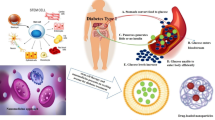
Abstract
Diabetic and chemotherapy-induced peripheral neuropathies are known for long-term complications that are associated with uncontrolled hyperglycemia and cancer treatment, respectively. Peripheral neuropathy often requires long-term therapy and could persist after treatment provoking detrimental effects on the patient’s quality of life. Despite continuous drug discoveries, development of efficient therapies is still needed for the significant management of diabetic and chemotherapy-induced peripheral neuropathy. Exosomes are nanosized extracellular vesicles that show great promise recently in tissue regeneration and injury repair compared to their parent stem cells. Herein, we provided a summary for the use of mesenchymal stem cell–derived exosomes in diabetic and chemotherapy-induced peripheral neuropathy in addition to recent advancements and ways proposed for the enhancement of their efficacy in these diseases.
Graphical abstract

Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Understanding the painful neuropathic disorders and mechanisms of current therapies is crucial for the recommendation of effective management strategies [1]. Peripheral neuropathy (PN) is associated with several diseases including diabetes, infections, autoimmunity, malignancy, and several metabolic diseases [2]. The most common causes of PN are diabetes and treatment with chemotherapy [3] with prevalence exceeding 50% in people with diabetes [4] and approximately 20 to 85% in patients treated with chemotherapy [5]. The exact mechanisms and pathogenesis of diabetic peripheral neuropathy (DPN) and chemotherapy-induced peripheral neuropathy (CIPN) remain unclear, although it has been suggested that both share mitochondrial dysfunction as a common pathogenic mechanism [6].
Diabetic sensorimotor polyneuropathy (DSPN) is one of the known type of diabetic neuropathy which demonstrates significant morbidity, affecting the quality of the patient’s life and increasing healthcare costs [7]. The progress of DSPN could be related mainly to defect in the antioxidant defense mechanisms. Other underlying mechanisms include changes in blood flow supplying the peripheral nerves, autoimmune and metabolic disorders leading to glial cell activation, changes in ion channel expression including sodium and calcium channels, and increased thalamic vascularity in addition to imbalance of stimulatory/inhibitory descending pathways with the involvement of central pain mechanisms [7, 8]. Other risk factors that contribute to DPN include old age, prolonged duration of uncontrolled diabetes, smoking, and drinking alcohol [9].
The clinical manifestations of DPN are comparable to those of CIPN. CIPN is a prominent complication associated with the long-term toxicity of chemotherapies including platinum-based drugs (cisplatin, carboplatin, and oxaliplatin), taxanes (paclitaxel and docetaxel), vincristine, and eribulin [10]. CIPN may persist for several years even after the cessation of chemotherapy causing disability in cancer survivors and seriously affecting their quality of life [11,12,13]. CIPN demonstrates sensory symptoms including pain, numbness, and tingling. However, some patients may have difficulties in fine motor coordination and sensory ataxia in addition to autonomic dysfunction [14]. The pathophysiological processes of CIPN are multi-factorial and have not been fully explained but differ among various classes of chemotherapeutic agents [15]. These processes involve oxidative stress, mitochondrial damage, apoptosis, altered ion channel activity, microtubular damage, axon degeneration and demyelination, and neuroinflammation [15, 16].
Several treatments have been proposed by international guidelines for controlling only symptoms of PN irrespective of the underlying cause. First-line therapies mostly include serotonin–noradrenaline reuptake inhibitors, tricyclic antidepressants, and anticonvulsants which act on sodium and calcium channels. Other treatments include the use of opioids in addition to topical agents such as lidocaine and capsaicin [9]. All traditional therapies reveal partial symptomatic relief of DPN and CIPN which reverses upon discontinuation in addition to having dose-limiting potential side effects [16]. These side effects often hinder the therapeutic options in clinical practice making the current treatments for DPN and CIPN suboptimal with urgent need for efficacious therapeutic strategies.
Mesenchymal stem cells (MSCs) are multipotent non-hematopoietic cells which show multiple advantages over other types of stem cells. MSCs can be readily isolated from different sources like adipose tissue, peripheral blood, and cord blood with minimally invasive approaches [17] and is known to be self-replicated to many passages [18]. MSCs have been widely considered a promising cell therapy for management of various neurological disorders [19, 20] with evidence of their efficacy in DPN [21,22,23] and CIPN [24, 25]. MSCs promote PN repair primarily via their paracrine effects with the secretion of angiogenic, anti-inflammatory, and neurotrophic factors [26,27,28]. However, MSC transplantation is compromised by prolonged induction period as well as possible risks of immunogenicity, tumor formation, and microcirculatory obstruction [29,30,31].
