81, 1660 (2009)
P. Sharma, S.C. Brown, N. Bengtsson, Q. Zhang, G.A. Walter, S.R. Grobmyer, S. Santra, H. Jiang, E.W. Scott, B.M. Moudgil, Chem. Mater. 20, 6087 (2008)
A. Habib, M.J. Tabata, Inorg. Biochem. 98, 1696 (2004)
W.D. Block, E.L. Knapp, J. Pharmacol. Exp. Ther. 83, 275 (1945)
M. Tuzen, K.O. Saygi, M. Soylak, J. Hazard. Mater. 156, 591 (2008)
M. Fujita, Comprehensive Supramolecular Chemistry, vol. 9 (Pergamon Press, Berlin, 1996), p. 253
C. Bazzicalupi, A. Bencini, I. Matera, S. Puccioni, B. Valtancoli, Inorg. Chim. Acta 381, 162 (2012)
A. Bencini, V. Lippolis, Coord. Chem. Rev. 256, 149 (2012)
M.T. Albelda, P. Díaz, E. García-España, J.C. Lima, C. Lodeiro, J.S. De Melo, A.J. Parola, F. Pina, C. Soriano, Chem. Phys. Lett. 353, 63 (2002)
I. Aoki, T. Sakaki, S.J. Shinkai, Chem. Soc. Chem. Commun. 9, 730 (1992)
J.M. Liu, Q.Y. Zheng, J.L. Yang, C.F. Chen, Z.-T. Huang, Tetrahedron Lett. 43, 9209 (2002)
Y.-D. Cao, Q.Y. Zheng, C.F. Chen, Z.T. Huang, Tetrahedron Lett. 44, 4751 (2003)
R. Metivier, I. Leray, B. Valeur, Chem. Eur. J. 10, 4480 (2004)
K.C. Chang, L.Y. Luo, E.W.G. Diau, W.S. Chung, Tetrahedron Lett. 49, 5013 (2008)
I. Leray, B. Valeur, Eur. J. Inorg. Chem. 2009, 3525 (2009)
M. Kumar, J.N. Babu, V. Bhalla, J. Incl. Phenom. Macrocycl. Chem. 66, 139 (2010)
N. Aksuner, E. Henden, I. Yilmaz, A. Cukurovali, Sens. Actuators, B 134, 510 (2008)
N. Aksuner, E. Henden, I. Yilmaz, A. Cukurovali, Dyes Pigm. 83, 211 (2009)
Z. Yang, M. She, J. Zhang, X. Chen, Y. Huang, H. Zhu, P. Liu, J. Li, Z. Shi, Sens. Actuators, B 176, 482 (2013)
L. Yang, W. Zhu, M. Fang, Q. Zhang, C. Li, Spectrochim. Acta, Part A 109, 186 (2013)
M. Kumar, J.N. Babu, V. Bhalla, J. Inclusion Phenom. Macrocycl. Chem. 66, 139 (2010)
L. Tang, F. Li, M. Liu, R. Nandhakumar, Spectrochim. Acta, Part A 78, 1168 (2011)
R.M. Izatt, J.S. Bradshaw, S.A. Nielsen, J.D. Lamb, J.J. Christian, D. Sen, Chem. Rev. 85, 271 (1985)
R.M. Izatt, J.S. Bradshaw, K. Pawlak, J.S. Bradshaw, R.L. Bruening, Chem. Rev. 91, 1721 (1991)
G.G. Talanova, H.S.Hwang,V.S. Talanov, R.A. Bartsch, Chem. Commun. 1329 (1998)
G.G. Talanova, N.S.A. Elkarim, V.S. Talanov, R.A. Bartsch, Anal. Chem. 71, 3106 (1999)
G.U. Akkus, S. Memon, M. Sezgin, M. Yilmaz, Clean 37, 109 (2009)
H.K. Alpoguz, A. Kaya, S. Memon, M. Yilmaz, J. Macromol. Sci. Pure 44, 17 (2007)
Ü. Ocak, M. Ocak, K. Surowiec, X. Liu, R.A. Bartsch, Tetrahedron Lond. 65, 7038 (2009)
S. Memon, A.A. Bhatti, A.A. Bhatti, Ü. Ocak, M. Ocak, J. Fluoresc. 25, 1507 (2015)
S. Memon, A.A. Bhatti, A.A. Bhatti, Ü. Ocak, M. Ocak, J. Iran. Chem. Soc. 12, 1739 (2015)
S. Memon, A.A. Bhatti, Ü. Ocak, M. Ocak, Poly. Arom. Com. 1–15 (2015)
S. Memon, A.A. Bhatti, Ü. Ocak, M. Ocak, Anal. Meth. 7, 5114 (2015)
M.A. Qazi, I. Qureshi, S. Memon, J Fluoresc. 21, 1703 (2011)
M.A. Qazi, Ü. Ocak, M. Ocak, S. Memon, I.B. Solangi, J. Fluores. 23, 575 (2013)
M.A. Qazi, I. Qureshi, S. Memon, J. Fluoresc. 21, 1231 (2011)
I.B. Solangi, A.A. Bhatti, M.A. Qazi, S. Memon, M.I. Bhanger, Pak. J. Anal. Environ. Chem. 13, 129 (2012)
C.D. Gutsche, M. Iqbal, D. Stewart, J. Org. Chem. 51, 742 (1986)
D. Maity, A. Chakraborty, R. Gunupuruand, P. Paul, Inorg. Chim. Acta 372, 126 (2011)
X. Chen, R.P.M. Dings, I. Nesmelova, S. Debbert, J.R. Haseman, J. Maxwell, T.R. Hoye, K.H. Mayo, J. Med. Chem. 49, 7754 (2006)
Z. Liang, Z. Liu, Y. Gao, Spectrochim. Acta. A. 68, 1231 (2007)
C.F. Chow, M.H.W. Lam, W.Y. Wong, Inorg. Chem. 43, 8387 (2004)
H.A. Benesi, J.H. Hildebrand, J. Am. Chem. Soc. 71, 2703 (1949)
D.C. Harris, Quantitative Chemical Analysis, 4th edn. (W.H. Freeman & Company, New York, 1995)
Ü. Ocak, M. Ocak, K. Surowiec, X. Liu, R.A. Bartsch, Tetrahedron 65, 7038 (2009)
Acknowledgments
We thank the National Center of Excellence in Analytical Chemistry, University of Sindh, Jamshoro/Pakistan and Scientific and Technological Research Council of Turkey (TUBITAK, B.02.1.TBT.0.06.01-216.01/895–6391) for the financial support of this work.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Memon, S., Bhatti, A.A., Bhatti, A.A. et al. A new calix[4]arene Schiff base sensor for Hg2+ and Au3+ . J IRAN CHEM SOC 13, 2275–2282 (2016). https://doi.org/10.1007/s13738-016-0946-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13738-016-0946-3