Abstract
This study investigates the freezing tolerance and cold-induced changes in amino acid metabolism in three Triticeae species (rye, wheat, and barley) with varying levels of freezing tolerance. Freezing tests confirmed that rye exhibited the highest tolerance, while barley showed the highest sensitivity. Cold acclimation significantly increased total free amino acid levels, with wheat and barley showing nearly twice the accumulation compared to rye. The glutamate family of amino acids, particularly proline (Pro), γ-aminobutyric acid (GABA), and glutamine (Gln), displayed substantial increase during cold treatment. Pro levels were notably higher in freezing-tolerant wheat and barley genotypes, suggesting its role in osmotic stress mitigation. However, this correlation was absent in rye. Gene expression analysis revealed that cold-induced proline accumulation is likely regulated at the post-transcriptional level, particularly involving the P5CS gene. These findings highlight the species-specific metabolic adjustments and regulatory mechanisms underlying freezing tolerance in Triticeae species, emphasizing the central role of proline and glutamate family amino acids in cold acclimation.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The changing climate poses a threat to global food security. Late spring frosts in temperate climate can result in severe damage and a reduction in the yield of cereals (Lamichhane 2021). It is evident that winter frosts cause severe damage and, consequently, yield loss for winter cereals. Their freezing tolerance increases during cold acclimation, which is induced by the gradual decrease in temperature during autumn. A coordinated reprogramming of gene expression and metabolism occurs which ensures their adjustment to the new environmental conditions (Kaplan et al. 2007; Zhu et al. 2007).
Transcriptome analysis revealed that the expression of many genes related to amino acid metabolism changed during cold acclimation. In Arabidopsis, the expression of genes related to the metabolism of 13 amino acids was affected by short-term (3–4 d) cold treatment (Kaplan et al. 2007; Usadel et al. 2008). For instance, the upregulation of Proline (Pro) biosynthesis-related genes was observed after cold acclimation in wheat seedlings with the downregulation of ProDH genes that are involved in Pro degradation (Zhao et al. 2019). Similar results were obtained in the case of beans as well, as cold-induced the expression of Pro synthesis related Δ-1-pyrroline-5-carboxylate synthetase (P5CS) gene (Chen et al. 2009). In barley and wheat, transcript levels related to Pro, Glu, and Gly metabolism were affected as shown by transcriptome profiling (Svensson et al. 2006; Kocsy et al. 2010) which results were also confirmed by qRT-PCR for Pro and Glu in wheat (Kovács et al. 2011). Transcriptome analysis revealed the cold induction of threonine synthase and asparaginase genes: the change of the latter one was greater in the freezing-tolerant wheat genotype compared to the sensitive one (Monroy et al. 2007). Cold also induced the accumulation of Glu by inducing the expression of Glu synthetase levels in five differently moderately tolerant Chinese Spring wheat lines (Ahad et al. 2023). These results indicate that cold-induced changes in amino acid concentration may derive from the activation of the related genes.
The amount of Ala, Arg, Asp, Asn, Glu, Gln, Gaba (γ-amino-butyrate), His, Ile, Leu, Lys, Phe, Pro, Thr, Trp, Tyr, and Val changed at low temperatures which derived from the corresponding alterations at the transcript and enzyme activity levels in Arabidopsis (Usadel et al. 2008). Thus, cold affected the activity of nitrate reductase, Gln and Glu synthase, Ala and Asp 2-oxoglutarate transaminase, and shikimate dehydrogenase. Similarly, significant accumulation of Ala, Pro, Leu, Phe and Val was observed in tobacco seedlings after 6 h of cold treatment (Xu et al. 2020). Moreover, cold-induced the expression of genes encoding β-alanine aminotransferase and glutamate decarboxylase in cold-tolerant maize, leading to the accumulation of osmoprotectants such as β-alanine betaine and GABA (Waititu et al. 2021). However, cold-induced changes related to amino acids are not always coordinated at transcript and metabolite levels as shown in a 4-day time-course experiment in Arabidopsis (Kaplan et al. 2007). This observation indicates the presence of regulatory processes being independent of transcript abundance. However, the gradual increase in the amount of the amino acids having a common precursor was very similar in this experimental system which suggested the co-ordinated control of the synthesis of members of a certain amino acid family. During a 3-week cold hardening period the amount of Asp, Glu, Gln, and Pro increased in wheat and a corresponding change was found for Glu and Pro at the transcript level (Kovács et al. 2011). A relationship between Pro levels and freezing tolerance was also observed in wheat (Kovács et al. 2011). Likewise, overexpression of Gln synthase affected cold tolerance in rice (Cai et al. 2009).
In order to clarify whether the cold acclimation-induced changes of the free amino acids at metabolite and gene expression levels have a relationship with freezing tolerance, these parameters were compared in three freezing-tolerant and three sensitive genotypes of three cereal species viz. winter rye, wheat, and barley.