Exosomes are endosomal-origin membranous nanovesicles with a diameter ranging from approximately 50 nm to 100 nm [32]. Exosomes are released mostly by all cell types and play a pivotal role in communication between cells by acting as biological transporters. Exosomes contain functional mRNAs, microRNAs (miRNAs), proteins, and lipids [33]. Current studies have shown that MSC-derived exosomes (MSC-exos) are with similar functions to their parent cells. However, unlike MSCs, exosomes possess lower immunogenicity because they possess fewer membrane-bound proteins [34] and have a reduced risk of microvasculature occlusion [35]. Exosomes can also cross biological barriers including the blood-brain barrier because of their nanoscale structures and their membrane composition [36, 37]. Moreover, compared to their parent cells, exosomes can be stored easier and without the use of potentially toxic cryopreservatives for long term, reducing their toxic side effects upon application [38].
The therapeutic effect of MSC-exos has been demonstrated in a variety of diseases, including tumors [39], neurodegenerative diseases [40], and cardiovascular [41] and cerebrovascular [42] disorders. Importantly, several studies demonstrated the therapeutic effect of MSC-exos in DPN. However, the impact of MSC-exos in CIPN is still limited, and it is important to highlight on this crucial type of PN to encourage more research to be done in this field. Future researches are also required to fully elucidate in detail the mechanism of actions of MSC-exos in different models of PN. Of note, a series of challenges and difficulties need to be overcome to verify and enhance the therapeutic efficacy of the derived exosomes and to promote their clinical application.
Role of MSCs in PN
To develop more effective therapies to neuropathic pain, the potential of stem cells has been assessed in various stages of sensory neuropathy [25, 43] where stem cell therapy demonstrates beneficial outcomes experimentally in sensory neuropathy associated with diabetes [23], spinal cord injury (SCI) [44, 45], and ligation of sciatic nerve [46]. These outcomes depend mainly on the neuroregenerative and neuroprotective potentials of stem cells as demonstrated previously [47,48,49,50]. More importantly, the analgesic actions of MSCs have been revealed both experimentally [22, 51] and in clinical trials [52, 53], highlighting the efficacy of this therapy in the management of neuropathic pain. In this context, MSCs have been the most investigated type due to their wide therapeutic potential, minor risk of tumorigenesis, and ease of isolation and expansion in vitro [54]. However, the mechanisms involved in the therapeutic effects of MSCs have not yet been fully verified, but most probably, multiple mechanisms are included, clarifying the broad biological properties of these cells [22, 23, 46, 51].
MSCs promote the repair and modulate the injured neuronal environment through secretion of trophic, anti-inflammatory, and antiapoptotic factors which support angiogenesis, immunomodulation, remyelination, and axonal growth in addition to providing protection from high apoptotic cell death [28]. Through the support of angiogenesis, MSCs augment the microcirculation supplying the peripheral nerves where the impairment of vascular supply has been significantly revealed in DN. MSCs also could be differentiated into endothelial cells and neurons in addition to the secretion of important factors, like angiogenin, basic fibroblast growth factor, vascular endothelial growth factor-A , and nerve growth factor [55, 56], which are crucial to both vascular and neuronal tissue health. Obviously, adipose-derived MSCs secrete increased levels of various angiogenic growth factors both directly and indirectly accelerating the healing of diabetic wounds in rats [57]. MSCs also provide a potential therapy for CIPN through suppressing the cascades of neuronal oxidative stress, inflammation, and apoptosis and promoting axonal repair and regeneration [16].
Role of Exosomes in PN
A number of in vivo and in vitro studies have shown the ability of exosomes to regulate immune responses, promote angiogenesis, and regenerate damaged tissue [58]. Exosomes are endosomal membranous nanovesicles which represent the main and vital components of small extracellular vesicles (< 100 nm) [59,60,61]. Exosomes could promote the communication between cells by transferring cargo genomic materials including miRNAs as well as proteins and lipids [62, 63]. Exosomes could be used for the management of DPN where the administration of exosomes intravenously from healthy Schwann cells remarkably improved PN in a model of diabetes in mice [64]. However, there is a limitation of studies using exosomes in CIPN. Obviously, exosome cargo shows the same properties as their parent cells [65, 66] where exosomes acquired from mesenchymal stromal cells contain peptides and miRNAs that promote neuronal repair and function [63]. MSC-exos have been found to be internalized by distal axons of cortical neurons with the mediation of axonal growth even under inhibitory conditions [67]. Moreover, exosomes derived from cerebral endothelial cells (CEC-exos) when used in combination with a platinum drug significantly decreased CIPN and increased the anti-tumor effect of oxaliplatin in mice with ovarian cancer. After being internalized into neuronal fibers, the intravenously administered CEC-exos modified miRNA and protein networks of sciatic nerve and thus reduced the toxicity of peripheral nerves [68]. A previous study also revealed that the application of bone marrow mesenchymal stem cell-derived exosomes (BM-MSC-exos) reduced the expression of tumor necrosis factor α (TNF-α) and transforming growth factor β and increased the levels of interleukin (IL)-10 and arginase 1 in a mouse model of DPN, indicating a decrease in macrophages M1/M2 ratio. These outcomes could be mediated through the polarization of macrophages M2 with subsequent inhibition of the expression of pro-inflammatory genes and improvement of neurovascular function [69]. Additionally, exosomes carrying miR-181c-5p alleviated chronic constriction injury–induced neuropathic pain through inhibition of neuropathic inflammation [70].
