Abstract
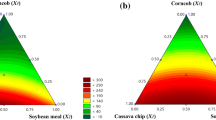
Poly (l-lactide) (PLLA) is an aliphatic polyester that can be obtained from renewable resources and degraded by various microorganisms. In previous reports, Actinomadura keratinilytica strain T16-1 demonstrated high ability to degrade PLLA under various conditions. PLLA-degrading enzyme production under solid state fermentation has been sparsely studied. PLLA-degrading enzyme production by A. keratinilytica strain T16-1 was investigated using agricultural wastes as substrate under solid state fermentation (SSF). Three agricultural wastes as soybean meal, cassava chips and duckweed were tested as substrates for PLLA-degrading enzyme production by statistical methods using mixture design. Results revealed that using duckweed as the substrate gave the highest enzyme production (138.66 ± 13.57 U/g dry substrate). Maximum enzyme activity of 391.24 ± 15.57 U/g dry substrate was obtained under 10 g duckweed, 10% inoculum size, 7 days of cultivation time, pH 7.0, 2.8% PLLA powder, and 60% moisture content at 45 °C. It can be concluded that duckweed is an inexpensive substrate, which reduces the costs of PLLA-degrading enzyme production, as an alternative to effective water weed management.






Similar content being viewed by others
References
Aciu C, Varvara DA, Manea DL, Orban YA, Babota F (2018) Recycling of plastic waste materials in the composition of ecological mortars. Procedia Manuf 22:274–279. https://doi.org/10.1016/j.promfg.2018.03.042
Aljammas HA, Fathi HA, Alkhalaf W (2018) Study the influence of culture conditions on rennin production by Rhizomucor miehei using solid-state fermentations. J Genet Eng Biotechnol 16:213–216. https://doi.org/10.1016/j.jgeb.2017.10.004
Bech L, Herbst FA, Grell MN, Hai Z, Lange L (2015) On-site enzyme production by Trichoderma asperellum for the degradation of duckweed. Fungal Genom Biol 5(2):1–10. https://doi.org/10.4172/2165-8056.1000126
Clais S, Boulet G, Van Kerckhoven M, Lanckacker E, Delputte P, Maes L, Cos P (2014) Comparison of viable plate count, turbidity measurement and real-time PCR for quantification of Porphyromonas gingivalis. Lett Appl Microbiol 60:79–84. https://doi.org/10.1111/lam.12341
de Castro RJS, Sato HH (2013) Synergistic effects of agroindustrial wastes on simultaneous production of protease and α-amylase under solid state fermentation using a simplex centroid mixture design. Ind Crops Prod 49:813–821. https://doi.org/10.1016/j.indcrop.2013.07.002
El-Naggar NE, Sherief AA, Hamza SS (2011) Bioconversion process of rice straw by thermotolerant cellulolytic Streptomyces viridiochromogenes under solid-state fermentation conditions for bioethanol production. Afr J Biotechnol 10(56):11998–12011. https://doi.org/10.5897/AJB11.1256
Fukuzaki H, Yoshida M, Asano M, Kumakura M (1989) Synthesis of copoly (D, L-lactic acid) with relative low molecular weight and in vitro degradation. Eur Polym J 25:1019–1026. https://doi.org/10.1016/0014-3057(89)90131-6
Hanphakphoom S, Maneewong N, Sukkhum S, Tokuyama S, Kitpreechavanich V (2014) Characterization of poly(L-lactide)- degrading enzyme produced by thermophilic filamentous bacteria Laceyella sacchari LP175. J Gen Appl Microbiol 60:13–22. https://doi.org/10.2323/jgam.60.13
Ishizawa H, Kuroda M, Morikawa M, Ike M (2017) Evaluation of environmental bacterial communities as a factor affecting the growth of duckweed Lemna minor. Biotechnol Biofuels 10(62):2–10. https://doi.org/10.1186/s13068-017-0746-8
Jarerat A, Tokiwa Y, Tanaka H (2006) Production of poly (L-lactide)-degrading enzyme by Amycolatopsis orientalis for biological recycling of poly (L-lactide). Appl Microbiol Biotechnol 72:726–731. https://doi.org/10.1007/s00253-006-0343-4
Kamath PV, Dwarakanath BS, Chaudhary A, Janakiraman S (2015) Optimization of culture conditions for maximal lovastatin production by Aspergillus terreus (KM017963) under solid state fermentation. Hayati J Biosci 22:174–180. https://doi.org/10.1016/j.hjb.2015.11.001
Kolstad JJ, Vink ETH, Wilde BD, Debeer L (2012) Assessment of anaerobic degradation of IngeoTM polylactides under accelerated landfill conditions. Polym Degrad Stab 97:1131–1141. https://doi.org/10.1016/j.polymdegradstab.2012.04.003
Krishna C (2005) Solid-state fermentation systems—an overview. Crit Rev Biotechnol 25:1–30. https://doi.org/10.1080/07388550590925383
Lazim H, Mankai H, Slama N, Barkallah I, Limam F (2009) Production and optimization of thermophilic alkaline protease in solid-state fermentation by Streptomyces sp. CN902. J Ind Microbiol Biotechnol 36:531–537. https://doi.org/10.1007/s10295-008-0523-6
Li GY, Cai YJ, Liao XR, Yin J (2011) A novel nonionic surfactant- and solvent-stable alkaline serine protease from Serratia sp. SYBC H with duckweed as nitrogen source: production, purification, characteristics and application. J Ind Microbiol Biotechnol 38:845–853. https://doi.org/10.1007/s10295-010-0855-x
Li C, Li D, Feng J, Fan X, Chen S, Zhang D, He R (2019) Duckweed (Lemna minor) is a novel natural inducer of cellulase production in Trichoderma reesei. J Biosci Bioeng 127(4):486–491. https://doi.org/10.1016/j.jbiosc.2018.09.017
Lim HA, Raku T, Tokiwa Y (2005) Hydrolysis of polyesters by serine proteases. Biotechnol Lett 27:459–464. https://doi.org/10.1007/s10529-005-2217-8
Lomthong T, Hanphakphoom S, Yoksan R, Kitpreechavanich V (2015) Co-production of poly(L-lactide)-degrading enzyme and raw starch-degrading enzyme by Laceyella sacchari LP175 using agricultural products as substrate, and their efficiency on biodegradation of poly(L-lactide)/thermoplastic starch blend film. Int Biodeter Biodegr 104:401–410. https://doi.org/10.1016/j.ibiod.2015.07.011
Lomthong T, Hanphakphoom S, Kongsaeree P, Srisuk N, Guicherd M, Cioci G, Kitpreechavanich V (2017) Enhancement of poly (L-lactide)-degrading enzyme production by Laceyella sacchari LP175 using agricultural crops as substrates and its degradation of poly (L-lactide) polymer. Int Biodeter Biodeger 143:64–73. https://doi.org/10.1016/j.polymdegradstab.2017.06.017
Lomthong T, Yoksan R, Lumyong S, Kitpreechavanich V (2020) Poly(L-lactide)-degrading enzyme production by Laceyella sacchari LP175 under solid state fermentation using low cost agricultural crops and its hydrolysis of poly(L-lactide) film. Waste Biomass Valor 11:1961–1970. https://doi.org/10.1007/s12649-018-0519-z
Lomthong T, Areesirisuka A, Suphana S, Panyachanakul T, Krajangsang S, Kitpreechavanichd V (2021) Solid state fermentation for poly (L-lactide)-degrading enzyme production by Laceyella sacchari LP175 in aerated tray reactor and its hydrolysis of poly (lactide) polymer. Agri Nat Resour 55:147–152. https://doi.org/10.34044/j.anres.2021.55.1.19
Lopesa MS, Jardini AL, Filho MR (2012) Poly (lactic acid) production for tissue engineering applications. Procedia Eng 42:1402–1413. https://doi.org/10.1016/j.proeng.2012.07.534
Masaki K, Ramuda N, Ikeda H, Iefuji H (2005) Cutinase-like enzyme from the yeast Cryptococcus sp. strain S-2 hydrolyzes polylactide acid and other biodegradable plastics. Appl Environ Microbiol 71:7548–7550. https://doi.org/10.1128/AEM.71.11.7548-7550.2005
Mienda SB, Idi A, Umar A (2011) Microbiological features of solid state fermentation and its applications-an overview. Res Biotechnol 2(6):21–26. https://www.researchgate.net/publication/236177189
Nair LS, Laurencin CT (2007) Biodegradable polymers as biomaterials. Prog Polym Sci 32(8–9):762–798. https://doi.org/10.1016/j.progpolymsci.2007.05.017
Ooi CK, Rasit N, Absullah WRW (2021) Optimization of protease from Aspergillus niger under solid-state fermentation utilizing shrimp shell substrate. Biointerface Res Appl Chem 11(6):14809–14824. https://doi.org/10.33263/BRIAC116.1480914824
Pandey A, Soccol CR, Mitchell D (2000) New developments in solid-state fermentation: Ibioprocesses and products. Proc Biochem 35:1153–1169. https://doi.org/10.1016/S0032-9592(00)00152-7
Panyachanakul T, Kitpreechavanich V, Tokuyama S, Krajangsang S (2017) Poly (D, L-lactide)-degrading enzyme production by immobilized Actinomadura keratinilytica strain T16–1 in a 5-L fermenter under various fermentation processes. Electron J Biotechnol 30:71–76. https://doi.org/10.1016/j.ejbt.2017.09.001
Panyachanakul T, Sorachart B, Lumyong S, Lorliam W, Kitpreechavanich V, Krajangsang S (2019) Development of biodegradation process for Poly(DL-lactic acid) degradation by crude enzyme produced by Actinomadura keratinilytica strain T16–1. Electron J Biotechnol 40:52–57. https://doi.org/10.1016/j.ejbt.2019.04.005
Prema P, Uma Maheswari Devi P (2015) Degradation of Polylactide plastic by PLA depolymerase isolated from thermophilic Bacillus. Int J Curr Microbiol Appl Sci 4(12):645–654. https://www.researchgate.net/publication/285925295
Sanjivkumar M, Vijayalakshmi K, Selvan ST, Sholkamy EN (2020) Biosynthesis, statistical optimization and molecular modeling of chitinase from crab shell wastes by a mangrove associated actinobacterium Streptomyces olivaceus (MSU3) using Box-Behnken design and its antifungal effects. Bioresour Technol Rep 11:100493. https://doi.org/10.1016/j.biteb.2020.100493
Sharma R, Bansal PP (2016) Use of different forms of waste plastic in concrete—a review. J Clean Prod 112:473–482. https://doi.org/10.1016/j.jclepro.2015.08.042
Soares VF, Castilho LR, Bon EPS, Freire DMG (2005) High-yield Bacillus subtilis protease production by solid-state fermentation. Appl Biochem Biotechnol 121–124:131–320
Sukkhum S, Tokuyama S, Tamura T, Kitpreechavanich V (2009) A novel poly (L-lactide) degrading actinomycetes isolated from Thai forest soils, phylogenic relationship and the enzyme characterization. J Gen Appl Microbiol 55(9):459–467. https://doi.org/10.2323/jgam.55.459
Sukkhum S, Tokuyama S, Kitpreechavanich V (2012) Poly (L-lactide) degrading enzyme production by Actinomadura keratinilytica T16–1 in 3L airlift bioreactor and its degradation ability for biological recycle. J Gen Appl Microbiol 22(1):92–99. https://doi.org/10.4014/jmb.1105.05016
Williams DF (1981) Enzymic hydrolysis of polylactic acid. Eng Med 10:5–7. https://doi.org/10.1243/EMED_JOUR_1981_010_004_02
Xu J, Zhoa H, Stomp AM, Cheng JJ (2012) The production of duckweed as a source of biofuels. Biofuels 3(5):589–601. https://doi.org/10.4155/bfs.12.31
Xu X, Song Z, Yin Y, Zhong F, Song J, Huang J, Ye W, Wang P (2021) Solid-state fermentation production of chitosanase by Streptomyces with waste mycelia of Aspergillus niger. Adv Enzyme Res 9:10–18. https://doi.org/10.4236/aer.2021.91002
Youngpreda A, Panyachanakul T, Kitpreechavanich V, Sirisansaneeyakul S, Suksamarn S, Tokuyama S, Krajangsang S (2017) Optimization of poly (DL-lactic acid) degradation and evaluation of biological re-polymerization. J Polymers Environ 25(4):1131–1139. https://doi.org/10.1007/s10924-016-0885-1
Zhao X, Moates GK, Elliston A, Wilson DR, Coleman MJ, Waldron KW (2015) Simultaneous saccharification and fermentation of steam exploded duckweed: improvement of the ethanol yield by increasing yeast titre. Bioresour Technol 194:263–269. https://doi.org/10.1016/j.biortech.2015.06.131
Acknowledgements
This research was supported by the National Research Council of Thailand: NRCT.
Funding
The National Research Council of Thailand: NRCT.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflicts of interest.
Ethical approval
This article does not contain any studies with human or animal subjects.
Rights and permissions
About this article
Cite this article
Krajangsang, S., Dechsresawut, N., Lomthong, T. et al. Production of poly (l-lactide)-degrading enzyme by Actinomadura keratinilytica strain T16-1 under solid state fermentation using agricultural wastes as substrate. 3 Biotech 11, 512 (2021). https://doi.org/10.1007/s13205-021-03060-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s13205-021-03060-8