Materials and methods
Plant material and cold hardening
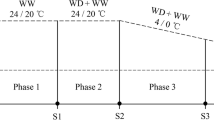
Our studies involved three cereal species: winter rye (Secale cereale L.) with genotypes LP1, LP2, and LP4 as freezing-tolerant ones and LP5, LP3, and LP6 (lines from Lochow-Petkus GmbH, Germany, Baruth) as freezing sensitive ones; winter wheat (Triticum aestivum L.) with genotypes Miranovskaja (Mir), Cheyenne (Che) and Uljanovka (Ulj) as freezing-tolerant ones and GK Tiszatáj (GKT), Capelle Desprez (CD) and Martonvásári 8 (Mv8) as freezing-sensitive ones (from Martonvásár Cereal Gene Bank); winter barley (Hordeum vulgare L.) with genotypes Hundred (Hun), Barbinak (Bar) and Kold (Kol) as freezing tolerant ones and Baishishek (Bai), Rex, Rodnik (Rod) as freezing sensitive ones (from Martonvásár Cereal Gene Bank) as shown in Fig. 1. The information about the level of freezing tolerance of the various genotypes derived from the provider (from Lochow-Petkus GmbH for rye; and from the Martonvásár Cereal Gene Bank for barley and wheat), and was confirmed in our present freezing tests. To initiate germination, the seeds were placed between wet filter papers and kept at a temperature of 25 °C for 1 day, followed by 4 °C for 3 days, and then again at 25 °C for 2 days. The seedlings were then cultivated on half-strength Hoagland medium for 10 days under specific growth conditions until reaching the 2-leaf stage (Hoagland and Arnon 1950). These conditions included a day/night temperature of 18/15 °C, a relative humidity of 75%, and 16 h of illumination with a photosynthetic photon flux density of 270 µmol m−2 s−1 in growth chambers. To induce cold acclimation for a period of 3 weeks, the temperature was consistently maintained at 2 °C, while the other growth conditions remained unchanged (Suppl. Fig. 1). Biochemical measurements were taken after 0, 1, 3, 7 and 21 days during the mid-point of the photoperiod, with three independent replications for each measurement.
Freezing tolerance of the investigated genotypes. 0: no re-growth after freezing, 5: very good re-growth. A rye (LP1-LP6 are lines from Lochow-Petkus GmbH, Germany, Baruth) frozen at − 21 °C, B: wheat (Mir: Miranoskaja, Che: Cheyenne, Ulj: Uljanovka, GKT: GK Tiszatáj, CD: Capelle Desprez, Mv8: Martonvásári 8 variety) frozen at − 15 °C, C: barley (Hun: Hundred, Bar: Barbinak, Kol: Kold, Bai: Baishishek, Rex: Rex, Rod: Rodnik variety) frozen at − 13 °C. The data were subjected to a general linear model for multivariate analysis of variance using SPSS package version 16.0. Tukey’s HSD test was performed to assess the significance of differences between the genotypes (three biological replicates with 10 plants). Values indicated with different letters are significantly different at p ≤ 0.05 level.
Freezing test
After the germination stage, the seedlings were transferred and planted in wooden boxes that were filled with a mixture of garden soil, humus, and sand in a ratio of 2:1:1 (volume:volume:volume). These seedlings were then cultivated for a period of 25 days. Subsequently, a cold-hardening process was implemented using the "FDA" program, as described by Tischner et al. 1997. The rye plants were subjected to freezing temperatures of either − 21 °C or − 19 °C, the wheat plants to − 15 °C or − 12 °C, and the barley plants to − 13 °C or − 10 °C for a duration of 12 h. Following this freezing treatment, the leaves of the plants were trimmed off, and the plants were allowed to regrow for 14 days at a temperature of 18/15 °C. The regrowth of the plants was evaluated using a scale ranging from 0 (no regrowth) to 5 (excellent regrowth) (Vágújfalvi et al. 1999).
Determination of free amino acid content
Shoot samples weighing 500 mg in fresh weight were crushed in liquid nitrogen. The samples were then extracted using 2 ml of cold 10% trichloroacetic acid for a duration of 1 h. The extraction process involved gentle agitation on a shaker (C. Gerhardt GmbH & Co., Germany), carried out at room temperature. Following extraction, each sample was filtered through a 0.2 μm pore membrane filter (Sartorius AG, Germany). The biochemical analysis was performed using an automatic amino acid analyzer (Ingos Ltd., Czech Republic). The analyzer was equipped with an Ionex Ostion LCP5020 cation-exchange column measuring 22 × 0.37 cm. Separation of free amino acids was achieved through stepwise gradient elution utilizing a Li+-citric buffer system (Ingos Ltd., Czech Republic). Colorimetric detection of the amino acids was conducted at wavelengths of 570 nm and 440 nm (for Pro) following post-column derivatization with ninhydrin reagent.
cDNA macroarray analysis and data collection
For macroarray analysis, we utilized the shoots of the plants (they were 17 d old by the start of the experiment and 38 d old at its end) that had undergone cold hardening for different durations (0, 1, 7, or 21 days at 2 °C). Total RNA extraction was carried out using TRIzol reagent (Invitrogen), and 12 µg of RNA from each biological replicate were combined to create a single hybridization sample. mRNA isolation was performed using the Dynabeads mRNA Purification Kit from Dynal (Oslo, Norway). To generate 33P-labeled second-strand cDNA probes, we followed the procedures outlined in a previous study (Zierold et al. 2005), with the exception of employing less stringent washing conditions (1 × SSC, 0.1% SDS; 0.5 × SSC, 0.1% SDS) for rye and wheat samples. The barley cDNA-macroarray utilized in the analysis contained over 13,000 PCR-amplified cDNA clones, arranged in 3 subarrays. After hybridization, the membranes were exposed to screens (BAS-III FujiFilm, Tokyo, Japan) for 24 h, and the signals were detected using a phosphoimager (FLA-3000, FujiFilm). Signal intensities were evaluated using Array Vision software (v8.0, Amersham Bioscience, Little Chalfont, UK). The background signal was determined by measuring a ring surrounding the spots (distance: 1 pixel, thickness: 1 pixel), and the measured background signal was subtracted from the spot intensities. The background-subtracted signals were quantile-normalized (Bolstad et al. 2003), and any double spots were averaged. Quality control measures were implemented by calculating the ratio of signal intensities from double spots, and features with a ratio exceeding 2 were excluded from the analysis. Additionally, spotted features with signal intensities below 2 times the background were also excluded. The experimental design, raw data, and normalized data were deposited in the Gene Expression Omnibus database under accession number GSE47882.