Application of MSC-exos in DPN
Exosomes play a pivotal role in the communication between cells by transporting biological molecules where they reveal low immunogenicity and the ability to cross the blood-brain barrier. The therapeutic effect of MSC-exos has been demonstrated in preclinical studies of different nervous system diseases [33, 71, 72] with significant impact in DPN (Fig. 1).
For example, the delivery of MSC-exos was shown to inhibit the inflammatory response and promote the neurovascular remodeling and functional recovery of DPN in diabetic mice [69]. MSC-exos also ameliorated DN by autophagy induction through the mammalian target of rapamycin (mTOR) signaling pathway in a rat model of streptozotocin-induced diabetes mellitus [73]. Moreover, exosomes derived from Schwann cells ameliorated PN in type 2 diabetic mice [64]. Furthermore, exosomes when used as biologic vehicles of miR-146a effectively enhanced the therapeutic action of MSCs in diabetic mice, providing a new therapeutic approach for the management of DPN [74]. Table 1 shows some studies on MSC-exos in the treatment of DPN.
Application of MSC-exos in CIPN
Therapeutic approaches for the management of CIPN are limited and have been based on evidences provided from other types of neuropathies including diabetes and post-herpetic infections [77, 78]. No currently available drugs are considered fully effective in the clinical management of painful neuropathy associated with chemotherapy. However, the choice depends mostly on the use of drugs that reduce neural excitability, such as gabapentin, which still has low efficacy in reducing pain [79, 80].
Several mechanisms have been proposed to mediate the beneficial actions of MSC-exos including the secretion of angiogenic and neurotrophic factors, enhanced production of immunosuppressive factors including IL-10, and healthy mitochondrial transfer from MSCs to the damaged neurons [81,82,83]. Notably, several reports have demonstrated that exosomes derived from BM-MSCs contain a reparative cargo with various cytokines, neurotrophic factors, and miRNA, which mediate significant anti-inflammatory and anti-apoptotic actions with well-recognized immunomodulatory properties (Fig. 2). BM-MSCs and BM-MSC-exos can modulate microglia and astrocyte reactivity, thereby promoting neuro-regeneration [84, 85]. However, assessment of the detailed molecular and cellular mechanisms that affect neural microenvironment after these therapies still requires more studies.
Experimentally, intrathecal administration of MSC-exos reduced mechanical and thermal hypersensitivity in L5/6 spinal nerve ligation neuropathic pain rat model. The anti-inflammatory actions of these exosomes have been demonstrated through diminishing c-fos, 2′,3′-cyclic nucleotide 3′-phosphodiesterase (CNPase), glial fibrillary acidic protein, ionized calcium–binding adaptor molecule 1 (Iba1), TNF-α, and IL-1β levels in addition to enhancing IL10, brain-derived neurotrophic factor (BDNF), and glial cell line–derived neurotrophic factor levels [86]. CEC-exos also reduced CIPN and enhanced the anti-tumor efficacy of platinum drugs [87]. Importantly, a recent study demonstrated that cannabidiol incorporated in extracellular vesicles derived from human umbilical cord MSCs had the ability to alleviate mechanical and thermal pain sensitivities in paclitaxel-induced PN. These outcomes were associated with mitochondrial protection in neuronal cells via activating 5HT1A receptors and AMP-activated protein kinase (AMPK) pathway [88]. In another study, BM-MSC-exos loaded with reduced graphene oxide (rGO), gelatin-methacryloyl (GelMA), and polycaprolactone (PCL) to create an rGO-GelMA-PCL nerve conduit revealed a superior ability to alleviate sciatic nerve injury via increasing the number of newly formed vessels as well as promoting peripheral nerve regeneration and function recovery [89]. Thus, despite being limited, the results of published studies give the hope for the possible significant impact of MSC-exos application in CIPN.
