Abstract
Oaxaca is one of the areas of early maize diversification, with 28 to 35 recognized agronomic races. Campesinos (farmers) cultivate at least ten different races in the Los Loxicha region. There is evidence of introgression between them; however, some traditional practices have promoted further differentiation evidenced by the presence of different landraces, clearly recognized by various morphological and phenological characteristics. Therefore, moderate levels of genetic diversity and low levels of population differentiation are expected. Seven communities belonging to two municipalities were selected in 2005. In total, 140 semistructured interviews were administered, inquiring about cultural practices, which included questions about maize seed, ear, and cob management and selection and maize cultivation. Based on interviews and permissions, 30 maize plants from each of 28 fields (milpas) with maize crop belonging to five landraces (Conejo, Hueso, Tablita, Tepecente, and Pinto) were sampled. From the 82 ISSR loci amplified, Hueso and Conejo exhibited the highest and lowest number of loci (70 and 56 loci, respectively); 72% of loci analyzed in the five populations were polymorphic. The Hueso landrace presented the highest percentage of polymorphism (73%) and Conejo the lowest (54%). The expected heterozygosity (HeT) for the five landraces was 0.225 ± 0.032, and the genetic diversity of Nei (HST) was 0.225 ± 0.034. The population differentiation values obtained with GST indicated that Tablita and Hueso were the least differentiated, while the most differentiated were Pinto and Tepecente. STRUCTURE analysis identified two genetically differentiated groups (k = 2), consistent with the length of their agricultural cycle (number of days until flowering). UPGMA analysis showed that milpas belonging to the same landrace and with a specific agricultural cycle length were grouped, regardless of their geographic distribution. These results indicate that traditional management of maize in the Los Loxicha region, particularly as it relates to the selection of ears and seeds and control of the agricultural cycle, maintains a balance between morphological differentiation, moderate levels of genetic diversity and gene flow, and low levels of population differentiation. This has contributed to the high richness and low evenness of agronomic races reported for the mountainous regions of the Sierra Madre del Sur and for Oaxaca.
Resumen
El manejo tradicional del maíz en la Sierra Sur, Oaxaca mantiene niveles moderados de diversidad genética y diferenciación poblacional entre las variedades locales Oaxaca es una de las áreas de diversificación temprana del maíz, con 28 a 35 razas agronómicas. En la región de Los Loxicha, los campesinos cultivan al menos 10 diferentes razas. Hay evidencia de introgresión entre ellas. Sin embargo, algunas prácticas tradicionales han promovido la presencia de diferentes variedades locales, claramente diferenciadas en varias características morfológicas y fenológicas, por lo que se espera encontrar niveles moderados de diversidad genética y bajos niveles de diferenciación genética entre ellas. Siete comunidades pertenecientes a dos municipios fueron seleccionadas en 2005. Se aplicaron 140 entrevistas semiestructuradas sobre el manejo y la selección de la semilla, olote y mazorca de maíz y prácticas culturales relacionadas con su cultivo. Con base en las entrevistas y permisos, se muestrearon 30 plantas de maíz procedentes de 28 campos de maíz (milpa)cultivados con cinco variedades locales (Conejo, Hueso, Tablita, Tepecente, Pinto). De los 82 loci de ISSR apmplificados para las cinco poblaciones, Hueso y Conejo exhibieron el mayor y menor número de loci (70 y 56, respectivamente); 72% de los loci analizados en las cinco poblaciones fueron polimórficos. La variedad local Hueso presentó el mayor porcentaje de polimorfismo (73%) y Conejo el menor (54%). La heterocigosis esperada (HeT) para las cinco variedades locales fue de 0.225 ± 0.032 y la diversidad genética de Nei (HST) fue de 0.225 ± 0.034. El valor de diferenciación poblacional obtenido con GST indicó que Tablita y Hueso fueron las menos diferenciadas, mientras que Pinto and Tepecente fueron las más diferenciadas. El análisis STRUCTURE identificó dos grupos genéticamente diferenciados (k= 2), consistentes con su ciclo agrícola. El análisis de UPGMA mostró que las milpas que pertenecen a la misma variedad local, con un ciclo agrícola o longitud de ciclo específico, se agruparon, independientemente de su distribución geográfica. Los resultados indican que el manejo tradicional del maiz en la región de Los Loxicha, en particular la selección de las mazorcas y las semillas y el control del ciclo agrícola, mantiene un equilibrio entre la diferenciación morfológica, los niveles moderados de diversidad genética y flujo génico, y niveles bajos de diferenciación poblacional, y ha contribuido a la alta riqueza y baja homogeneidad de razas agronómicas reportadas para las regiones montañosas de la Sierra Madre del Sur y para Oaxaca.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The state of Oaxaca, along with Jalisco, Michoacán, and Chiapas, is considered one of the areas where the early diversification of maize has occurred (Bellon and Berthaud 2004; Kato et al. 2009; Matsuoka et al. 2002; Prasanna 2010; Sánchez et al. 2000), and it is currently one of the states with the greatest diversity of maize races, a concept that refers to a group of related individuals with enough characteristics in common to permit their recognition as a group. “From the standpoint of genetics, a race is a group of individuals with a significant number of genes in common, major races having a smaller number in common than do subraces” (Anderson and Cutler 1942; Sánchez and Goodman 1992). Based on the 64 races reported by the Comisión Nacional para el Conocimiento y Uso de la Biodiversidad (CONABIO) (2011), 28 to 35 races are currently recognized for Oaxaca (Aragón-Cuevas 2006; Orozco-Ramírez et al. 2017), representing 44 to 55% of the diversity of maize in Mexico. Within races, there is also a spectacular morphological variation, recognized as local varieties, landraces, subraces, or traditional varieties, molded by the high outcrossing nature of maize, the environmental conditions, as well as the cultural practices that campesinos (farmers) have followed since ancestral times (Brush et al. 1988; Perales et al. 2003b). One of these cultural practices is concerned with ear and seed selection. Each year, campesinos select the best ears that will be used for the next planting cycle (large, with healthy seeds, full, and with a desirable color) and seeds (healthy, large, and complete). Campesinos’ selection promotes the diversification of landraces while preserving the desirable characteristics of ears and seeds associated with each landrace (e.g., color, seed texture, and size). The second cultural practice relates to seed movement, which occurs through the acquisition of seeds within the same community, as well as the introduction of seeds from other communities or regional markets, often through exchange, gift, or purchase. Seed movement occurs when many generations of inbreeding cause a decrease in yield (campesinos would say that the “maize is tired”) or when campesinos become interested in different varieties (Aragón-Cuevas et al. 2005; Aragón-Cuevas 2006; Bellon and Risopoulos 2001; Bellon et al. 2003; Bellon and Berthaud 2004; Bellon et al. 2011; Berthaud and Gepts 2004). The effect of seed movement is reflected in frequent introgression events between races, landraces, and introduced varieties. In addition to seed movement, natural phenomena such as pollen dispersal by insects and wind may influence morphological and genetic diversity (Bellon and Berthaud 2004; Hellin 2006).
The analysis of molecular data reveals that traditional maize management has maintained genetic variability and the identity of landraces (Bracco et al. 2009; Doebley and Goodman 1984; Hartings et al. 2008; Lia et al. 2009; Perales et al. 2005; Qi-Lun et al. 2008; Reif et al. 2006; Sánchez et al. 2000; Solomon et al. 2010; Vigouroux et al. 2005). Landraces are organized along a continuum such that their morphological and genetic differentiation (which refers to the level of physical and genetic isolation between populations, subpopulations, or demes) is influenced by cultural practices, geographical isolation, and agroecological conditions (Bellon et al. 2011; Hartings et al. 2008; Ignjatović-Micić et al. 2008; Ortega-Paczka 2003; Perales et al. 2005; Pressoir and Berthaud 2004; Qi-Lun et al. 2008; Solomon et al. 2010; Vigouroux et al. 2005; Vigouroux et al. 2008).
Different aspects of genetic diversity, genetic structure, and gene flow have been documented for some landraces and races in Oaxaca. Pressoir and Berthaud (2004) used molecular markers and quantitative traits to assess the role of human selection and gene flow in the population structure of different populations of white maize (e.g., Bolita race) from the Central Valleys of Oaxaca. Based on their agricultural cycle length, which is defined as the number of days until flowering (anthesis), López et al. (2010) estimated the levels of genetic diversity and genetic differentiation between the Zapalote Chico and Maiz Grande races, as well as other races growing in the Isthmus of Tehuantepec. More recently, Orozco-Ramírez et al. (2016) estimated the genetic differentiation of maize samples from villages at low and middle elevations in two adjacent municipalities with different ethnic compositions.
The Loxicha Region, located in the Sierra Madre del Sur of Oaxaca, is a cultural region represented by five municipalities inhabited by the Zapotec ethnic group, who have lived there for thousands of years. They have been involved in different adaptation processes, which are reflected not only in traditions and beliefs but also in ancestral knowledge and use of plants (Luna-José and Rendón-Aguilar 2008), the existence of traditional classification systems (Luna-José and Rendón-Aguilar 2012), and the presence of traditional agricultural systems, such as traditional shade-grown coffee (Montalvo 2006) and milpa in monoculture and polyculture (Aguilera 2005; Cardoso 2004). In this region, Rendón-Aguilar et al. (2015) found high levels of maize diversity, represented by at least 36 different landraces corresponding to 10 agronomic races, which represents 29–36% of those registered for Oaxaca (Aragón-Cuevas et al. 2005; Aragón-Cuevas 2006; Orozco-Ramírez et al. 2017; Soleri et al. 2006). They also found that campesinos have specific criteria to recognize and describe these 36 landraces as different morphological units. Campesinos understand that even when different landraces are distributed in the same area, they maintain their characteristics, but they also understand that some landraces can share characteristics of others. Based on this study, we wanted to know if traditional practices such as seed selection and seed exchange are the primary processes that promote morphological variation in maize and determine levels of gene flow and genetic diversity.
Our investigation sought to answer the following questions: What are the main traditional practices that generate and sustain intra- and interracial morphological diversity, and how do they work? What and how do these traditional practices affect levels of genetic diversity and genetic differentiation?
Methods
Study Sites
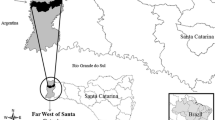
In 2005, we selected the localities of San Agustín Loxicha and Candelaria Loxicha, which are part of the cultural region known as “Los Loxicha” in the Sierra Sur de Oaxaca (Table 1, Fig. 1). This region is located between 15°56 46.2″ N and 90°33 15.5″ W, with elevations ranging from 100 to 2250 m. Candelaria, San Agustin, San Bartolome, San Baltazar, and Santa Catarina are the five municipalities that comprise the region. It is included in the Priority Terrestrial Region (PTR) 129 (Fig. 1), one of the CONABIO’s (the Mexican National Commission for the Knowledge and Use of Biodiversity) 152 defined Priority Terrestrial Regions (Arriaga et al. 2000). PTR 129 is regarded as one of the Oaxaca’s best-preserved areas, and it is characterized by the diversity of ecosystems, primarily semideciduous and temperate forests with a small proportion of montane cloud forest, and extensive connectivity among ecosystem types. Moreover, this region has a high level of species richness (Arriaga et al. 2000).
Vegetation types are also diverse and complex, occurring across the elevation zones as well as within small refuges (Chávez-González 2007). Seven types of vegetation are present in San Agustín Loxicha (montane cloud forest, tropical deciduous forest, tropical evergreen forest, tropical semideciduous forest, oak forest, pine forest, and pine-oak forest) and four are present in Candelaria Loxicha (montane cloud forest, tropical deciduous forest, tropical semideciduous forest, and pine-oak forest), because of the complex orography of the study area (Luna-José and Rendón-Aguilar 2008). Agriculture is the primary activity in both municipalities. Shifting cultivation is a common practice that involves planting different varieties of maize together, or just one variety, or maize associated or interplanted with different species and varieties of beans (Phaseolus vulgaris L., P. coccineus L., and Vigna unguiculata (L.) Walp.), squash (Cucurbita spp.), peanut (Arachis hypogaea L.), and roselle (Hibiscus sabdariffa L.). In the middle elevations, shade-grown coffee plantations are the most important economic resource (Luna-José and Rendón-Aguilar 2008) and shade trees provide a variety of nontimber products (food, medicine, wood, fuel, and forage) (Montalvo 2006) (Table 1).
Fieldwork
Selection and Interviews of Participants
We contacted the Comisariado de Bienes Comunales, the local authorities in each of these communities, established to protect their right of self-governance and self-determination as indigenous peoples. Each Comisariado is comprised of 6 to 9 people appointed by the inhabitants, who regulate all aspects related to land use including agriculture, cattle grazing, forest management, and the exploitation of natural resources. In some cases, Comisariados held special meetings to inform residents about our project, which were well attended. In others, they sent topiles (community members nominated by the Comisariado) to visit campesinos who they thought would be interested in participating. Topiles were responsible for explaining the purpose of the study. They spoke in Spanish or Zapoteco as appropriate. Authorities and potential collaborators inquired about the study’s benefits, including questions about financial renumeration. We responded that we would try to add some technical data to supplement the traditional knowledge that they already have, because their traditional practices have been responsible for preserving the diversity of native maize. We believed that recognizing their important role in preserving their native maize was sufficient to gain their approval. Previous studies had been conducted in the “Loxicha region” and in neighboring communities from 2002 to 2004 (Project # R38234-B, National Council of Science and Technology) (Rebollar-Dominguez 2004), so obtaining approval from the residents was simple. We had previously worked with some of the campesinos. Throughout the study, we followed local regulations, and we were careful to seek permission before accessing sites or fields that we wanted to visit, to collect and to photograph maize plants.
Semistructured interviews with 20 campesinos from each community were used to gather information about cultural practices (total N=140) (Alexiades 1996; Badstue et al. 2002; Smale et al. 1999). Participants were chosen by their availability and by the local authorities, with an eye toward selecting those campesinos who might be interested in participating or those with more experience. We let the authorities decide out of respect for local customs. The interviews included questions about seed management and the criteria for ear and seed selection, as well as cultural practices used during maize cultivation, such as type of cultivation (monoculture or polyculture), storage methods (ear and seed use), and harvesting methods.
DNA Analysis of Maize
Based on the interviews and previous information about the richness of maize races and landraces (Aguilera 2005; Cardoso 2004), as well as the local terms that campesinos use to describe various aspects of the maize crop, we will use the following terms: race, “a group of individuals who share enough characteristics to be recognized as a group” (Anderson and Cutler 1942); landrace, the variety shared by a group of campesinos regardless of where they live, based on specific characteristics (ear, plant height, and agricultural cycle length); milpa, a maize field planted in monoculture or polyculture with one or more landraces (for example, milpa of Hueso or milpa of Conejo); mata, a traditional cultivation practice in Mexico, where campesinos plant three to five seeds/hole. For the genetic analysis, the landraces were labeled as populations and milpas as the subpopulations, so the terms will be used interchangeably.
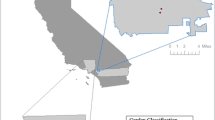
We chose five landraces based on their agricultural cycle length (short, middle, and long), as well as their elevation distribution within each of the three elevation zones established in this study: low, from 0 to 500 m; intermediate, from 501 to 1000 m; and high, over 1000 m: Conejo (short, intermediate); Tablita (middle, low); Tepecente (long, high), Pinto (long, high), and Hueso (middle, high). We also considered their distribution in at least two communities (Fig. 2) (Table 2). The Hueso landrace was an exception because it had a significant middle to highlands distribution, but it was found only in one community. It is important to note that all the landraces chosen for this study are widely distributed throughout the study area (Rendón-Aguilar et al. 2015), but we chose them based on the local knowledge of their presence in a specific elevation zone. Conejo, for example, was classified as a lowland landrace, Tepecente as a middle land landrace, and Hueso and Pinto as highland landraces. Tablita is a widely distributed landrace, but we only sampled it in lowland communities.
Each landrace was sampled for five to six milpas, for a total of twenty-eight milpas (Table 2). In 94% of them, campesinos had planted only one landrace/milpa. 30 individual plants were sampled from each for DNA analysis, following a diagonal line throughout the field, and considering a separation of 10 m between plants. We selected the most vigorous plant from each mata.
A foliar blade from each plant was selected, wrapped in aluminum foil, and stored in plastic bags with hermetic closure to separate leaves from different milpas (Table 2). All samples were duplicated. They were kept in two freezers at −80 °C, one at the Laboratory of the Division of Molecular Biology on the Universidad Autónoma Metropolitana Iztapalapa campus (UAM-I) and the other in the Molecular Biology Laboratory of the National Center for Research and Environmental Training (CENICA). The geographical location and elevation of each milpa was recorded using the Garmin Etrex global positioning system (GPS) model.
Laboratory Procedure for DNA Analysis
DNA was extracted from 834 individuals using the modified hexadecyl trimethyl ammonium bromide (CTAB) method (Doyle and Doyle 1990). The extract was electrophoresed in 0.8% agarose gels at 90 V for 90 minutes to test the DNA integrity. The gel was stained with ethidium bromide, and the reading was done with a photodocumenter (UVP BioImaging Systems). Dilutions were made at a concentration of 30 ng/μl.
D01 [(AG) 9 C] and D02 [(AC) 9 G] (Domenyuk et al. 2002) and M01 [(AC) 8 CG], M10 [(CA) 6 RG] (Osipova et al. 2003) oligos were chosen and tested. We chose three polymorphic ones, M01, D01, and D02. They were amplified using the following technique: PCR buffer (New England Biolabs) [1X] 2.5 μl, MgCl2 (New England Biolabs) [2.5 mM] 2.5 μl, dNTPs (New England Biolabs) [0.5 mM] 1.25 μl, Oligo (Invitrogen) [0.5 mM] 1.25 μl, DNA [30 ng] 0.5 or 1 μl (depending on the oligo and variety), Taq (New England Biolabs) [1U] 0.2 μl and Q solution (Qiagen) from 1 to 0.5 μl (depending on the oligo and variety). PCR products were separated by electrophoresis on 1.5% agarose gels with 1X TBE buffer at 90 V for 3 hrs. The gel was stained with ethidium bromide and bands were recorded in a data matrix (presence/absence) to obtain the image in the photo documenter.
Statistical Analysis
Maize Landrace Genetic Diversity and Population Structure
We studied five populations (landraces) (e.g., Conejo, Pinto, Tepecente, Hueso, and Tablita), as well as five or six subpopulations (milpas) within each population.
A presence/absence matrix containing data on observed bands was created and used to estimate genetic variation with dominant multi locus markers (ISSRs).
For all populations (landraces), the average percentage of polymorphic loci (%P), the expected population heterozygosity (He), the population genetic diversity (HS) of Nei (1972, 1973), and the Shannon index (IShannon) were estimated with the POPGENE v.1.32 (Yeh et al. 1999) and TEPGA v.1.3 (Miller 1997) programs.
One-way ANOVA for each index was used to compare the diversity of the different landraces. For parameters that did not follow the normal distribution, the Kruskal-Wallis test was used. Tukey tests were used to evaluate the differences between landraces (Zar 1999).
Genetic structure was investigated using different approaches:
(a) GST and FST values were estimated using the Nei method (1973) and the POPGENE program v. 3.2 (Yeh et al. 1999) to assess differences between landraces and milpas. In addition, an estimate of the FST, which is the coefficient of coancestry (θ), was calculated using Weir and Cockerham’s (1984) method which assumes that the population is in equilibrium and is defined as the probability that two individuals drawn at random from a population are identical by descent. As a result, it denotes the degree of differentiation between populations (Reynolds et al. 1983). When there is a hierarchy in the data, the TFPGA v.1.3 (Miller 1997) program provides two values of θ. In this case, it provides a value of θS that indicates the differentiation of the milpas within the landraces and a value of θP that indicates the differentiation between the landraces. This program also estimates the variation of θ by performing 5000 iterations of jackknife and bootstrapping procedures to generate 95% confidence intervals (CIs).
(b) Arlequin software v.3.5 was used to analyze genetic variation among and within milpas and landraces using analysis of molecular variance (AMOVA) (Excoffier 2009). The genetic membership of individuals in a milpa group was determined using a Bayesian clustering approach. With 10,000 iterations, a 10% burn-in period, and 10 replicates for each K group, STRUCTURE v.2.3 (Pritchard et al. 2000) was used to test for 1 to 10 groups. All other parameters were left at their default values. The method of Evanno et al. (2005) was used to estimate the most likely number of K groups.
Gene flow between landraces was estimated using the method proposed by Slatkin and Barton (1989) and McDermott and McDonald (1993) in POPGENE software v.3.2 (Yeh et al. 1999).
The same software was used to generate a dendrogram of genetic distances between subpopulations based on the Nei pairwise distances matrix and the arithmetic clustering algorithm’s unweighted pair group method (UPGMA). A Mantel test was performed to determine the significance of the correlation between the genetic distance (as estimated by TFPGA v.1.3) (Miller 1997) and the geographic distance of subpopulations estimated by geographic coordinates using ArcGIS 9.1. (ESRI 2005).
Cultural Practices Associated with Maize Cultivation
Data from interviews were analyzed using frequency tables to determine the role of traditional practices in the management, selection, and preservation of landraces, as well as their role in the levels and distribution of genetic diversity. Analyses were conducted at the community level.
Results
Genetic Diversity and Population Structure
Of the 82 loci amplified for 834 individuals, the Hueso landrace exhibited the highest number of loci (70) and Conejo showed only 56 loci. 72% (60% ± 1%) of the loci analyzed in the five populations were polymorphic. Hueso presented the highest (73%) and Conejo presented the lowest percentage (54%) of polymorphic loci. Between milpas, the highest percentage of polymorphism was found in a Hueso milpa (79%) and the lowest value was found in a Conejo milpa (43%) (Table 2).
The expected heterozygosity (HeT) for the five landraces was 0.225± 0.032 and the Nei genetic diversity (HST) was 0.225 ± 0.034. Only the expected heterozygosity between the Hueso and Conejo landraces (p = 0.004) revealed significant differences. Hueso had the highest level of unbiased genetic diversity (0.242), while Conejo had the lowest (0.180). Between milpas, a Hueso milpa had the highest value (0.298) and a Tepecente milpa had the lowest (0.144) (Table 2). The Shannon index obtained was 0.378 ± 0.209, and it followed the same pattern as genetic diversity. It ranged from 0.382 for the Hueso landrace to 0.289 for the Conejo landrace (Table 2). The population differentiation values obtained with GST (Table 3) revealed that Tablita and Hueso were the least differentiated while Pinto and Tepecente were the most differentiated. This value was similar to θ, which was obtained by evaluating each of the populations θP = 0.070, with a maximum value of 0.105 and a minimum value of 0.058. The value between subpopulations (milpas) is θS = 0.175 with a maximum value of 0.204 and a minimum value of 0.148, indicating that there was less genetic differentiation between landraces than between milpas.
The AMOVA revealed that the highest variance was found within milpas (73%), followed by landraces (13%), and between milpas within landraces (14%) (Table 3).
The STRUCTURE model-based analysis identified two genetically distinct groups (k= 2). The bar plot depicts the distribution of landraces in two groups (group one is colored red and group two is colored green) (Fig. 3). The landraces were classified based on their agricultural cycle length: Group 1 consisted of Conejo and Tablita landraces, distinguished by their short and middle agricultural cycle length, respectively; Group 2, consisted of Hueso, Tepecente, and Pinto. The first has a medium agricultural cycle length and is in the intermediate elevation zone. The others have a long cycle length and are found at higher elevations. Even after these groups were identified, populations belonging to Hueso and Tepecente landraces (Group 2) occurred within Group 1 (Fig. 3).
Inferred population structure for K = 2 groups. The red group is represented by the Conejo and Tablita landraces; the green group by the Hueso, Tepecente, and Pinto landraces. Each vertical line corresponds to one individual divided into two colored segments representing its estimated occurrence within each group (1, short agricultural cycle length; 2, middle agricultural cycle length; 3, long agricultural cycle length).
The estimated gene flow for the five landraces was greater than one. The Tablita and Hueso varieties had higher gene flow values than the others (Nm = 6.75 and 6.38, respectively), while the Pinto landrace had the lowest gene flow (3.40). This pattern is consistent with the GST and Θ values (Table 4).
Milpas belonging to the same landrace were grouped together, regardless of their geographic distribution, according to the dendrogram (UPGMA) obtained with the Nei genetic distance matrix. They were also classified based on their agricultural cycle length (Fig. 4). The long length landraces were divided into two groups at the bottom: Pinto and Tepecente. The third group included the two middle cycle length landraces, Tablita and Hueso, as well as the short cycle length landrace Conejo.
There was no statistical relationship between milpas’ genetic and geographical distances (Mantel test r = 0.129; p = 0.933). Even some milpas with the same landrace were geographically close, they were found to be genetically distant (e.g., Tablita23 and Tablita25 with the lowest genetic distance (0.0122), although geographically the closest were Hueso17 and Hueso18; the Conejo3 and Pinto8 milpas were more genetically distant (0.1162)).
The Role of Traditional Management in the Levels of Genetic Diversity and Genetic Differentiation Within and Between Maize Landraces
The levels of genetic diversity and population differentiation found in the five landraces are the result of traditional seed management and seed selection, as well as agroecological characteristics.
Management of Landraces
Campesinos identified the five landraces chosen for the current study based on phenotypic and agroecological characteristics. They mentioned that some landraces were cultivated primarily to eat elotes (corn on the cob) because they have a short agricultural cycle length (Conejo). Other landraces have a long agricultural cycle length but campesinos like them because the elotes have a sweeter flavor (Tablita, Hueso, Pinto), the kernels have a different flavor, it cooks faster (Tepecente), they are pest resistant (Hueso, Pinto, Tablita), or the ears have a higher yield (Hueso, Tablita). Campesinos avoid mixing landraces because they prefer to preserve the unique characteristics of each. Some campesinos dislike the flavor or the color of the tortillas produced by the mixtures. Others avoided mixing so that they could harvest the landraces at different times and maintain control over the landraces they already knew. Introgression between landraces is recognized as a common process by campesinos.
All the collaborators agreed that harvesting ears with different colored seeds is common when landraces are planted in nearby milpas or when two or more varieties are grown in the same milpa. On occasion, some experimented with mixing the seeds to produce improved morphological characteristics such as larger ears, thinner cobs, and heavier and larger seeds. However, campesinos mentioned that accidental mixing of varieties affects yields because the attributes for which they were selected can be lost. All campesinos preferred their landraces because they know them, and they are confident that seeds from these landraces will germinate and produce maize. They also believe that hybrids are more susceptible to rains, drought, or to the attack of weevils.
Seed Management
The majority of campesinos (85%) saved seeds from previous harvests, thereby preserving landraces. Campesinos stated that seed turnover occurs occasionally when maize yield falls. When this happens, they try to obtain seeds of the same landrace from people in the same community. Campesinos from Magdalena Loxicha, Juquilita, and San Vicente Yogondoy primarily mentioned this, whereas campesinos from San Francisco Loxicha, Los Horcones, and Río Molino obtain seeds from the same community or from other communities (Fig. 5a).
Traditional practices related to maize cultivation: a seed source: previous cycle (black), buying inside or outside the community (gray); b area destinated to maize production: less than one ha (black), more than one ha (gray); c crop system: polyculture (black), monoculture (gray). ML = Magdalena Loxicha, SFL = San Francisco Loxicha, SVY = San Vicente Yogondoy, J = Juquilita, H= Los Horcones, C = El Chilar, RM= Río Molino.
Seed Selection
When seeds are separated from ears for sowing the following year, almost all campesinos (96%) mentioned some specific characteristics: ears must be larger, of higher quality and color than the rest, and cobs must be thin. 40% of the campesinos use only the seeds from the middle of the ear. Campesinos stated that when selecting seeds, they look for characteristics such as large size, cleanliness, the highest degree of hardness, nice color, and homogeneity. Seeds must be free of fungi and intact (not chopped).
Other Cultural Practices
Campesinos cultivated maize in small plots of less than one ha, where one to three landraces may be cultivated (Fig. 5b). Maize was planted with different annual food crops and medicinal species as polyculture. The additional crops were destined for home consumption (Fig. 5c).
Discussion
Genetic Diversity
We found moderate levels of polymorphism as well as moderate levels of expected heterozygosity (He), genetic diversity (HS), and population genetic diversity (HSpob). Many studies have analyzed genetic diversity in maize samples from different areas of Mexico using different molecular markers, such as microsatellites (Reif et al. 2006, Rocandio-Rodríguez et al. 2014; Vega-Álvarez et al. 2017), isoenzymes (Doebley and Goodman 1984; Doebley et al. 1985; Sánchez et al. 2000), RFLPs (Dubreuil and Charcosset 1998), AFLPs (Hartings et al. 2008), and ISSRs (Hernández-Ramos et al. 2017; Santos et al. 2017). In Oaxaca, some studies with maize accessions used co-dominant molecular markers to address this topic (López et al. 2010; Pressoir and Berthaud 2004). Regardless of the molecular markers used, all studies reported medium to high genetic diversity levels. Our findings suggest that recurring processes for selecting seeds to plant each year maintained genetic diversity levels. Its allogamous nature is important in mitigating the potential to reduce genetic diversity through human selection. Although the presence of many transposing elements and intragenic recombination processes favors the evolution of new alleles and the maintenance of levels of genetic diversity in this species (Peterson 1997; Hartings et al. 2008). Human selection distributes and shapes it through the landraces, and occasional introgression allows this diversity to move throughout them. This is one of the reasons why genetic diversity reservoirs in rural communities in Mexico must be preserved. The more landraces there are, the greater the genetic diversity that will be retained in cultivated plants. Campesinos empirically understand this benefit when they cultivate different maize landraces.
Genetic Structure and Population Differentiation
The STRUCTURE model-based analysis identified two genetically distinct groups that clearly classified landraces based on agricultural cycle length and, to a lesser extent, elevation distribution. Campesinos appear to have experimented with both agroecological characteristics. Even when there was little evidence of gene flow via seed exchange or cross-pollination, campesinos’ selection criteria ensured the maintenance of their landraces. Conejo and Pinto, which are landraces with contrasting agricultural cycle lengths (e.g., short and long, respectively), exhibited a high percentage of membership in one of both groups (e.g., red or green, respectively). This is also consistent with their high levels of GST, but not with the elevation distribution (e.g., intermediate and high) of the landraces analyzed in the study. These findings are consistent with previous reports in which local varieties are clearly distinguished by vegetative characteristics, ear characteristics, and agricultural cycle length (Bellon and Brush 1994; Louette et al. 1997). However, our findings differ from previous studies in which landrace clustering reflected geographic origin or distribution (Hartings et al. 2008; Hernández-Ramos et al. 2017; Santos et al. 2017).
The genetic structure using dominant markers revealed moderate levels of population differentiation between landraces (GST= 0.0701) and high levels between milpas (GST= 0.1737). These findings imply that traditional landrace management through agricultural cycle length is the primary source of gene flow and occasional introgression occurs between them. Campesinos indicated that the coloring of kernels in landraces was an important selection factor each year. Seed exchange appears to be insignificant. It was practiced by few campesinos.
Milpas are the campesinos’ laboratories where they have experimented with their own varieties. Campesinos select better ears, cultivate different varieties in time and place, alone or mixed, retained the main characteristics that characterize each landrace by endogamy, and promoted their isolation or introgression, regardless of their geographical proximity. Few studies have examined population differentiation and gene flow in maize in Mexico. Most of the studies used codominant markers and found low levels of genetic differentiation (Doebley and Goodman 1984; Perales et al. 2005; Pressoir and Berthaud 2004; Sánchez et al. 2000).
Some landraces appear to be more susceptible to introgression than others (Chávez-Servia et al. 2011; Rendón-Aguilar et al. 2015). Hueso and Tepecente, members of Group 2, appear to have exhibited major introgression levels. Introgression has been observed between agronomic races (Rendón-Aguilar 2011; Rendón-Aguilar et al. 2015; Sánchez and Goodman 1992) and landraces (Hartings et al. 2008; Lia et al. 2009).
Rendón-Aguilar (2011) and Rendón-Aguilar et al. (2015) reported that the Tepecente landrace belonged to three agronomic races: Tepecente, Olotillo, and Tuxpeño. All of them exhibited high levels of introgression, consistent with the pattern detected in the STRUCTURE analysis and the GST and Nm values. This means that the Tepecente landrace was highly susceptible to introgression events regardless of the agronomic race to which it belonged. Hernández-Ramos et al. (2017) discussed the possible role of interracial origin in the morphological, phenological, and agroecological characteristics and local adaptation of local varieties.
Genetic Distance and Gene Flow
The dendrogram based on Nei genetic distances separated the landraces, supporting the explanation that campesinos’ selection was effective in separating and maintaining them as distinct units based on their morphological characteristics and agricultural cycle length. Carvalho et al. (2004) used ISSR and RAPDs to analyze 81 native maize varieties in Brazil and discovered an association between three groups that were correlated with endosperm colors and fourteen subgroups that were characterized by their flowering season and other ear characteristics. Molecular data revealed that small-scale campesinos’ maize management contributed to the preservation of genetic variability, and that due to isolation in the field, the various identities were preserved (Carvalho et al. 2004). Other authors, such as Pressoir and Berthaud (2004), concluded that a maize landrace should not be considered a separate entity but rather an open genetic system and emphasized the importance of campesinos in determining gene flow between creole populations of maize. Our study supports these findings.
We found no significant relationship between genetic distances and geographical isolation. Bracco et al. (2009) and Orozco-Ramírez et al. (2016) reported similar findings. We propose that, regardless of geographical distance or elevation distribution between milpas and landraces, natural pollination is the most important mechanism that has reduced population differentiation and that gene flow between landraces in the Los Loxicha region was determined by landrace agricultural cycle length. This gene flow could be caused by introgression between short and middle agricultural cycle length landraces and the middle with long agricultural cycle length landraces. In this context, sowing dates are important because they allow some levels of gene flow to occur between individuals of two landraces with different agricultural cycle lengths. The Loxicha region is characterized by seasonal agriculture with sowing dates limited to May–June. Torres-Morales et al. (2022) compared agronomic and morphological data between seven agronomic races and found that they clustered primarily based on flowering dates. They did, however, show significant complexity in their groupings and evidence of introgression processes. Previous research has identified seed exchange as an important source of gene flow (Bracco et al. 2009; Carvalho et al. 2004), but this practice was not common in the Loxicha region because most campesinos save the seeds from the previous cycle (Badstue et al. 2002; Brush and Perales 2007; Fenzi et al. 2017).
Traditional Management of Landraces
Traditional maize management by campesinos in the Loxicha region has promoted the presence of landraces that can be distinguished by morphological and agroecological characteristics, as well as moderate levels of genetic diversity and low levels of genetic differentiation (Aguilera 2005; Cardoso 2004). Traditional landrace management has relied on recognizing morphological characteristics such as ear size, seed size, and color, as well as agricultural cycle length. Campesinos recognized that some landraces grew better only within one elevation zone (low or high). Thus, Hueso or Pinto is a highland landrace, whereas Conejo is a lowland landrace. However, elevation zone distribution was not a factor in landrace differentiation.
We found that seed movement was not an important mechanism of gene flow because campesinos still select and store ears and seeds from the previous cycle. This pattern differs from other studies in which seed movement via exchange is a significant and determinant source of gene flow (Badstue et al. 2006; Bellon and Risopoulos 2001; Bellon et al. 2003; Bellon et al. 2011; Louette 1995; Rice et al. 1998; Smale et al. 1999).
Although some levels of introgression occur between landraces (ears exhibit a mixture of seeds of different colors known as “pintas,” “mezcladas,” or “injertadas”), campesinos separate seeds of different colors and plant them separately. These activities promote the preservation of the characteristics of each landrace (Louette 1995; Ortega-Paczka 2003).
Other cultural practices, such as the area selected for maize production, the preference for maize in a polyculture rather than a monoculture, and production primarily for self-consumption, contribute to maintaining maize landraces because campesinos obtain more than economic benefits, as Escobar indicated previously (2006). Campesinos prefer more than one color, flavor, or other organoleptic property, and these cultural preferences determine the presence of different landraces. For example, campesinos mentioned they like to eat seeds from all landraces, but they prefer white maize for tortillas. The presence of different landraces in the same field can either promote or inhibit introgression, depending on which of them the campesinos decide to plant and where they are planted within the milpa. In our study, 94% of the plots were planted with only one landrace, but this type of management was documented during interviews rather than through direct observation.
The susceptibility of maize landraces to insect pests during storage is becoming more of an issue, which may be linked to elevation (Perales et al. 2003a). Landraces in the Loxicha region exhibited this pattern (Callejas-Chavero et al. 2018). It would be interesting to conduct additional research on this topic to examine the differences in pest responses.
Oaxaca has the most maize races in Mexico (Orozco-Ramírez et al. 2017). The Sierra Madre del Sur, which encompasses a significant portion of Oaxaca and is one of the Mexico’s Priority Terrestrial Regions, has been identified as one of the mountain areas with high richness and low evenness of agronomic maize races. Los Loxicha region, as a small part of this important mountain region, clearly demonstrates this richness and complexity as we found a large number of landraces located within a few kilometers of each other (Aguilera 2005; Cardoso 2004; Rendón-Aguilar et al. 2011, 2015). The five landraces exhibited moderate levels of genetic diversity and population differentiation.
Conclusion
Campesinos (farmers) in the Loxicha region preserved the morphological and genetic diversity of maize landraces, as well as low levels of population differentiation, through a variety of traditional practices. They kept landraces as morphological units through seed selection, which may result in relatively high endogamy and low genetic diversity. Agricultural cycle length of the landraces determines gene flow and occasional introgression events, which occurs between milpas or within the same milpa when campesinos planted two or more landraces. Campesinos regulate genetic diversity and landrace differentiation empirically. Introgression processes occur when they plant landraces with similar agricultural cycle lengths together. This planting pattern is an empirical strategy for maintaining genetic diversity and low levels of landrace differentiation. Planting landraces with different agricultural cycle lengths promotes differentiation.
Each published paper adds to the body of evidence for genetic diversity in maize. Maize has a remarkable genome in which new alleles emerge through transposons and intragenic recombination processes. Traditional management shapes this genetic diversity to produce dozens of landraces and agronomic races in its center of origin, transforming them into genetic and morphological reservoirs that can be used in the present and future. Maize evolution continues, and the introgression-differentiation processes allow for genome rearrangement and the emergence of new landraces. Monitoring for in situ conservation areas is necessary in Mexico. Our study shows the importance of local landraces to campesinos and the need for landrace production programs rather than the introduction of improved varieties or hybrids.
Data Availability
The authors confirm that the data supporting the findings of this study are available within the article and its supplementary materials.
Literature Cited
Aguilera, L. C. A. 2005. Evaluación de los recursos vegetales en tres Municipios zapotecos de la Sierra Madre del Sur de Oaxaca. Social service final report. CDMX (Mexico City), México: Universidad Autónoma Metropolitana-Unidad Iztapalapa
Alexiades, N. M. 1996. Collecting ethnobotanical data: An introduction to basic concepts and techniques. In: Selected guidelines for ethnobotanical research: A field Manual, ed. N. M. Alexiades, 53-94. New York: The New York Botanical Garden
Anderson, E., and H. C. Cutler. 1942. Races of Zea mays. I. Their recognition and classification. Annals of the Missouri Botanical Garden 29: 69–88
Aragón-Cuevas, F., S. Taba, F. H. Castro-García, J. M. Hernández, J. M. Cabrera-Toledo, L. Osorio-Alcalá, and N. Dillánes-Ramírez. 2005. In situ conservation and use of local maize races in Oaxaca, Mexico: A participatory and decentralized approach. In: Latin American Maize Germoplasm Conservation: Regeneration, In situ Conservation, Core Subsets, and Prebreeding; Proceedings of a Workshop held at CIMMYT, ed. S. Taba, 26–38. México: CIMMYT
Aragón-Cuevas F. 2006. Actualización de la información sobre los maíces criollos de Oaxaca. Bases de datos SNIB-CONABIO, proyecto CS002. DF, México: Instituto Nacional de Investigaciones Forestales, Agrícolas y Pecuarias. http://www.conabio.gob.mx/institucion/proyectos/resultados/InfCS002.pdf
Arriaga L, J. M. Espinoza, C. Aguilar, E. Martínez, L. Gómez, and E. Loa. 2000. Regiones terrestres prioritarias de México. DF, México: Comisión Nacional para el Conocimiento y Uso de la Biodiversidad
Badstue, L. B., M. R. Bellon, X. Juárez, X. I. M. Rosas, and A. M. Solano. 2002. Social relations and seed transactions among small-scale maize peasants’ access to maize landraces in the Central Valleys of Oaxaca, México. DF, México: Centro Internacional para el Mejoramiento de Maíz y Trigo
Badstue, L. B., M. R. Bellon, J. Berthaud, X. Juárez, I. M. Rosas, A. M. Solano, and A. Ramírez. 2006. Examining the role of collective action in an informal seed system: A case study from the Central Valleys of Oaxaca, México. Human Ecology 34(2): 249–273. https://doi.org/10.1007/s10745-006-9016-2
Bellon, M. R., and S. B. Brush. 1994. Keepers of maize in Chiapas, Mexico. Economic Botany 48(2): 196–209. https://doi.org/10.1007/BF02908218
Bellon, M. R., and J. Risopoulos. 2001. Small-scale peasants expand the benefits of improved maize germplasm: A case study from Chiapas, México. World Development 29(5): 799–811. https://doi.org/10.1016/S0305-750X(01)00013-4
Bellon, M. R., J. Berthaud, M. Smale, J. A. Aguirre, S. Taba, F. Aragón, J. Díaz, and H. Castro. 2003. Participatory landrace selection for on-farm conservation: An example from the Central Valleys of Oaxaca, México. Genetic Resources and Crop Evolution 50: 401–416. https://doi.org/10.1023/A:1023967611495
Bellon, M. R., and J. Berthaud. 2004. Transgenic maize and the evolution of landrace diversity in México. The importance of peasants´ behavior. Plant Physiology 134(3): 883–888. https://doi.org/10.1104/pp.103.038331
Bellon, M. R., D. Hodson, and J. Hellin. 2011. Assessing the vulnerability of traditional maize seed systems in Mexico to climate change. Proceedings of the Natural Academy of Sciences 108(33): 13432–13437. https://doi.org/10.1073/pnas.1103373108
Berthaud, J., and P. Gepts. 2004. Assessment of effects on genetic diversity. In: Maize and biodiversity: The effects of transgenic maize in Mexico, ed. Commission for Environmental Cooperation. Montreal, Canada: Commission for Environmental Cooperation
Bracco, M., V. V. Lia, A. M. Gottlieb, J. Cámara, and L. Poggio. 2009. Genetic diversity in maize landraces from indigenous settlements of Northeastern Argentina. Genetica 135(1): 39–49. https://doi.org/10.1007/s10709-008-9252-z
Brush, S. B., M. Bellon, and E. Schmidt. 1988. Agricultural development and maize diversity in Mexico. Human Ecology 16(3): 307–326. http://www.jstor.org/stable/4602889 (29 June 2023)
Brush, S. B., and H. R. Perales. 2007. A maize landscape: Ethnicity and agro-biodiversity in Chiapas Mexico. Agriculture, Ecosystems & Environment 121(3): 211–221. https://doi.org/10.1016/j.agee.2006.12.018
Callejas-Chavero, A., A. Cornejo-Romero, A. Serrato-Díaz, and B. Rendón-Aguilar. 2018. Diversity and trophic structure of insects associated with grains of three maize landraces in San Agustín Loxicha, Oaxaca, Mexico. Entomological Science 22: 42-47. https://doi.org/10.1111/ens.12335
Cardoso, D. J. 2004. Estudio etnobotánico del agroecosistema milpa en la comunidad de Candelaria Loxicha, Oaxaca. Social service final report. CDMX (Mexico City), México: Universidad Autónoma Metropolitana-Unidad Xochimilco
Carvalho, V. P., C. F. Ruas, J. M. Ferreira, R. M. P. Moreira, and P. M. Ruas. 2004. Genetic diversity among maize (Zea mays L.) landraces assessed by RAPD markers. Genetics and Molecular Biology 27(2): 228–236. https://doi.org/10.1590/S1415-47572004000200017
Chávez-González, A. D. 2007. Variación aloenzimática y estructura genética de Ceratozamia longifolia Miquel (Zamiaceae) en la Sierra Madre del Sur, Oaxaca. Tesis de licenciatura. Facultad de Estudios Superiores Zaragoza. CDMX (Mexico City), México: Universidad Nacional Autónoma de México. https://ru.dgb.unam.mx/handle/DGB_UNAM/TES01000616548 (2 June 2023)
Chávez-Servia, J. L., P. Diego-Flores, and J. C. Carrillo-Rodríguez. 2011. Complejos raciales de poblaciones de maíz evaluadas en San Martín Huamelulpan, Oaxaca. Ra Ximhai 7(1). http://www.redalyc.org/articulo.oa?id=46116742010
CONABIO. 2011. Proyecto Global de Maíces Nativos. CDMX (Mexico City), México: Comisión Nacional para el Conocimiento y Uso de la Biodiversidad; Instituto Nacional de Investigaciones Forestales, Agrícolas y Pecuarias; Instituto Nacional de Ecología y cambio Climático. https://biodiversidad.gob.mx/diversidad/proyectoMaices (11 May 2023)
Doebley, J. F., and M. M. Goodman. 1984. Isoenzymatic variation in Zea (Gramineae). Systematic Botany 9(2): 203–218. https://doi.org/10.2307/2418824
Doebley, J. F., M. M. Goodman, and C. W. Stuber. 1985. Isozyme variation in the races of maize from Mexico. American Journal of Botany 72(5): 629-639. https://doi.org/10.2307/2443674
Domenyuk, V. P., T. G. Verbitskaya, A. A. Belousov, and Y. M. Sivolap. 2002. Marker analysis of quantitative traits in maize by ISSR-PCR. Russian Journal of Genetics 38: 1161–1168. https://doi.org/10.1023/A:1020600804246
Doyle, J. J., and J. L. Doyle. 1990. Isolation of plant DNA from fresh tissue. Focus 12(1): 413–415
Dubreuil, P., and A. Charcosset. 1998. Genetic diversity within and among maize populations: A comparison between isozyme and nuclear RFLP loci. Theoretical and Applied Genetics 96(5): 577–587. https://doi.org/10.1007/s001220050776
Escobar, D.A. 2006. Valoración campesina de la diversidad del maíz. Estudio de caso de las comunidades indígenas en Oaxaca, México. PhD dissertation. Spain: Universidad Autónoma de Barcelona
ESRI. 2005. ArcGIS 9: What is ArcGIS 9.1. Redlands, California: Environmental Systems Research Institute
Evanno G, S. Regnaut, and J. Goudet. 2005. Detecting the number of clusters of individuals using the software STRUCTURE: assimilation study. Molecular Ecology 14: 2611–2620. https://doi.org/10.1111/j.1365-294X.2005.02553.x
Excoffier, L. 2009. Arlequin ver. 3.5.1.2 computational and molecular population genetics lab. Berne, Switzerland: Institute of Ecology and Evolution, University of Berne Swiss Institute of Bioinformatics. February 2010 http://cmpg.unibe.ch/software/arlequin3/
Fenzi, M., D. Jarvis, L. Arias Reyes, L. Latournerie Moreno, and J. Tuxill. 2017. Longitudinal analysis of maize diversity in Yucatan, Mexico: Influence of agro-ecological factors on landraces conservation and modern variety introduction. Plant Genetic Resources 15(1): 51–63. https://doi.org/10.1017/S1479262115000374
Hartings, H., N. Berardo, G. F. Mazzinelli, P. Valoti, A. Verderio, and M. Motto. 2008. Assessment of genetic diversity and relationships among maize (Zea mays L.) Italian landraces by morphological traits and AFLP profiling. Theoretical and Applied Genetics 117(6): 831–842. https://doi.org/10.1007/s00122-008-0823-2
Hellin, J. 2006. Agricultores mexicanos de escasos recursos cultivan más maíz "mejorado" de lo que podría creerse. CIMMYT E-News 3(2)
Hernández-Ramos, M. A., L. A. Rodríguez-Larramendi, F. Guevara-Hernández, M. D. L. A. Rosales-Esquinca, R. Pinto-Ruiz, and R. Ortiz-Pérez. 2017. Caracterización molecular de maíces locales de la Reserva de la Biosfera La Sepultura, México. Agronomía Mesoamericana 28(1): 69–83. https://doi.org/10.15517/am.v28i1.21612
Ignjatović-Micić, D., S. Mladenović-Drinić, A. Nikolić, and V. Lazić-Jančić. 2008. SSR analysis for genetic structure and diversity determination of maize local populations from former Yugoslavia territories. Russian Journal of Genetics 44(11): 1317–1324. https://doi.org/10.1134/S1022795408110100
Kato, T. A., C. Mapes, L. M. Mera, J. A. Serratos, and R. Bye. 2009. Origen y diversificación del maíz: Una revisión analítica. México: Universidad Nacional Autónoma de México y Comisión Nacional para el Conocimiento y Uso de la Biodiversidad
Lia, V., L. Poggi, and V. A. Confalonieri. 2009. Microsatellite variation in maize landraces from Northwestern Argentina: Genetic diversity, population structure and racial affiliations. Theoretical and Applied Genetics 119(6): 1053–1067. https://doi.org/10.1007/s00122-009-1108-0
López, R. G., A. Santacruz, A. Muñoz, F. Castillo, L. Córdova, and H. Vaquera. 2010. Perfil Isoenzimático de maíces nativos del Istmo de Tehuantepec, Oaxaca, México. I. Caracterización de grupos. Revista Fitotecnia Mexicana 33(1): 1–10. https://doi.org/10.35196/rfm.2010.1.1
Louette, D. 1995. Intercambio de semillas entre agricultores y flujo genético entre variedades de maíz en sistemas agrícolas tradicionales. In: Memoria del Foro: Flujo Genético entre Maíz Criollo, Maíz Mejorado y Teocintle: Implicaciones para el Maíz Transgénico, eds. J. A. Serratos, M. C. Willcox, and F. Castillo, 60-71. Estado de México: INIFAP-CIMMYT-CNBA
Louette, D., A. Charrier, and J. Berthaud. 1997. In situ conservation of maize in Mexico: Genetic diversity and maize seed management in a traditional community. Economic Botany 51(1): 20–38. https://doi.org/10.1007/BF02910401
Luna-Jose, A de L . 2006. Cuantificación de los recursos vegetales recolectados en tres municipios zapotecos de la Sierra Madre del Sur, Oaxaca. Tesis de maestría. Montecillos, Mexico: Colegio de Posgraduados
Luna-Jose, A de L, and B. Rendón-Aguilar. 2008. Recursos vegetales útiles en diez comunidades de la Sierra Madre del Sur, Oaxaca, México. Polibotánica (26): 193–242. https://www.redalyc.org/articulo.oa?id=62102611
Luna-José, A de L., and B. Rendón-Aguilar. 2012. Traditional knowledge among Zapotecs of Sierra Madre Del Sur, Oaxaca. Does it represent a base for plant resources management and conservation? Journal of Ethnobiology and Ethnomedicine 8: 24 https://doi.org/10.1186/1746-4269-8-24
Matsuoka, Y., Y. Vigouroux, M. M. Goodman, J. Sánchez, E. Buckler, and J. Doebley. 2002. A single domestication for maize shown by multilocus microsatellite genotyping. Proceedings of the Natural Academy of Sciences 99(9): 6080–6084. https://doi.org/10.1073/pnas.052125199
McDermott, J. M., and B. A. McDonald. 1993. Gene flow in plant pathosystems. Annual Review of Phytopathology 31(1): 353–373. https://doi.org/10.1146/annurev.py.31.090193.002033
Miller, M. P. 1997. Tools for population genetic analyses (TFPGA) 1.3: A Windows program for the analysis of allozyme and molecular population genetic data. Computer software distributed by the author
Montalvo, L. 2006. Manejo y estructura de la vegetación leñosa de los cafetales en la Sierra Madre del Sur, Oaxaca. Tesis de maestría. Montecillos, Mexico: Colegio de Posgraduados
Nei, M. 1972. Genetic distance between populations. The American Naturalist 106(949): 283–292. https://www.jstor.org/stable/2459777
Nei, M. 1973. Analysis of gene diversity in subdivided populations. Proceedings of the National Academy of Sciences 70(12): 3321–3323. https://doi.org/10.1073/pnas.70.12.3321
Orozco-Ramírez, Q., J. Ross-Ibarra, A. Santacruz-Varela, and S. Brush. 2016. Maize diversity associated with social origin and environmental variation in Southern Mexico. Heredity 116: 477–484. https://doi.org/10.1038/hdy.2016.10
Orozco-Ramírez, Q., H. Perales, and R. J. Hijmans. 2017. Geographical distribution and diversity of maize (Zea mays L. subsp. mays) races in Mexico. Genetic Resources and Crop Evolution 64: 855–865 https://doi.org/10.1007/s10722-016-0405-0
Ortega-Paczka, R. 2003. La diversidad del maíz en México. In: Sin maíz no hay país, eds. C. Esteva, and C. Marielle. México: Consejo Nacional para la Cultura y las Artes, Dirección General de Culturas Populares e Indígenas
Osipova, E. S., O. V. Koveza, A. V. Troitskij, Y. I. Dolgikh, Z. B. Shamina, and S. A. Gostimskij. 2003. Analysis of specific RAPD and ISSR fragments in maize (Zea mays L.) somaclones and development of SCAR markers on their basis. Russian Journal of Genetics 39(12): 1412–1419. https://doi.org/10.1023/B:RUGE.0000009156.74246.bc
Perales, H., S. B. Brush, and C. O. Qualset. 2003a. Landraces of maize in Central Mexico: An altitudinal transect. Economic Botany 57: 7–20. https://doi.org/10.1663/0013-0001(2003)057[0007:LOMICM]2.0.CO;2
Perales, H., S. B. Brush, and C. O. Qualset. 2003b. Dynamic Management of Maize Landraces in Central Mexico. Economic Botany 57: 21–34. https://doi.org/10.1663/0013-0001(2003)057[0021:DMOMLI]2.0.CO;2
Perales, H., B. F. Benz, and S. B. Brush. 2005. Maize diversity and ethnolinguistic diversity in Chiapas, México. Proceedings of the Natural Academy of Sciences 3(102): 949–954. https://doi.org/10.1073/pnas.0408701102
Peterson, P.A. 1997. Mechanisms contributing to genetic diversity in maize populations. In: The genetics and exploitation of heterosis in crops: An International Symposium, eds. J. G. Coors, and S. Pandey, 149–161. USA: ASA, CSSA, and SSSA Books. https://doi.org/10.2134/1999.geneticsandexploitation.c14
Prasanna, B. M. 2010. Phenotypic and molecular diversity of maize landraces: Characterization and utilization. Indian Journal of Genetics and Plant Breeding 70(4): 315–327
Pressoir, G., and J. Berthaud. 2004. Patterns of population structure in maize landraces from the Central Valleys of Oaxaca in Mexico. Heredity 92: 88–9 https://doi.org/10.1038/sj.hdy.6800387
Pritchard, J. K., M. Stephens, and P. Donnelly. 2000. Inference of population structure using multilocus genotype data. Genetics 155: 945–959. https://doi.org/10.1093/genetics/155.2.945
Qi-Lun, Y., F. Ping, K. Ke-Cheng, and P. Guang-Tang. 2008. Genetic diversity based on SSR markers in maize (Zea mays L.) landraces from Wuling mountain region in China. Journal of Genetics 87(3): 287–291. https://doi.org/10.1007/s12041-008-0046-y
Rebollar-Domínguez, S. 2004. Evaluación de los recursos vegetales de tres municipios zapotecos en la Sierra Madre del Sur de Oaxaca. M. en C. Proyecto R38234-B, financiado por el Consejo Nacional de Ciencia y Tecnología (CONACYT)
Reif, J. C., M. L. Warburton, X. C. Xia, D. A. Hoisington, J. Crossa, S. Taba, J. Muminović, M. Bohn, M. Frisch, and A. E. Melchinger. 2006. Grouping of accessions of Mexican races of maize revisited with SSR markers. Theoretical and Applied Genetics 113(2): 177–185. https://doi.org/10.1007/s00122-006-0283-5
Rendón-Aguilar, B. 2011. Diversidad y distribución altitudinal de maíces nativos en la región de los Loxicha, Sierra Madre del Sur Oaxaca. Informe final CONABIO proyecto FZ003 México D.F. http://www.conabio.gob.mx/institucion/cgi-bin/datos2.cgi?Letras=FZ&Numero=3 (19 April 2023)
Rendón-Aguilar, B., V. Aguilar-Rojas, M. C. Aragón- Martínez, J. F. Ávila-Castañeda, L. A. Bernal- Ramírez, D. Bravo-Avilez, G. Carrillo-Galván, A. Cornejo-Romero, E. Delgadillo-Durán, G. Hernández-Cárdenas, M. Hernández-Hernández, A. López-Arriaga, J. M. Sánchez-García, E. Vides-Borrell, and R. Ortega-Packzca. 2015. Diversidad de maíz en la Sierra Sur de Oaxaca, México. Polibotánica 39: 151–174. https://www.redalyc.org/articulo.oa?id=62137097009
Reynolds, J., B. S. Weir, and C. C. Cockerham. 1983. Estimation of the coancestry coefficient: Basis for a short-term genetic distance. Genetics 105(3): 767–779. https://doi.org/10.1093/genetics/105.3.767
Rice, E., M. Smale, and J. L. Blanco. 1998. Farmers’ use of improved seed selection practices in Mexican maize: Evidence and issues from the Sierra de Santa Marta. World Development 26(9): 1625–1640. https://doi.org/10.1016/S0305-750X(98)00079-5
Rocandio-Rodríguez, M., A. Santacruz-Varela, L. Córdova-Téllez, H. López-Sánchez, F. Castillo-González, R. Lobato-Ortiz, and J. J. García-Zavala. 2014. Detection of genetic diversity of seven maize races from the high central valleys of Mexico using microsatellites. Maydica 59(2): 144–151. https://journals-crea.4science.it/index.php/maydica/article/view/990 (19 April 2023)
Sánchez G. J. J., and M. M. Goodman. 1992. Relationships among the Mexican races of maize. Economic Botany 46: 72–85. https://doi.org/10.1007/BF02985256
Sánchez G. J. J., M. M. Goodman, and C. M. Stuber. 2000. Isozymatic and morphological diversity in the races of maize of Mexico. Economic Botany 54(1): 43–59. https://doi.org/10.1007/BF02866599
Santos L. F. C., R. H. Andueza-Noh, E. S. Ruíz, L. L. Moreno, R. Garruña, J. O. Mijangos-Cortes, and J. Martínez-Castillo. 2017. Characterization of the genetic structure and diversity of maize (Zea mays L) landrace populations from Mexico. Maydica 62(2): 7. https://journals-crea.4science.it/index.php/maydica/article/view/1576 (19 April 2023)
Slatkin, M., and N. H. Barton. 1989. A comparison of three indirect methods for estimating average levels of gene flow. Evolution 43(7): 1349–1368. https://doi.org/10.1111/j.1558-5646.1989.tb02587.x
Smale, M., A. Aguirre, M. Bellon, J. Mendoza, and I. Manuel. 1999. Farmer management of maize diversity in the Central Valleys of Oaxaca, México. CIMMYT Economics Working Paper México, D.F: CIMMYT. 99–09. https://repository.cimmyt.org/bitstream/handle/10883/991/68263.pdf?sequence=1&isAllowed=y (19 April 2023)
Soleri, D., D. A. Cleveland, and F. Aragón-Cuevas. 2006. Transgenic crops and crop varietal diversity: the case of maize in Mexico. BioScience 56: 503–13. https://doi.org/10.1641/0006-3568(2006)56[503:TCACVD]2.0.CO;2
Solomon, K. F., I. Martin, and A. Zeppa. 2010. Temporal genetic structure patterns in tropical maize populations under reciprocal recurrent selection. Euphytica 176(2): 239–249. https://doi.org/10.1007/s10681-010-0238-4
Torres-Morales, B., M. Rocandio-Rodríguez, A. Santacruz-Varela, L. Córdova-Téllez, B. Coutiño-Estrada, and H. López-Sánchez. 2022. Diversidad morfológica y agronómica de siete razas de maíz del estado de Chiapas. Revista Mexicana de Ciencias Agrícolas 13(4): 687–699. https://doi.org/10.29312/remexca.v13i4.2956
Vega-Alvarez, I., A. Santacruz-Varela, M. Rocandio-Rodríguez, L. Córdova-Téllez, H. López-Sánchez, A. Muñoz-Orozco, and A. Hernández-Bautista. 2017. Genetic diversity and structure of native maize races from Northwestern Mexico. Pesquisa Agropecuária Brasileira 52: 1023–1032. https://doi.org/10.1590/S0100-204X2017001100008
Vigouroux, Y., S. Mitchell, Y. Matsuoka, M. Hamblin, S. Kresovich, J. Stephen, C. Smith, J. Jaqueth, O. S. Smith, and J. Doebley. 2005. An analysis of genetic diversity across the maize genome using microsatellites. Genetics 169(3): 1617–1630. https://doi.org/10.1534/genetics.104.032086
Vigouroux, Y., J. C. Glaubitz, Y. Matsuoka, M. M. Goodman, J. Sánchez, and J. Doebley. 2008. Population structure and genetic diversity of New World maize races assessed by DNA microsatellites. American Journal of Botany 95(10): 1240–1253. https://doi.org/10.3732/ajb.0800097
Weir, B. S., and C. C. Cockerham. 1984. Estimating F-statistics for the analysis of population structure. Evolution 38(6): 1358–1370. https://doi.org/10.2307/2408641
Yeh, F. C., R. C. Yang, and T. Boyle. 1999. POPGENE Version 1.32: Microsoft Window-based freeware for population genetics analysis. Edmonton, Canada: University of Alberta
Zar, J. H. 1999. Biostatistical analysis. Hoboken, NJ: Prentice Hall
Acknowledgements
We deeply appreciate the permission, trust, and support of all the people and local authorities of the municipalities of San Agustin Loxicha and Candelaria Loxicha, to authorize and to facilitate the field work. To the Zapotec people who share their ancestral knowledge about maize diversity. Beatriz Rendón Aguilar, Fabiola Ramírez Corona, Juan Núñez Farfán, Martha Rocha Munive, and Fabián Vargas Mendoza review the preliminary version of the manuscript. Gabriela Contreras, Lourdes Luna, Mireya Hernández, Luis Bernal, Francisco Ávila, Verónica Aguilar, Alejandro López, David Bravo, Eric Vides, Guadalupe Carrillo, they help during field work and laboratory. Without their help, it would be difficult to get interviews and maize samples in a relatively short time. Gilberto Hernández Cárdenas and Miguel Bravo Rivera elaborated the map. To the Laboratory of Molecular Biology at Universidad Autónoma Metropolitana Iztapalapa for the support in the analysis of molecular samples. We deeply thank Alejandra Serrato-Díaz for all logistic support, advice, and guidance to the first author. Dr. Blair Orr volunteered with English language editing of the final draft.
Funding
This research was made possible through financial support from the National Council of Science and Technology throughout the master’s in biology program at the Universidad Autónoma Metropolitana Iztapalapa with a scholarship to the first author and personal resources of the authors.
Author information
Authors and Affiliations
Author notes
Alejandra Serrato-Díaz is deceased. This paper is dedicated to her memory.
- Alejandra Serrato-Díaz
Contributions
MCAM and BRA designed the project, applied the interviews, and sampled the maize blades. MCAM analyzed data and wrote the first version through her master’s thesis. MGRM gave feedback on molecular analysis in the laboratory and statistical analysis. FRC and CFVM helped with statistical analysis. All the authors reviewed and edited the final version of the manuscript.
Corresponding author
Ethics declarations
Competing Interests
The authors declare no competing interests.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Aragón-Martínez, M., Serrato-Díaz, A., Rocha-Munive, M.G. et al. Traditional Management of Maize in the Sierra Sur, Oaxaca, Maintains Moderate Levels of Genetic Diversity and Low Population Differentiation Among Landraces. Econ Bot 77, 282–304 (2023). https://doi.org/10.1007/s12231-023-09580-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12231-023-09580-z