Data analysis
The freezing test data and amino acid measurements were subjected to a general linear model for multivariate analysis of variance to show the general effect of genotype, temperature and their interaction. Two biological replicates with three technical repetitions were used for the analyses. To analyze the cDNA macroarray features, linear models were applied using the Bionconductor LIMMA package (Ritchie et al. 2015), and p-values were adjusted using the Benjamini–Hochberg false discovery rate procedure. Data analysis was performed using the statistical programming language R (Version 2.11). Differentially expressed genes were identified using a significance threshold of 0.05 and a minimum log2-fold change of ± 1. The sequences were annotated using the Mercator pipeline (Lohse et al. 2014) with default parameters.
Results
Freezing tolerance
Three Triticeae species with different levels of freezing tolerance were compared. For this purpose, the most tolerant rye was frozen at the lowest temperatures (− 21 °C and − 19 °C) and the most sensitive barley at the highest temperatures (− 13 °C and − 10 °C) based on previous results (Campoli et al. 2009). The freezing test showed that there was a great difference in the freezing tolerance between the three sensitive and tolerant genotypes in the case of wheat and barley, while there was a continuous transition in this parameter between the three tolerant and three sensitive rye genotypes (Fig. 1).
Changes in the total free amino acid level and the ratios of amino acid families
The average total free amino acid level exhibited a gradual increase during the 3-week hardening period in the three Triticeae species (Fig. 2). Although the starting values were similar in all 3 species, the maximum values were nearly two-fold greater in wheat and barley compared to rye. This difference resulted from the far larger accumulation of free amino acids after 7 and 21 days of hardening in wheat and barley. Although there were differences between the individual genotypes in the time course of the changes in the total free amino acid levels, these alterations did not have any relationship with their freezing tolerance (Fig. 2).
Effect of cold hardening on total free amino acid (TFAA) concentration. Sampling was done after 0 (control), 1, 3, 7, and 21 days of cold hardening. A: rye (LP1-LP6 are lines from Lochow-Petkus GmbH, Germany, Baruth), B: wheat (Mir: Miranoskaja, Che: Cheyenne, Ulj: Uljanovka, GKT: GK Tiszatáj, CD: Capelle Desprez, Mv8: Martonvásári 8 variety), C: barley (Hun: Hundred, Bar: Barbinak, Kol: Kold, Bai: Baishishek, Rex: Rex, Rod: Rodnik variety). The data were subjected to a general linear model for multivariate analysis of variance using SPSS package version 16.0. Tukey’s HSD test was performed to assess the significance of differences between the genotypes (two biological replicates with three parallels). Values indicated with different letters are significantly different at p ≤ 0.05 level
Cold acclimation induced great changes not only in the total free amino acid levels but also in the ratio of the amino acids belonging to the individual families. The glutamate family was the most important since about 50% of the total free amino acid content was covered by its components in all 3 species (Fig. 3, Suppl. Figs. 2–4). Its ratio increased to 60–80%, while the ratio of other families (aspartate, pyruvate, serine, aromatic amino acids) decreased at 2 °C. Similar to the total free amino acid levels, these changes could not be related to the level of freezing tolerance of the individual genotypes.
Cold-induced changes in the ratio of amino acid families in the most tolerant rye, wheat, and barley genotypes. Sampling was done after 0 (control), 1, 3, 7, and 21 days of cold hardening. Arom: aromatic family, Ser: serine family, Pyr: pyruvate family, Asp: aspartate family, Glu: glutamate family. The ratios in all genotypes are shown in Suppl. Figs. 1–3
Alterations in the concentrations of the individual amino acids
Within the glutamate family, the ratio of GABA (γ-amino butyric acid) was about 60–80% and the ratio of Gln was 20–40% before cold acclimation in nearly all genotypes (Fig. 4, Suppl. Figs. 4–6). After 7 and 21 d cold hardening, the ratio of GABA decreased to 20–30% with a parallel increase in the ratio of Pro to 40–60%. The great elevation in the Pro ratio was a result of the large (100–1000-fold) increase in the Pro concentration (Fig. 5). The Pro content was nearly two-fold greater in the tolerant barley and wheat genotypes compared to the sensitive ones, except for the wheat variety GK Tiszatáj after 3 weeks of cold hardening. In addition, the Gln content was also greater in the tolerant wheat and barley genotypes than in the sensitive ones, except for the wheat variety Che and the barley variety Kol after 21 d at 2 °C (Fig. 6). Such a relationship between Pro, Gln levels and freezing tolerance was not found in rye. The Gln, Glu, and GABA concentrations exhibited a greater increase only during the first 3 d of cold hardening in all 3 genotypes (Suppl. Fig. 8). In contrast, Pro and Arg contents showed greater changes after 7 and 21 d at 2 °C (Fig. 5, Suppl. Fig. 9A-9C).
Time-course of the cold-induced changes in the free proline content. Sampling was done after 0 (control), 1, 3, 7, and 21 days of cold hardening. The data were subjected to a general linear model for multivariate analysis of variance using SPSS package version 16.0. The Pro level was significantly affected by the genotype, cold and their interactions except for the genotype in barley (Suppl. Table 1). The order of the genotypes can be seen in Fig. 1. T: freezing-tolerant, S: freezing-sensitive
Time-course of the cold-induced changes in the free glutamine content. Sampling was done after 0 (control), 1, 3, 7, and 21 days of cold hardening. The data were subjected to a general linear model for multivariate analysis of variance using SPSS package version 16.0. The Gln level was significantly affected by the genotype, cold and their interactions except for the interaction between genotype and cold in wheat (Suppl. Table 1). The order of the genotypes can be seen in Fig. 1. T: freezing-tolerant, S: freezing-sensitive
Within the aspartate family, the concentration of Asn and Asp had a greater increase in most of the studied Triticeae species in the second half of the hardening period, while the amount of Thr and Ile slightly increased and that of Lys decreased during the first 3 d of cold treatment in certain genotypes (Suppl. Figs. 10–12). Regarding the pyruvate family, an increase in the Leu content was observed during the first 3 d of cold hardening in all 3 species and the Val concentration became greater later (Suppl. Fig. 13–14). Among the amino acids of the serine family, the amount of Gly increased during the first 3 d of cold treatment in all genotypes, and the Ser and Cys content exhibited a small transient decrease during the first 3 d at 2 °C in several genotypes. Later only smaller or no changes occurred in the levels of amino acids of serine family in all 3 genotypes (Suppl. Figs. 14–15). Considering the aromatic amino acids, the amount of Tyr showed greater changes during the first week of the cold hardening (Suppl. Fig. 16). The level of Phe showed a smaller decrease after the first week of hardening in some genotypes (Suppl. Fig. 9D–F).
Changes at the gene expression level
Since the greatest cold-induced changes occurred in the ratio of the amino acids belonging to the glutamate and aspartate families, the effect of cold on the expression of several genes related to their metabolism was monitored by cDNA-macroarrays for a better understanding of the biochemical background of the observed alterations. Similar to the Pro levels, the expression of the P5CS gene encoding a key enzyme in Pro biosynthesis was greatly induced (significantly at p < 5% level) by low temperature in all genotypes compared to the corresponding controls (Fig. 7). However, this change has no relationship with the level of freezing tolerance in the 3 species. The expression of P5CDH participating in the Pro degradation was not affected by cold, but it was, in general, greater in rye and wheat compared to barley. The transcription of the other genes related to Pro metabolism was similar in all genotypes at all sampling points. Interestingly, the expression of the gene encoding a Gln synthetase was lower in barley than in wheat and rye. The transcription of the genes encoding enzymes of Glu, Asp, and Asn metabolism was not affected by the cold. Thus, the observed changes in the amounts of Glu, Asp, and Asn probably derives from the cold-induced alterations in the activity of the enzymes involved in their metabolism.
Time course of the cold-induced changes in the expression of the genes related to the Pro, Gln, Glu, Asn, and Asp metabolism. Sampling was done after 0 (control), 1, 3, 7, and 21 days of cold hardening. The order of the genotypes can be seen in Fig. 1. T: freezing-tolerant, S: freezing-sensitive, r: rye, w: wheat, and b: barley. A significant difference (p < 5%) between control and cold-treated plants was found for the gene encoding ∆-1-pyrroline-5-carboxylate synthase in all genotypes (multivariate analysis of variance, Tukey’s HSD test, two biological replicates with three parallels)
Discussion
The freezing test results confirmed the suitability of our system to examine the potential correlation between freezing tolerance levels and cold-induced alterations in amino acid composition and levels in Triticeae species. We were able to effectively demonstrate differences in freezing tolerance both among the three species and among the six genotypes studied within each species. Consistent with Campoli et al. (2009), our findings indicate that rye exhibited the highest level of tolerance, while barley showed the highest sensitivity to freezing.
Apart from the alterations in the levels of amino acids as observed in this investigation, some of the ultrastructure changes also confer stress tolerance. For instance, there are different changes in the protoplast of cold-acclimatised and non-acclimatised winter rye seedlings. The changes in non-acclimatised seedlings’ protoplast, freeze-induced osmotic contractions which result in the irreversible formation of endocytotic vesiculation of the plasma membrane. However, in the case of cold acclimatised seedlings’ protoplast, the freeze-induced osmotic contraction results in the reversible formation of exocytotic extrusions of the plasma membrane. Moreover, the loss of responsiveness of non-acclimatised winter rye protoplast occurs at -6 °C. Whereas, in the case of cold-acclimated seedlings protoplast, the injury and loss of responsiveness occur at -20 °C (Webb et al. 1994). Similarly, the alterations in the ultrastructural organization of cells were observed as a response to adapt to low temperatures in winter wheat. After low-temperature hardening, the cells’ size increased with the cytoplasm and accumulation of vesicles, however, the size of the vacuole decreased. In addition, changes in the shapes of chloroplast and mitochondria were also observed as they became more rounded and dumbbell-shaped, respectively. Peroxisomes and mitochondria also increased in number and clustered near chloroplast along with the disappearance of starch inclusions (Venzhik and Moshkov 2023). The roots of the winter wheat plants when subjected to a low temperature of 2 °C caused structural changes in the mesophyll cells of the leaves. The chloroplast increased in size along with the total length of thylakoid membranes (Venzhik et al. 2012). Likewise, similar observations of anatomical alterations and functional reorganisation of the photosynthetic apparatus as an adaptation to cold were also elucidated in winter wheat (Venzhik et al. 2013). Thus, the available literature hints the structural reorganisation as an adaptation exhibited by the cold-tolerant species.