Enhancement of the Therapeutic Effects of MSC-exos in Different Models of Neuropathy
MSC-exos exert clear therapeutic effects in various diseases. Obviously, exosome properties vary depending on the status of MSCs from which they are derived where MSC status is modified in response to external stimuli. Therefore, several studies have investigated whether preconditioning MSCs could enhance the therapeutic activities of their derived exosomes. Preconditioning of MSCs with cytokines, hypoxia, and chemicals improve their immunosuppressive, immunomodulatory, and regenerative effects [90]. Moreover, genetic and cell surface modification can enhance the therapeutic efficacy of exosomes [91].
Different Preconditioning Strategies
Preconditioning of MSCs with Hypoxia
The administration of MSC-exos subjected to hypoxia preconditioning could enhance the neurologic functions by specific biomolecule transport to the recipient damaged cells [41]. For example, exosomes derived from the exposure of MSCs to hypoxia preconditioning restored the synaptic dysfunction and reduced the inflammatory responses through the regulation of miR-21 secretion and thus improvement of the learning and memory capabilities of APP/PS1 mice [41]. Hypoxia preconditioning represents one of the most effective approaches to enhance the therapeutic effects of MSC-exos. A possible underlying mechanism could be through enhanced exosomal miR-216a-5p secretion which directs the shifting of microglia from the pro-inflammatory M1 phenotype to the anti-inflammatory M2 phenotype and thus suppressing toll-like receptors (TLRs)/nuclear factor kappa B (NF-κB) and activating phosphatidylinositol 3-kinase (PI3K)/protein kinase B (PKB) signaling pathways [92]. Another study indicated that human umbilical cord mesenchymal stem cell (HUCMSC)-derived exosomes could improve the viability and migration of olfactory ensheathing cells in hypoxic conditions by activating BDNF signaling and thus promoting sciatic nerve regeneration and functional recovery [93]. Additionally, exosomes derived from hypoxia-preconditioned MSCs improved cognitive decline through amelioration of synaptic dysfunction and regulation of inflammatory responses in APP/PS1 mice [41]. Hypoxia-treated MSCs were also found to overexpress antiapoptotic proteins like IL-8, IL-10, and Fas Ligand (FasL) and thus enhancing immunoregulation which in turn would inhibit inflammation and encourage tissue repair and regeneration [94].
Preconditioning of MSCs with Pharmacological Agents
Preconditioning of MSCs with pharmacological agents is considered another effective way to enhance their actions [95, 96]. The preconditioning of MSCs has been examined through the use of pharmacological agents known for their neuroprotective and neurotrophic actions such as mood stabilizers like lithium and valproic acid [97]. Importantly, lithium and valproic acid control the signaling pathway mediating neurotrophic and neuroprotective functions through the inhibition of histone deacetylase (HDAC). In turn, the inhibition of HDAC prevents apoptosis and suppresses cellular glycogen synthase kinase-3 which mediates glycogen metabolism, cell proliferation and migration, and cellular transport [98]. Experimentally, deferoxamine preconditioning significantly increased total antioxidant, pro-angiogenic, neuroprotective, and anti-inflammatory factors secreted by MSCs and thus improving the therapeutic potential of adipose tissue-MSCs in DN [99]. The enhancement of exosome efficacy using different culture conditions in different nervous system diseases gives the hope of their possible efficacy in PN.
Modified MSC-exos
Previous studies revealed different ways of MSC-exos modification to improve their therapeutic potential [100] through loading exosomal lumen with endogenous or exogenous biomolecules such as nucleic acids, peptides, or drugs or through modifying MSC-exos’ surface for targeting of a particular type of cells or tissues [101].
Therapeutic Exosome Loading
MSC-exos have recently been introduced as promising biological carriers owing to their remarkable small size, biocompatibility, and ability to carry specific and various therapeutic molecules to their targeted cells [102]. The functional biomolecules in MSC-exos such as proteins and nucleotides are included in tissue regeneration, immunoregulation, and angiogenesis in various experimental models [103]. Exosome loading includes the addition of the therapeutic molecule to the exosome from its parent cell (endogenous loading) or direct depositing of the therapeutic molecule into purified exosomes (exogenous loading) [104].
As an example of endogenous loading, MSCs can be genetically modified to enhance the expression of endogenous biomolecules inside MSC-exos by using plasmids or viral vectors. Therefore, loading the targeted therapeutic biomolecules inside exosomes depends mainly on the genetic modifications of the parent cells for the improvement of the therapeutic actions of MSC-exos and prevention of heterogeneity between different exosome batches [103]. Exosomal miRNAs are also key regulators to their function. For example, treatment of DPN with MSC-exos enriched with miRNA-146a provided enhanced therapeutic efficacy in PN [74]. The administration of exosomes containing miR-133b also contributed to the inhibition of ras homolog gene family member A (RhoA) and activation of extracellular signal-regulating kinase 1/2 (ERK1/2) and cAMP response element-binding protein (CREB) pathway and thus amelioration of neuronal apoptosis and neurodegeneration in rats after intracerebral hemorrhage [105]. Besides, miR-133b-modified MSC-exos allowed miR-133b transfer to astrocytes and thus regulated their expression and promoted neuronal functional recovery after stroke in rats [106]. In addition, MSC-exos enriched with miR-126 and miR-26a enhanced the recovery of limb motor function and improved axon regeneration after SCI [107, 108]. Transplantation of engineered exosomes derived from BM-MSCs after being fused with polypyrrole nanoparticles containing liposomes improved DPN along with electrical stimulation. This approach normalized the velocity of nerve conduction and muscle action potential compared to the healthy control [75]. Notably, several research studies have revealed that MSC-exos could be used as drug carriers in addition to their traditional cargoes like miRNAs and peptides. The membrane structure of MSC-exos can preserve the biological activity and integrity of drugs in addition to possible achievement of targeted drug delivery through the modification of the membrane structure of MSC-exos [109]. For example, chemotherapeutic drugs (adriamycin and paclitaxel) were loaded into MSC-exos for the management of tumors with reduced drug side effects [110, 111]. In the future, MSC-exos can be used as carriers for potential drugs for the management of diabetes, cancer, and their related neuropathic complications.
Surface Modification of Exosomes
Three common strategies are present for surface membrane modification of MSC-exos such as genetic engineering, chemical modification, and membrane fusion. Genetic engineering consolidates gene sequences of guided proteins or peptides with those of selected exosomal membrane proteins to effectively present specific peptides and proteins on exosomal surface and thus enhancing its targeting or function. On the other hand, chemical modifications reveal a variety of synthetic and natural receptors or ligands on the membrane surface of exosomes through noncovalent or covalent modifications. Finally, membrane fusion uses extrusion to combine exosomes with other membrane structures, an approach that confers new functional and therapeutic benefits to them [112]. For example, Yang et al. innovated a membrane-editing technique to confer the required functional membrane proteins into cellular membranes directly using a virus-mimetic fusogenic exosomes, enabling the exosomes to target specific tissues [113]. Another study used the fusion extrusion technique to modify MSC-exos with monocyte mimics to enhance their delivery to myocardial tissues with ischemic damage [114]. Collectively, the membrane fusion technique to modify MSC-exos opens new approaches for improvement of their cell-targeting therapeutic efficacy.
Limitations of Current Studies and Future Directions
MSC-exos have been clearly recognized as the main regulators of the paracrine mechanism that promotes regeneration mediated by stem cells [115]. These nanoparticles reveal similar therapeutic effects to those exerted by MSCs and can be considered a powerful tool for cell-free–based therapy with several advantages compared to MSCs. Of note, the outcomes of use of MSC-exos in peripheral nerve injury are encouraging [114]. Although being safe and effective experimentally, this field needs further research and much work to fully determine the potential of MSC-exos clinically and to overcome the limitations of their application [116, 117].
MSC-exos are commonly isolated via differential or density-gradient ultracentrifugation [118]. Despite being the most cost-effective available method, ultracentrifugation is time-consuming, needs intensive work, and may yield impurities [119]. Other isolation methods include ultrafiltration, size exclusion chromatography, precipitation, and immune affinity capture [119] in addition to size-dependent and immunoaffinity-based microfluidic technologies [120]. Though being limited compared to the traditional methods, these new methods require relatively small amount of samples and can achieve high purity and rapid separation [119]. Limitations of MSC-exos isolation include also low yield under conventional culture conditions [121] in addition to low efficacy upon application after multiple passages in vitro due to MSC senescence [122]. Thus, improving the yield of MSC-exos and enhancing their efficacy or targeting ability are crucial through altering MSC cell culture conditions [123, 124] or modifying exosomes with different approaches [125]. Furthermore, concerns regarding changes in morphology and bioactivity of exosomes have been reported following long-term storage [126]. Cryopreservation using liquid nitrogen and other cryoprotective agents may overcome these issues [127]. Notably, the components of MSC-exos as well as their exact actions that mediate tissue repair and regeneration need to be fully investigated. There is also no guidance for the safety and the exact dose or amount of MSC-exos needed for treatment. More research is still required to select the best route of systemic administration of MSC-exos (intravenous, subcutaneous, or intramuscular routes) [128]. Collectively, the risks, safety, and challenges included in the use of MSC-exos need to be fully investigated before their clinical use in tissue repair and regenerative medicine.
Conclusion
MSC-exos have received great attention recently as an emerging therapeutic agent for the management of PN because of their ability to suppress inflammation, regulate the immune system and angiogenesis, and promote tissue regeneration in a similar way to MSCs. Exosomes reveal several advantages, fewer side effects, and lower risks compared to their parent MSCs. Exosomes are relatively non-immunogenic and with very low tumorigenic potential. However, a series of challenges and difficulties need to be overcome to verify their purity, reproducibility, safety, biodistribution, clearance, and their molecular characteristics or contents. Furthermore, their long-term therapeutic effect is still unknown.
Importantly, future research is required to enhance the therapeutic efficacy of exosomes and to promote their clinical application where preconditioning of MSCs with hypoxia or pharmacological agents has been considered an effective approach to optimize the actions of their derived exosomes. The surface of exosomes can also be modulated for targeting of particular cell types. Moreover, exosomes can be loaded with targeted drugs, peptides, or genes with facilitated delivery due to their ability to cross biological barriers. Meanwhile, the signals and pathways involved in the actions of MSC-exos in the target cells and organs should be identified in detailed studies before being considered as a potential cell-free based therapy in PN.
Data Availability
Not applicable
Abbreviations
- AMPK:
-
AMP-activated protein kinase
- BM-MSCs:
-
Bone marrow mesenchymal stem cells
- CEC-exos:
-
Exosomes derived from cerebral endothelial cells
- CIPN:
-
Chemotherapy-induced peripheral neuropathy
- CNPase:
-
2′,3′-cyclic nucleotide 3′-phosphodiesterase
- CREB:
-
cAMP response element-binding protein
- DPN:
-
Diabetic peripheral neuropathy
- DSPN:
-
Diabetic sensorimotor polyneuropathy
- ERK1/2:
-
Extracellular signal regulating kinase 1/2
- GelMA:
-
Gelatin-methacryloyl
- HDAC:
-
Histone deacetylase
- HUCMSC:
-
Human umbilical cord mesenchymal stem cell
- Iba1:
-
Ionized calcium binding adaptor molecule 1
- IL:
-
Interleukin
- miRNAs:
-
MicroRNAs
- MSC-exos:
-
MSC-derived exosomes
- MSCs:
-
Mesenchymal stem cells
- mTOR:
-
Mammalian target of rapamycin
- NGCs:
-
Nerve guidance channels
- NF-κB:
-
Nuclear factor kappa B
- PN:
-
Peripheral neuropathy
- PCL:
-
Polycaprolactone
- PI3K:
-
Phosphatidylinositol 3-kinase
- PKB:
-
Protein kinase B
- rGO:
-
Reduced graphene oxide
- RhoA:
-
Ras homolog gene family member A
- SCI:
-
Spinal cord injury
- TLRs:
-
Toll-like receptors
References
Gore M, Dukes E, Rowbotham DJ, Tai KS, Leslie D (2007) Clinical characteristics and pain management among patients with painful peripheral neuropathic disorders in general practice settings. Eur J Pain 11:652–664. https://doi.org/10.1016/j.ejpain.2006.10.004
Roth B, Schiro DB, Ohlsson B (2021) Diseases which cause generalized peripheral neuropathy: a systematic review. Scand J Gastroenterol 56:1000–1010. https://doi.org/10.1080/00365521.2021.1942542
Wu CJ, Huang KJ, Tsai YC, Yeh TP, Hsieh CF, Wang YJ (2021) Peripheral neuropathy: comparison of symptoms and severity between colorectal cancer survivors and patients with diabetes. Clin J Oncol Nurs 1(25):395–403. https://doi.org/10.1188/21.CJON.395-403
Pop-Busui R, Ang L, Boulton AJM, Feldman EL, Marcus RL, Mizokami-Stout K et al (2022) Diagnosis and treatment of painful diabetic peripheral neuropathy. Compendia 2022:1–32. https://doi.org/10.2337/db2022-01
Michalová Z, Székiová E, Blaško J, Vanický I (2023) Prevention and therapy of chemotherapy-induced peripheral neuropathy: a review of recent findings. Neoplasma 70:15–35. https://doi.org/10.4149/neo_2022_221007N992
Flatters SJL, Bennett GJ (2006) Studies of peripheral sensory nerves in paclitaxel-induced painful peripheral neuropathy: evidence for mitochondrial dysfunction. Pain 122:245–257. https://doi.org/10.1016/j.pain.2006.01.037
Callaghan BC, Cheng HT, Stables CL, Smith AL, Feldman EL (2012) Diabetic neuropathy: clinical manifestations and current treatments. Lancet Neurol 11:521–534. https://doi.org/10.1016/S1474-4422(12)70065-0
Javed S, Petropoulos IN, Alam U, Malik RA (2015) Treatment of painful diabetic neuropathy. Ther Adv Chronic Dis 6:15–28. https://doi.org/10.1177/2040622314552071
Khdour MR (2020) Treatment of diabetic peripheral neuropathy: a review. J Pharm Pharmacol 72:863–872. https://doi.org/10.1111/jphp.13241
Wolf S, Barton D, Kottschade L, Grothey A, Loprinzi C (2008) Chemotherapy-induced peripheral neuropathy: prevention and treatment strategies. Eur J Cancer 44:1507–1515. https://doi.org/10.1016/j.ejca.2008.04.018
Ewertz M, Qvortrup C, Eckhoff L (2015) Chemotherapy-induced peripheral neuropathy in patients treated with taxanes and platinum derivatives. Acta Oncol 54:587–591. https://doi.org/10.3109/0284186X.2014.995775
Pachman DR, Qin R, Seisler DK et al (2015) Clinical course of oxaliplatin-induced neuropathy: results from the randomized phase III trial N08CB (Alliance). J Clin Oncol 33:3416–3422. https://doi.org/10.1200/JCO.2014.58.8533
Boyette-Davis JA, Walters ET, Dougherty PM (2015) Mechanisms involved in the development of chemotherapy-induced neuropathy. Pain Manag 5:285–296. https://doi.org/10.2217/pmt.15.19
Miltenburg NC, Boogerd W (2014) Chemotherapy-induced neuropathy: a comprehensive survey. Cancer Treat Rev 40:872–882. https://doi.org/10.1016/j.ctrv.2014.04.004
Argyriou AA, Bruna J, Marmiroli P, Cavaletti G (2012) Chemotherapy-induced peripheral neurotoxicity (CIPN): an update. Crit Rev Oncol Hematol 82:51–77. https://doi.org/10.1016/j.critrevonc.2011.04.012
Al-Massri KF, Ahmed LA, El-Abhar HS (2020) Mesenchymal stem cells in chemotherapy-induced peripheral neuropathy: a new challenging approach that requires further investigations. J Tissue Eng Regen Med 14:108–122. https://doi.org/10.1002/term.2972
Ullah I, Subbarao RB, Rho GJ (2015) Human mesenchymal stem cells - current trends and future prospective. Biosci Rep 35:e00191. https://doi.org/10.1042/BSR20150025
Marion NW, Mao JJ (2006) Mesenchymal stem cells and tissue engineering. Methods Enzymol. 420:339–361. https://doi.org/10.1016/S0076-6879(06)20016-8
Katakowski M, Buller B, Zheng X et al (2013) Exosomes from marrow stromal cells expressing miR-146b inhibit glioma growth. Cancer Lett 335:201–204. https://doi.org/10.1016/j.canlet.2013.02.019
Han JW, Choi D, Lee MY, Huh YH, Yoon YS (2016) Bone marrow-derived mesenchymal stem cells improve diabetic neuropathy by direct modulation of both angiogenesis and myelination in peripheral nerves. cell Transplant 25:313–326. https://doi.org/10.3727/096368915X688209
Hosseini M, Yousefifard M, Aziznejad H, Nasirinezhad F (2015) The effect of bone marrow-derived mesenchymal stem cell transplantation on allodynia and hyperalgesia in neuropathic animals: a systematic review with meta-analysis. Biol Blood Marrow Transplant 21:1537–1544. https://doi.org/10.1016/j.bbmt.2015.05.008
Monfrini M, Donzelli E, Rodriguez-Menendez V et al (2017) Therapeutic potential of mesenchymal stem cells for the treatment of diabetic peripheral neuropathy. Exp Neurol. 288:75–84. https://doi.org/10.1016/j.expneurol.2016.11.006
Evangelista AF, Vannier-Santos MA, de Assis Silva GS et al (2018) Bone marrow-derived mesenchymal stem/stromal cells reverse the sensorial diabetic neuropathy via modulation of spinal neuroinflammatory cascades. J Neuroinflammation 15:189. https://doi.org/10.1186/s12974-018-1224-3
Al-Massri KF, Ahmed LA, El-Abhar HS (2019) Mesenchymal stem cells therapy enhances the efficacy of pregabalin and prevents its motor impairment in paclitaxel-induced neuropathy in rats: role of Notch1 receptor and JAK/STAT signaling pathway. Behav Brain Res 360:303–311. https://doi.org/10.1016/j.bbr.2018.12.013
Dos Santos GGL, Oliveira ALL, Santos DS et al (2021) Mesenchymal stem cells reduce the oxaliplatin-induced sensory neuropathy through the reestablishment of redox homeostasis in the spinal cord. Life Sci 265:118755. https://doi.org/10.1016/j.lfs.2020.118755
Siniscalco D, Giordano C, Galderisi U et al (2010) Intra-brain microinjection of human mesenchymal stem cells decreases allodynia in neuropathic mice. Cell Mol Life Sci 67:655–669. https://doi.org/10.1007/s00018-009-0202-4
Waterman RS, Morgenweck J, Nossaman BD, Scandurro AE, Scandurro SA, Betancourt AM (2012) Anti-inflammatory mesenchymal stem cells (MSC2) attenuate symptoms of painful diabetic peripheral neuropathy. Stem Cells Transl Med 1:557–565. https://doi.org/10.5966/sctm.2012-0025
Shibata T, Naruse K, Kamiya H et al (2008) Transplantation of bone marrow-derived mesenchymal stem cells improves diabetic polyneuropathy in rats. Diabetes 57:3099–3107. https://doi.org/10.2337/db08-0031
Zurita M, Vaquero J, Oya S, Bonilla C, Aguayo C (2007) Neurotrophic Schwann-cell factors induce neural differentiation of bone marrow stromal cells. Neuroreport 18:1713–1717. https://doi.org/10.1097/WNR.0b013e3282f0d3b0
Jeong JO, Han JW, Kim JM et al (2011) Malignant tumor formation after transplantation of short-term cultured bone marrow mesenchymal stem cells in experimental myocardial infarction and diabetic neuropathy. Circ Res 108:1340–1347. https://doi.org/10.1161/CIRCRESAHA.110.239848
Shojaei S, Hashemi SM, Ghanbarian H, Salehi M, Mohammadi-Yeganeh S (2019) Effect of mesenchymal stem cells-derived exosomes on tumor microenvironment: tumor progression versus tumor suppression. J Cell Physiol 234(4):3394–3409. https://doi.org/10.1002/jcp.27326
Van Niel G, D'Angelo G, Raposo G (2018) Shedding light on the cell biology of extracellular vesicles. Nat Rev Mol Cell Biol 19:213–228. https://doi.org/10.1038/nrm.2017.125
Zhang ZG, Buller B, Chopp M (2019) Exosomes - beyond stem cells for restorative therapy in stroke and neurological injury. Nat Rev Neurol 15:193–203. https://doi.org/10.1038/s41582-018-0126-4
Konala VB, Mamidi MK, Bhonde R, Das AK, Pochampally R, Pal R (2016) The current landscape of the mesenchymal stromal cell secretome: a new paradigm for cell-free regeneration. Cytotherapy 18:13–24. https://doi.org/10.1016/j.jcyt.2015.10.008
Zhao T, Sun F, Liu J, Ding T, She J, Mao F, Xu W, Qian H, Yan Y (2019) Emerging role of mesenchymal stem cell-derived exosomes in regenerative medicine. Curr Stem Cell Res Ther 14:482–494. https://doi.org/10.2174/1574888X14666190228103230
Lopez-Verrilli MA, Picou F, Court FA (2013) Schwann cell-derived exosomes enhance axonal regeneration in the peripheral nervous system. Glia 61:1795–1806. https://doi.org/10.1002/glia.22558
Qing L, Chen H, Tang J, Jia X (2018) Exosomes and their microRNA cargo: new players in peripheral nerve regeneration. Neurorehabil Neural Repair 32:765–776. https://doi.org/10.1177/1545968318798955
Webber J, Clayton A (2013) How pure are your vesicles? J Extracell Vesicles 10:2. https://doi.org/10.3402/jev.v2i0.19861
Luo T, von der Ohe J, Hass R (2021) MSC-derived extracellular vesicles in tumors and therapy. Cancers (Basel) 13:5212. https://doi.org/10.3390/cancers13205212
Riazifar M, Mohammadi MR, Pone EJ et al (2019) Stem cell-derived exosomes as nanotherapeutics for autoimmune and neurodegenerative disorders. ACS Nano 13:6670–6688. https://doi.org/10.1021/acsnano.9b01004
Cui GH, Wu J, Mou FF, Xie WH, Wang FB, Wang QL, Fang J, Xu YW, Dong YR, Liu JR, Guo HD (2018) Exosomes derived from hypoxia-preconditioned mesenchymal stromal cells ameliorate cognitive decline by rescuing synaptic dysfunction and regulating inflammatory responses in APP/PS1 mice. FASEB J 32:654–668. https://doi.org/10.1096/fj.201700600R
Moon GJ, