Abstract
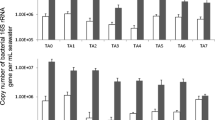
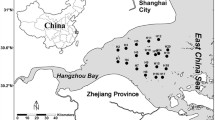
Nitrospira, a genus of nitrite-oxidizing bacteria, plays an important role in the global nitrogen cycle. However, the distribution patterns of Nitrospira communities and their responses to environmental variables in the estuary are relatively unknown. In this study, Nitrospira communities were collected at two size fractions, 0.2–2.0 μm (free-living) and >2.0 μm (particle-associated), across a physicochemical gradient in the Pearl River Estuary (PRE). The abundance and community composition of Nitrospira bacteria within these two fractions were analysed using qPCR and high-throughput sequencing targeting the nxrB gene. The abundance of particle-associated bacteria decreased dramatically from upstream to downstream, but no significant changes were observed in free-living bacteria, leading to a drastic decrease in the ratio of particle-associated to free-living bacteria (from 36.56 to 0.81). Nitrospira community composition in the PRE also exhibited remarkable spatial variation but no distinct differences in the community composition between particle-associated and free-living fractions. Seven Nitrospira species were detected in water samples from the PRE, of which Nitrospira lenta (N. lenta) and Candidatus Nitrospira defluvii (Ca. N. defluvii) were found to be dominant, and Ca. N. defluvii was observed for the first time in the estuarine environment. A marine Nitrospira, Nitrospira marina (N. marina), was more abundant in winter than in summer, possibly due to strong seawater intrusion in winter. Multivariate statistics revealed that ammonia rather than nitrite was the key factor controlling the community structure of Nitrospira, though different species responded differently to environmental variables. Community network analysis revealed tighter interactions in particle-associated Nitrospira communities than in free-living communities, suggesting that particle association favored interactions. Our analyses indicate that particles play a dominant role in shaping the structure of Nitrospira bacterial communities and imply their major function of the nitrite oxidation in the waters of the PRE.







Similar content being viewed by others
References
Acinas, S.G., J. Anton, and F. Rodriguez-Valera. 1999. Diversity of free-living and attached bacteria in offshore Western Mediterranean waters as depicted by analysis of genes encoding 16S rRNA. Applied and Environmental Microbiology 65 (2): 514–522.
Alawi, M., A. Lipski, T. Sanders, Spieck Eva-Maria-Pfeiffer, and E. 2007. Cultivation of a novel cold-adapted nitrite oxidizing betaproteobacterium from the Siberian Arctic. ISME Journal 1 (3): 256–264.
Bartosch, S., I. Wolgast, E. Spieck, and E. Bock. 1999. Identification of nitrite-oxidizing bacteria with monoclonal antibodies recognizing the nitrite oxidoreductase. Applied and Environmental Microbiology 65 (9): 4126–4133.
Bastian M., S. Heymann, and M. Jacomy. 2009. Gephi: an open source software for exploring and manipulating networks. Available at: https://www.aaai.org/ocs/index.php/ICWSM/09/paper/view/154/1009. Accesses 3 May 2020.
Beman, J.M., S.J. Leilei, and B.N. Popp. 2013. Nitrite oxidation in the upper water column and oxygen minimum zone of the eastern tropical North Pacific Ocean. ISME Journal 7 (11): 2192–2205.
Bidle, K.D., and M. Fletcher. 1995. Comparison of free-living and particle-associated bacterial communities in the chesapeake bay by stable low-molecular-weight RNA analysis. Applied and Environmental Microbiology 61 (3): 944–952.
Black, E.M., and C.L. Just. 2018. The genomic potentials of nob and comammox Nitrospira in river sediment are impacted by native freshwater mussels. Frontiers in Microbiology 9: 2061.
Castellani, A.G., and C.F. Niven Jr. 1955. Factors affecting the bacteriostatic action of sodium nitrite. Applied Microbiology 3 (3): 154–159.
Crump, B.C., E.V. Armbrust, and J.A. Baross. 1999. Phylogenetic analysis of particle-attached and free-living bacterial communities in the Columbia river, its estuary, and the adjacent coastal ocean. Applied and Environmental Microbiology 65 (7): 3192–3204.
Daims, H., E.V. Lebedeva, P. Pjevac, P. Han, C. Herbold, M. Albertsen, N. Jehmlich, M. Palatinszky, J. Vierheilig, A. Bulaev, R.H. Kirkegaard, M. von Bergen, T. Rattei, B. Bendinger, P.H. Nielsen, and M. Wagner. 2015. Complete nitrification by Nitrospira bacteria. Nature 528 (7583): 504–509.
Daims, H., S. Lucker, and M. Wagner. 2016. A new perspective on microbes formerly known as nitrite-oxidizing bacteria. Trends in Microbiology 24 (9): 699–712.
Daims, H., J.L. Nielsen, P.H. Nielsen, K.H. Schleifer, and M. Wagner. 2001. In situ characterization of Nitrospira-like nitrite-oxidizing bacteria active in wastewater treatment plants. Applied and Environmental Microbiology 67 (11): 5273–5284.
Dai, M., X. Guo, W. Zhai, L. Yuan, B. Wang, L. Wang, P. Cai, T. Tang, and W.-J. Cai. 2006. Oxygen depletion in the upper reach of the Pearl River estuary during a winter drought. Marine Chemistry 102 (1-2): 159–169.
Dworkin, M. 2012. Sergei Winogradsky: a founder of modern microbiology and the first microbial ecologist. FEMS Microbiology Review 36 (2): 364–379.
Ehrich, S., D. Behrens, E. Lebedeva, W. Ludwig, and E. Bock. 1995. A new obligately chemolithoautotrophic, nitrite-oxidizing bacterium, Nitrospira moscoviensis sp. nov. and its phylogenetic relationship. Archives of Microbiology 164 (1): 16–23.
Fuchsman, C.A., J.T. Staley, B.B. Oakley, J.B. Kirkpatrick, and J.W. Murray. 2012. Free-living and aggregate-associated Planctomycetes in the Black Sea. FEMS Microbiology Ecology 80 (2): 402–416.
Fujitani, H., Y. Aoi, and S. Tsuneda. 2013. Selective enrichment of two different types of Nitrospira-like nitrite-oxidizing bacteria from a wastewater treatment plant. Microbes and Environments 28 (2): 236–243.
Fujitani, H., N. Ushiki, S. Tsuneda, and Y. Aoi. 2014. Isolation of sublineage I Nitrospira by a novel cultivation strategy. Environmental Microbiology 16 (10): 3030–3040.
Füssel, J., P. Lam, G. Lavik, M.M. Jensen, M. Holtappels, M. Günter, and M.M.M. Kuypers. 2012. Nitrite oxidation in the Namibian oxygen minimum zone. ISME Journal 6 (6): 1200–1209.
Grossart, H.P., T. Kiorboe, K. Tang, and H. Ploug. 2003. Bacterial colonization of particles: growth and interactions. Applied and Environmental Microbiology 69 (6): 3500–3509.
Guan, F., Y. Hong, J. Wu, Y. Wang, L. Bin, B. Tang, and G. Xie. 2017. A fast sodium hypobromite oxidation method for the sequential determination of ammonia nitrogen in small volume. Ecological Science 36: 42–48.
Hentschel, U., J. Hopke, M. Horn, A.B. Friedrich, M. Wagner, J. Hacker, and B.S. Moore. 2002. Molecular evidence for a uniform microbial community in sponges from different oceans. Applied and Environmental Microbiology 68 (9): 4431–4440.
Holmes, A.J., N.A. Tujula, M. Holley, A. Contos, J.M. James, P. Rogers, and M.R. Gillings. 2001. Phylogenetic structure of unusual aquatic microbial formations in Nullarbor caves, Australia. Environmental Microbiology 3 (4): 256–264.
Hong, Y., L. Jiao, and J. Wu. 2020. New primers, taxonomic database and cut-off value for processing nxrB gene high-throughput sequencing data by MOTHUR. Journal of Microbiological Methods 173: 105939. https://doi.org/10.1016/j.mimet.2020.105939.
Hou, L., X. Xie, X. Wan, S.-J. Kao, N. Jiao, and Y. Zhang. 2018. Niche differentiation of ammonia and nitrite oxidizers along a salinity gradient from the Pearl River estuary to the South China Sea. Biogeosciences Discussions 15 (16): 5169–5187.
Hovanec, T.A., L.T. Taylor, A. Blakis, and E.F. Delong. 1998. Nitrospira-like bacteria associated with nitrite oxidation in freshwater aquaria. Applied and Environmental Microbiology 64 (1): 258–264.
Jiao, L., J. Wu, X. He, X. Wen, Y. Li, and Y. Hong. 2018. Significant microbial nitrogen loss from denitrification and anammox in the land-sea interface of low permeable sediments. International Biodeterioration and Biodegradation 135: 80–89.
Jorgensen, S.L., B. Hannisdal, A. Lanzén, T. Baumberger, K. Flesland, R. Fonseca, L. Øvreås, I.H. Steen, I.H. Thorseth, R.B. Pedersen, and C. Schleper. 2012. Correlating microbial community profiles with geochemical data in highly stratified sediments from the Arctic Mid-Ocean Ridge. Proceedings of the National Academy of Sciences 109 (42): E2846–E2855.
Ke, X., R. Angel, Y. Lu, and R. Conrad. 2013. Niche differentiation of ammonia oxidizers and nitrite oxidizers in rice paddy soil. Environmental Microbiology 15 (8): 2275–2292.
Keuter, S., M. Kruse, A. Lipski, and E. Spieck. 2011. Relevance of Nitrospira for nitrite oxidation in a marine recirculation aquaculture system and physiological features of a Nitrospira marina-like isolate. Environmental Microbiology 13 (9): 2536–2547.
Könneke, M., A.E. Bernhard, J.R. de la Torre, C.B. Walker, J.B. Waterbury, and D.A. Stahl. 2005. Isolation of an autotrophic ammonia-oxidizing marine archaeon. Nature 437 (7058): 543–546.
Kruse, M., S. Zumbrägel, E. Bakker, E. Spieck, T. Eggers, and A. Lipski. 2013. The nitrite-oxidizing community in activated sludge from a municipal wastewater treatment plant determined by fatty acid methyl ester-stable isotope probing. Systematic and Applied Microbiology 36 (7): 517–524.
LaMontagne, M.G., and P.A. Holden. 2003. Comparison of free-living and particle-associated bacterial communities in a coastal lagoon. Microbial Ecology 46 (2): 228–237.
Lebedeva, E.V., M. Alawi, F. Maixner, P.G. Jozsa, H. Daims, and E. Spieck. 2008. Physiological and phylogenetic characterization of a novel lithoautotrophic nitrite-oxidizing bacterium, 'Candidatus Nitrospira bockiana'. International Journal of Systematic and Evolutionary Microbiology 58 (Pt 1): 242–250.
Lebedeva, E.V., M. Alawi, C. Fiencke, B. Namsaraev, E. Bock, and E. Spieck. 2005a. Moderately thermophilic nitrifying bacteria from a hot spring of the Baikal rift zone. FEMS Microbiology Ecology 54: 297–306.
Lebedeva, E.V., S. Off, S. Zumbragel, M. Kruse, A. Shagzhina, S. Lucker, and E. Spieck. 2005b. Isolation and characterization of a moderately thermophilic nitrite-oxidizing bacterium from a geothermal spring. FEMS Microbiology Ecology 75 (2): 195–204.
Levipan, H.A., V. Molina, and C. Fernandez. 2014. Nitrospina-like bacteria are the main drivers of nitrite oxidation in the seasonal upwelling area of the Eastern South Pacific (Central Chile ~36°S). Environmental Microbiology Reports 6 (6): 565–573.
Lu, Z., and J. Gan. 2015. Controls of seasonal variability of phytoplankton blooms in the Pearl River Estuary. Deep Sea Research Part II Topical Studies in Oceanography 117: 86–96.
Lücker, S., M. Wagner, F. Maixner, E. Pelletier, H. Koch, B. Vacherie, T. Rattei, J.S.S. Damste, E. Spieck, D. Le Paslier, and H. Daims. 2010. A Nitrospira metagenome illuminates the physiology and evolution of globally important nitrite-oxidizing bacteria. Proceedings of the National Academy of Sciences 107 (30): 13479–13484.
Maixner, F., D.R. Noguera, B. Anneser, K. Stoecker, G. Wegl, M. Wagner, and H. Daims. 2006. Nitrite concentration influences the population structure of Nitrospira-like bacteria. Environmental Microbiology 8 (8): 1487–1495.
Meincke, M., E. Bock, D. Kastrau, and P.M.H. Kroneck. 1992. Nitrite oxidoreductase from Nitrobacter hamburgensis: redox centers and their catalytic role. Archives of Microbiology 158 (2): 127–131.
Ngugi, D.K., J. Blom, R. Stepanauskas, and U. Stingl. 2016. Diversification and niche adaptations of Nitrospina-like bacteria in the polyextreme interfaces of Red Sea brines. ISME Journal 10 (6): 1383–1399.
Nowka, B., S. Off, H. Daims, and E. Spieck. 2014. Improved isolation strategies allowed the phenotypic differentiation of two Nitrospira strains from widespread phylogenetic lineages. FEMS Microbiology Ecology 91 (3): fiu031.
Nunoura, T., Y. Takaki, M. Hirai, S. Shimamura, A. Makabe, O. Koide, T. Kikuchi, J. Miyazaki, K. Koba, N. Yoshida, M. Sunamura, and K. Takai. 2015. Hadal biosphere: Insight into the microbial ecosystem in the deepest ocean on Earth. Proc. Proceedings of the National Academy of Sciences 112 (11): E1230–E1236.
Pachiadaki, M.G., E. Sintes, K. Bergauer, J.M. Brown, N.R. Record, B.K. Swan, M.E. Mathyer, S.J. Hallam, L.-G. Purificacion, Y. Takaki, T. Nunoura, T. Woyke, G.J. Herndl, and R. Stepanauskas. 2017. Major role of nitrite-oxidizing bacteria in dark ocean carbon fixation. Science 358 (6366): 1046–1051.
Park, H., and D. Noguera. 2008. Nitrospira community composition in nitrifying reactors operated with two different dissolved oxygen levels. Journal of Microbiology and Biotechnology 18 (8): 1470–1474.
Pester, M., F. Maixner, D. Berry, T. Rattei, H. Koch, S. Lücker, B. Nowka, A. Richter, E. Spieck, E. Lebedeva, A. Loy, M. Wagner, and H. Daims. 2014. NxrB encoding the beta subunit of nitrite oxidoreductase as functional and phylogenetic marker for nitrite-oxidizing Nitrospira. Environmental Microbiology 16 (10): 3055–3071.
Ramanathan, B., A.M. Boddicker, T.M. Roane, and A.C. Mosier. 2017. Nitrifier gene abundance and diversity in sediments impacted by acid mine drainage. Frontiers in Microbiology 8: 2136.
Schloss, P.D., S.L. Westcott, T. Ryabin, J.R. Hall, M. Hartmann, E.B. Hollister, R.A. Lesniewski, B.B. Oakley, D.H. Parks, C.J. Robinson, J.W. Sahl, B. Stres, G.G. Thallinger, D.J. Van Horn, and C.F. Weber. 2009. Introducing mothur: open-source, platform-independent, community-supported software for describing and comparing microbial communities. Applied and Environmental Microbiology 75 (23): 7537–7541.
Shimizu, T., K. Ohtani, H. Hirakawa, K. Ohshima, A. Yamashita, T. Shiba, N. Ogasawara, M. Hattori, S. Kuhara, and H. Hayashi. 2002. Complete genome sequence of Clostridium perfringens, an anaerobic flesh-eater. Proceedings of the National Academy of Sciences 99 (2): 996–1001.
Smith, M.W., L.Z. Allen, A.E. Allen, L. Herfort, and H.M. Simon. 2013. Contrasting genomic properties of free-living and particle-attached microbial assemblages within a coastal ecosystem. Frontiers in Microbiology 4: 120.
Sorokin, D.Y., S. Lucker, D. Vejmelkova, N.A. Kostrikina, R. Kleerebezem, W.I. Rijpstra, J.S. Damste, D. Le Paslier, G. Muyzer, M. Wagner, M.C. van Loosdrecht, and H. Daims. 2012. Nitrification expanded: discovery, physiology and genomics of a nitrite-oxidizing bacterium from the phylum Chloroflexi. ISME Journal 6 (12): 2245–2256.
Spieck, E., C. Hartwig, I. McCormack, F. Maixner, M. Wagner, A. Lipski, and H. Daims. 2006. Selective enrichment and molecular characterization of a previously uncultured Nitrospira-like bacterium from activated sludge. Environmental Microbiology 8 (3): 405–415.
Ushiki, N., H. Fujitani, Y. Aoi, and S. Tsuneda. 2013. Isolation of Nitrospira belonging to Sublineage II from a Wastewater Treatment Plant. Microbes and Environments 28 (3): 346–353.
van Kessel, M.A., D.R. Speth, M. Albertsen, P.H. Nielsen, H.J. Op den Camp, B. Kartal, M.S. Jetten, and S. Lucker. 2015. Complete nitrification by a single microorganism. Nature 528 (7583): 555–559.
Vanparys, B., E. Spieck, K. Heylen, L. Wittebolle, J. Geets, N. Boon, and P. De Vos. 2007. The phylogeny of the genus Nitrobacter based on comparative rep-PCR, 16S rRNA and nitrite oxidoreductase gene sequence analysis. Systematic and Applied Microbiology 30 (4): 297–308.
Wang, Y., Y. Hong, M. Ma, S. Wu, H.J.M. Op den Camp, G. Zhu, W. Zhang, and F. Ye. 2019. Anthropogenic pollution intervenes the recovery processes of soil archaeal community composition and diversity from flooding. Frontiers in Microbiology 10: 2285.
Watson, S.W., E. Book, F.W. Valois, J.B. Waterbury, and U. Schlosser. 1986. Nitrospira marina gen. nov. sp. nov.: a chemolithotrophic nitrite-oxidizing bacterium. Archives of Microbiology 144 (1): 1–7.
Watson, S.W., and J.B. Waterbury. 1971. Characteristics of two marine nitrite oxidizing bacteria, Nitrospina gracilis nov. gen. nov. sp. and Nitrococcus mobilis nov. gen. nov. sp. Archives of Microbiology 77: 203–230.
Winogradsky, S. 1892. Contributions à la morphologie des organismes de la Archives of Science Biology St. Petersb. 1: 88–137.
Wu, J., Y. Hong, F. Guan, Y. Wang, Y. Tan, W. Yue, M. Wu, L. Bin, J. Wang, and J. Wen. 2016. A rapid and high-throughput microplate spectrophotometric method for fieldmeasurement of nitrate in seawater and freshwater. Scientific Reports 6 (1): 20165.
Zhai, W.D., M.H. Dai, W.J. Cai, Y.C. Wang, and Z.H. Wang. 2005. High partial pressure of CO2 and its maintaining mechanism in a subtropical estuary: the Pearl River estuary, China. Marine Chemistry 93 (1): 21–32.
Zhang, H., H. Fu, J. Wang, L. Sun, Y. Jiang, L. Zhang, and H. Gao. 2013. Impacts of nitrate and nitrite on physiology of Shewanella oneidensis. PLoS ONE 8 (4): e62629.
Zhang, Y., W. Xiao, and N.Z. Jiao. 2016. Linking biochemical properties of particles to particle-attached and free-living bacterial community structure along the particle density gradient from freshwater to open ocean. Journal of Geophysical Research – Biogeosciences 121 (8): 2261–2274.
Zimmerman, G.M., H. Goetz, and P.W. Mielke. 1985. Use of an improved statistical method for group comparisons to study effects of prairie fire. Ecology 1985: 66(2).
Funding
This work was funded by National Natural Science Foundation of China (No. 91851111, No. 31870100, and No. 42006122), Basic and Applied Basic Research Foundation of Guangdong Province (No. 2019B1515120066 and No. 2020A1515110597), Young Talent Research Project of Guangzhou Education Bureau (No. 202032795), and Research Foundation for Talented Scholars of Guangzhou University (Grant No. GU2017001).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Communicated by James L Pinckney
Supplementary Information
ESM 1
(DOCX 355 kb)
Rights and permissions
About this article
Cite this article
Hong, Y., Wu, J., Jiao, L. et al. Shifts in the Abundance and Community Composition of Particle-Associated and Free-Living Nitrospira Across Physicochemical Gradients in the Pearl River Estuary. Estuaries and Coasts 44, 1931–1945 (2021). https://doi.org/10.1007/s12237-021-00909-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12237-021-00909-2