Cold-induced changes in the total amount of free amino acids and their composition
In terms of cold-induced changes in total free amino acids and their composition, although the concentration of total free amino acids was similar among the Triticeae species under control conditions and during the initial three days of cold treatment, it was twice as high in barley and wheat compared to rye after three weeks of cold acclimation. This suggests that the increase in total free amino acid content during long-term cold acclimation may serve as an indicator of greater freezing sensitivity in barley and wheat relative to rye. However, regarding individual genotypes, the cold-induced changes in total free amino acid levels did not exhibit a consistent relationship with their freezing tolerance, which is in line with the findings of Kovács et al. (2011) when comparing different wheat genotypes.
During cold treatment, the accumulation of free amino acids followed a similar pattern in all three species and across all genotypes, resulting in an elevated proportion of the glutamate family (from 40–60% to 60–80%) and a decreased proportion of the aspartate family (from 20–40% to 10–20%) and serine family (from 10 to 5%). These alterations in the ratios of amino acid families can be attributed to the significant cold-induced increase (100–1000-fold) in proline (a member of the glutamate family) concentration. This observation suggests that rather than a general shift in the overall level and composition of free amino acids at low temperatures, there is a targeted adjustment in the level of specific amino acids. The observed changes in the amount of the individual amino acids were affected in general by the genotype, cold treatment and the interactions of these two parameters in all three species (Suppl. Table 1).
Role of amino acids in the short-term response to cold
During the initial three days of cold treatment in all genotypes of the three Triticeae species, a significant accumulation of Glu, Gln, and GABA, which are part of the glutamate family, was observed. This finding aligns with previous studies that demonstrated coordinated metabolic reprogramming of these amino acids at low temperatures in wheat and Arabidopsis (Kaplan et al. 2007; Kovács et al. 2011)). The involvement of Glu in the short-term response to cold stress has also been observed in Lolium perenne (Bocian et al. 2015). Glu plays a central role in this amino acid family as a precursor for Gln, GABA, Pro, and Arg. Therefore, the increased concentration of Glu is essential for the enhanced synthesis of these amino acids, which was observed concurrently for Gln and GABA, and after one week for Pro and Arg in our experiments. The increased Glu content facilitates the accumulation of polyamines, which play a crucial role in the response to low temperature stress (Kovács et al. 2011). In addition to Glu, Gln, and GABA, a rapid induction of Leu, Gly, and Phe content was observed during the initial days of cold treatment. The synthesis of these three amino acids is linked to various steps of glycolysis and the pentose-phosphate cycle, suggesting that changes in their concentrations may arise from well-known cold-dependent alterations in carbohydrate metabolism (Asghar et al. 2021). The simultaneous cold-induced rapid increase in Gly and Glu contents enables greater synthesis of glutathione (γ-Glu-Cys-Gly, GSH), which has been previously described in cold-acclimated wheat (Kocsy et al. 2000). The increase in glutathione plays a crucial role in protecting against low temperature-induced damage, as it acts as an antioxidant and participates in the removal of excess reactive oxygen species generated under unfavourable environmental conditions.
The quick induction of Glu, Gln, GABA, Leu, Gly, and Phe accumulation suggests their involvement in regulatory processes. Indeed, Glu has been shown to influence stress response, root development, and growth in Arabidopsis (Forde 2014; Weiland et al. 2016). The addition of Gln to the nutrient solution increased the expression of genes encoding transcription factors involved in the regulation of nitrogen metabolism and stress response (Kan et al. 2011). The regulatory role of GABA in low temperature tolerance has been indicated in barley, wheat, and tomato with different freezing tolerance levels (Mazzucotelli et al. 2006; Liu et al. 2020). The importance of Leu in the stress response has been demonstrated in Arabidopsis through investigations of gene expression and Leu content under changing environmental conditions, Leu treatments, or using Leu overproducing plants (Hannah et al. 2010). Based on these findings, it can be proposed that these amino acids also play regulatory roles during cold acclimation in cereals.
Involvement of free amino acids in the long-term response to cold
After 7 or 21 days of cold treatment, several amino acids, namely Pro, Arg, Asn, Asp, His, and Val, exhibited increased accumulation. These amino acids may play a role in long-term adaptation to cold conditions. They could be involved in nitrogen storage during unfavorable conditions or in reducing damage caused by low temperatures. The coordination of these long-term changes in amino acid levels is evidenced by the increased concentration of two members from the glutamate family (Pro and Arg) and two members from the aspartate family (Asn and Asp) in response to cold. The cold-induced accumulation of Pro has been observed in previous wheat experiments as well (Wang et al. 2022). Similarly, increased levels of Asn have been shown to participate in long-term adaptation to low temperatures in Lolium perenne, and its concentration, like in the studied Triticeae genotypes, did not correlate with freezing tolerance levels (Bocian et al. 2015). In contrast to barley, wheat, and rye, Asp accumulated during the initial days of cold acclimation in this particular species. Notably, Asp, Pro, Arg, and Val concentrations also increased after only 4 days of cold treatment in Arabidopsis, indicating a different time-course of low-temperature-induced reprogramming of amino acid metabolism across various species (Kaplan et al. 2007).
Pro may contribute to long-term adaptation to cold as an osmolyte, reducing damage associated with osmotic stress induced by low temperatures (Alvarez et al. 2022). Arg, as a precursor to protective polyamines (positively charged macromolecules that safeguard negatively charged proteins), plays a crucial role in reducing stress-induced injuries (Alcázar et al. 2011). Asn has a central role in nitrogen storage and transport (Lea et al. 2007), while Asp indirectly participates in these processes through its conversion to Asn. In summary, four out of the six amino acids that accumulate during long-term cold treatment serve important functions in low-temperature adaptation. His and Val may also have similar protective roles, although further research is needed to explore their specific contributions in this context.
Relationship between free amino acid levels and freezing tolerance
In accordance with the well-known positive role of Pro as an osmolyte, molecular chaperone, antioxidant, and growth regulator in stress tolerance (Alvarez et al. 2022). This investigation confirmed its involvement in the cold acclimation process. Its concentration was found to be higher in freezing-tolerant genotypes of wheat and barley compared to the sensitive ones. In wheat, there was a positive correlation between the level of freezing tolerance and proline content (r = 0.63 at − 15 °C), and a similar positive correlation was observed in barley (r = 0.86 at − 13 °C). The protective role of Pro was also demonstrated in spinach, in which treatment with 10 mM Pro increased freezing tolerance (Shin et al. 2018). Additionally, in barley, a positive correlation was also found between freezing tolerance and Gln content (r = 0.67 at − 15 °C), which is involved in Pro synthesis through Glu. However, in rye, similar to turf grass (Dionne et al. 2001), Pro levels did not correlate with freezing tolerance, and there were no significant differences in Pro content between the sensitive and tolerant genotypes. Thus, the accumulation of Pro is not universally associated with the level of freezing tolerance across all cereal species and external application of Pro may or may not increase the freezing tolerance. Contrasting results were observed in Arabidopsis, where the levels of Pro, Gln, Asn, Gly, Asp, Ser, and Ala changed differently in cold-treated sensitive and tolerant ecotypes (Cook et al. 2004).
In our experimental system, the gene encoding P5CS, an enzyme involved in Pro biosynthesis, was induced by cold treatment in all the cereal species studied. A post-transcriptional regulation of its expression by miR164c during the response to cold can be proposed based on the results obtained in drought-stressed Arabidopsis (Gupta et al. 2020). The activation of P5CS gene is likely mediated by C-repeat-binding transcription factors (CBF), which are key regulators of freezing tolerance, as demonstrated in transgenic Arabidopsis (Chen et al. 2023). The constitutive overexpression of CBF3 in Arabidopsis led to increased Pro levels due to elevated P5CS expression, even without cold acclimation. The specific role of the P5CS gene in the substantial accumulation of Pro during cold acclimation is evident, as the expression of other investigated genes related to Pro metabolism was not influenced by cold. This phenomenon is likely a general feature of cold acclimation in cereals since the level of P5CS transcripts in both tolerant and sensitive genotypes was similar. Therefore, the differences in Pro content associated with freezing tolerance in wheat and barley might result from regulatory mechanisms at the translational or enzyme activation level, as described in Arabidopsis (Kaplan et al. 2007). Furthermore, genes involved in Glu, Gln, Asp, and Asn metabolism were unaffected, and the levels of these amino acids showed a much smaller increase compared to Pro.
Conclusions
A comparison of free amino acid and related transcript levels in three freezing-tolerant and three sensitive rye, wheat, and barley genotypes indicated that alterations in Gln and Pro concentration in barley and wheat and changes in Gln content in barley are related to the level of freezing tolerance. Interestingly, in the case of rye, which is the most tolerant cereal species, there was no relationship between freezing tolerance and amino acid levels. It was a general phenomenon that the huge increase in Pro level had a major role in the elevation of the ratio of glutamate family and the total free amino acid content in each species. The time-course of changes during the 3-week cold acclimation indicates that Gln, Glu, Gaba, Leu, Gly, Phe are involved in the short-term and Gln, Glu, Gaba, Leu, Gly, Phe participates in the long-term response to cold. Among the amino acid metabolism-related genes, only the expression of P5CS was induced by cold.
Data availability
All data generated or analysed during this study are included in this article (and its supplementary information files). The experimental design, raw data, and normalized data of cDNA macroarray analysis were deposited in the Gene Expression Omnibus database (Accession No. GSE47882).
Abbreviations
- Pro:
-
Proline
- Glu:
-
Glutamic acid
- Gly:
-
Glycine
- Thr:
-
Threonine
- Ala:
-
Alanine
- Arg:
-
Arginine
- Asp:
-
Aspartic acid
- Asn:
-
Asparagine
- Gln:
-
Glutamine
- GABA:
-
γ-Amino butyric acid
- His:
-
Histidine
- Ile:
-
Isoleucine
- Leu:
-
Leucine
- Lys:
-
Lysine
- Phe:
-
Phenylalanine
- Trp:
-
Tryptophan
- Val:
-
Valine
- Mir:
-
Miranoskaja
- Che:
-
Cheyenne
- Ulj:
-
Uljanovka
- GKT:
-
GK Tiszatáj
- CD:
-
Capelle Desprez
- Mv8:
-
Martonvásári 8
References
Ahad A, Gul A, Batool TS et al (2023) Molecular and genetic perspectives of cold tolerance in wheat. Mol Biol Rep 50:6997–7015. https://doi.org/10.1007/s11033-023-08584-1
Alcázar R, Cuevas JC, Planas J et al (2011) Integration of polyamines in the cold acclimation response. Plant Sci 180:31–38. https://doi.org/10.1016/j.plantsci.2010.07.022
Alvarez ME, Savouré A, Szabados L (2022) Proline metabolism as regulatory hub. Trends Plant Sci 27:39–55. https://doi.org/10.1016/j.tplants.2021.07.009
Asghar MA, Mednyánszky Z, Simon-Sarkadi L, Kocsy G (2021) Different induction of biogenic amine accumulation during cold acclimation in Triticeae genotypes with varying freezing tolerance. Braz J Bot 44:11–15. https://doi.org/10.1007/s40415-020-00690-9
Bocian A, Zwierzykowski Z, Rapacz M et al (2015) Metabolite profiling during cold acclimation of Lolium perenne genotypes distinct in the level of frost tolerance. J Appl Genet 56:439–449. https://doi.org/10.1007/s13353-015-0293-6
Bolstad BM, Irizarry R, Åstrand M, Speed TP (2003) A comparison of normalization methods for high density oligonucleotide array data based on variance and bias. Bioinformatics 19:185–193. https://doi.org/10.1093/bioinformatics/19.2.185
Cai H, Zhou Y, Xiao J et al (2009) Overexpressed glutamine synthetase gene modifies nitrogen metabolism and abiotic stress responses in rice. Plant Cell Rep 28:527–537. https://doi.org/10.1007/s00299-008-0665-z
Campoli C, Matus-Cádiz MA, Pozniak CJ et al (2009) Comparative expression of Cbf genes in the Triticeae under diVerent acclimation induction temperatures. Mol Genet Genomics 282:141–152. https://doi.org/10.1007/s00438-009-0451-9
Chen J-B, Wang S-M, Jing R-L, Mao X-G (2009) Cloning the PvP5CS gene from common bean (Phaseolus vulgaris) and its expression patterns under abiotic stresses. J Plant Physiol 166:12–19. https://doi.org/10.1016/j.jplph.2008.02.010
Chen N, Pan L, Yang Z et al (2023) A MYB-related transcription factor from peanut, AhMYB30, improves freezing and salt stress tolerance in transgenic Arabidopsis through both DREB/CBF and ABA-signaling pathways. Front Plant Sci. https://doi.org/10.3389/fpls.2023.1136626
Cook D, Fowler S, Fiehn O, Thomashow MF (2004) From the cover: a prominent role for the CBF cold response pathway in configuring the low-temperature metabolome of Arabidopsis. Proc Natl Acad Sci 101:15243–15248. https://doi.org/10.1073/pnas.0406069101
Dionne J, Castonguay Y, Nadeau P, Desjardins Y (2001) Amino acid and protein changes during cold acclimation of green-type annual Bluegrass (L.) ecotypes. Crop Sci 41:1862. https://doi.org/10.2135/cropsci2001.1862
Forde BG (2014) Glutamate signalling in roots. J Exp Bot 65:779–787
Gupta A, Patil M, Qamar A, Senthil-Kumar M (2020) ath-miR164c influences plant responses to the combined stress of drought and bacterial infection by regulating proline metabolism. Environ Exp Bot 172:103998. https://doi.org/10.1016/j.envexpbot.2020.103998
Hannah MA, Caldana C, Steinhauser D, et al (2010) Combined transcript and metabolite profiling of arabidopsis grown under widely variant growth conditions facilitates the identification of novel metabolite-mediated regulation of gene expression. Plant Physiol 152
Hoagland D, Arnon D (1950) The water-culture method for growing plants without soil. Circ Calif Agric Exp Stn. https://doi.org/10.1097/00010694-193910000-00022
Kan C-C, Chung T-Y, Juo Y-A, Hsieh M-H (2011) Glutamine rapidly induces the expression of key transcription factor genes involved in nitrogen and stress responses in rice roots. BMC Genomics. https://doi.org/10.1186/s12864-015-1892-7
Kaplan F, Kopka J, Sung DY et al (2007) Transcript and metabolite profiling during cold acclimation of Arabidopsis reveals an intricate relationship of cold-regulated gene expression with modifications in metabolite content. Plant J 50:967–981. https://doi.org/10.1111/j.1365-313X.2007.03100.x
Kocsy G, Athmer B, Perovic D et al (2010) Regulation of gene expression by chromosome 5A during cold hardening in wheat. Mol Genet Genomics 283:351–363
Kocsy G, Szalai G, Vágújfalvi A et al (2000) Genetic study of glutathione accumulation during cold hardening in wheat. Planta 210:295–301
Kovács Z, Simon-Sarkadi L, Sovány C et al (2011) Differential effects of cold acclimation and abscisic acid on free amino acid composition in wheat. Plant Sci 180:61–68
Lamichhane JR (2021) Rising risks of late-spring frosts in a changing climate. Nat Clim Chang 11:554–555. https://doi.org/10.1038/s41558-021-01090-x
Lea PJ, Sodek L, Parry MAJ et al (2007) Asparagine in plants. Ann Appl Biol 150:1–26. https://doi.org/10.1111/j.1744-7348.2006.00104.x
Liu T, Jiao X, Yang S et al (2020) Crosstalk between GABA and ALA to improve antioxidation and cell expansion of tomato seedling under cold stress. Environ Exp Bot 180:104228. https://doi.org/10.1016/j.envexpbot.2020.104228
Lohse M, Nagel A, Herter T et al (2014) Mercator: a fast and simple web server for genome scale functional annotation of plant sequence data. Plant Cell Environ 37:1250–1258. https://doi.org/10.1111/pce.12231
Machacckova I, Hanisova A, Krekule J (1989) Levels of ethylene, ACC, MACC, ABA and proline as indicators of cold hardening and frost resistance in winter wheat. Physiol Plant 76:603–607. https://doi.org/10.1111/j.1399-3054.1989.tb05486.x
Mazzucotelli E, Tartari A, Cattivelli L, Forlani G (2006) Metabolism of ?-aminobutyric acid during cold acclimation and freezing and its relationship to frost tolerance in barley and wheat. J Exp Bot 57:3755–3766. https://doi.org/10.1093/jxb/erl141
Monroy AF, Dryanova A, Malette B et al (2007) Regulatory gene candidates and gene expression analysis of cold acclimation in winter and spring wheat. Plant Mol Biol 64:409–423. https://doi.org/10.1007/s11103-007-9161-z
Ritchie ME, Phipson B, Wu D et al (2015) limma powers differential expression analyses for RNA-sequencing and microarray studies. Nucleic Acids Res 43:e47–e47. https://doi.org/10.1093/nar/gkv007
Shin H, Oh S, Kim D et al (2018) Induced freezing tolerance and free amino acids perturbation of spinach by exogenous proline. J Plant Biotechnol 45:357–363. https://doi.org/10.5010/JPB.2018.45.4.357
Svensson JT, Crosatti C, Campoli C, et al (2006) Transcriptome analysis of cold acclimation in barley albina and xantha mutants. Plant Physiol 141
Tischner T, Koszegi B, Veisz O (1997) Climatic programmes used in the Martonvásár phytotron most frequently in recent years. Acta Agron Hungarica 45:85–104
Usadel B, Blasing OE, Gibon Y et al (2008) Multilevel genomic analysis of the response of transcripts, enzyme activities and metabolites in Arabidopsis rosettes to a progressive decrease of temperature in the non-freezing range. Plant Cell Environ 31:518–547. https://doi.org/10.1111/j.1365-3040.2007.01763.x
Vágújfalvi A, Kerepesi I, Galiba G et al (1999) Frost hardiness depending on carbohydrate changes during cold acclimation in wheat. Plant Sci 144:85–92. https://doi.org/10.1016/S0168-9452(99)00058-8
Venzhik YV, Moshkov IE (2023) The role of ultrastructural organization of cells in adaptation of winter wheat to low temperature. Russ J Plant Physiol 70:100. https://doi.org/10.1134/S1021443723600873
Venzhik YV, Titov AF, Koteyeva NK et al (2012) The effect of low temperature on wheat roots causes rapid changes in chloroplast ultrastructure in wheat leaves. Dokl Biol Sci 445:230–234. https://doi.org/10.1134/S0012496612040047
Venzhik YV, Titov AF, Talanova VV et al (2013) Structural and functional reorganization of the photosynthetic apparatus in adaptation to cold of wheat plants. Cell Tissue Biol 7:168–176. https://doi.org/10.1134/S1990519X13020132
Waititu JK, Cai Q, Sun Y et al (2021) Transcriptome profiling of maize (Zea mays L.) leaves reveals key cold-responsive genes, transcription factors, and metabolic pathways regulating cold stress tolerance at the seedling stage. Genes (Basel) 12:1638. https://doi.org/10.3390/genes12101638
Wang W, Wang X, Lv Z et al (2022) Effects of cold and salicylic acid priming on free proline and sucrose accumulation in winter wheat under freezing stress. J Plant Growth Regul 41:2171–2184. https://doi.org/10.1007/s00344-021-10412-4
Webb MS, Uemura M, Steponkus PL (1994) A Comparison of freezing injury in oat and rye: two cereals at the extremes of freezing tolerance. Plant Physiol 104:467–478. https://doi.org/10.1104/pp.104.2.467
Weiland M, Manusco S, Baluska FB (2016) Signalling via glutamate and GLRs in Arabidopsis thaliana. Funct Plant Biol 43:1–25
Xu J, Chen Z, Wang F et al (2020) Combined transcriptomic and metabolomic analyses uncover rearranged gene expression and metabolite metabolism in tobacco during cold acclimation. Sci Rep 10:5242. https://doi.org/10.1038/s41598-020-62111-x
Zhao Y, Zhou M, Xu K et al (2019) Integrated transcriptomics and metabolomics analyses provide insights into cold stress response in wheat. Crop J 7:857–866. https://doi.org/10.1016/j.cj.2019.09.002
Zhu J, Dong C-H, Zhu J-K (2007) Interplay between cold-responsive gene regulation, metabolism and RNA processing during plant cold acclimation. Curr Opin Plant Biol 10:290–295. https://doi.org/10.1016/j.pbi.2007.04.010
Zierold U, Scholz U, Schweizer P (2005) Transcriptome analysis of mlo -mediated resistance in the epidermis of barley. Mol Plant Pathol 6:139–151. https://doi.org/10.1111/j.1364-3703.2005.00271.x
Acknowledgements
The authors wish to thank A. Horváth and M. Fehér for their help in plant cultivation and treatment. This work was funded by the National Research, Development and Innovation Office (Grant TKP2021-NKTA-06).
Funding
Open access funding provided by HUN-REN Centre for Agricultural Research. This work was funded by the National Research, Development and Innovation Office (grant TKP2021-NKTA-06) and Kalpita Singh is supported by the Stipendium Hungaricum program of the Tempus Public Foundation (SHE-079837-004/2022).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethics approval
N/A.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Singh, K., Gulyás, Z., Athmer, B. et al. Comparative study of free amino acids at metabolite and gene expression levels in Triticeae during cold acclimation. J. Plant Biochem. Biotechnol. (2024). https://doi.org/10.1007/s13562-024-00912-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s13562-024-00912-1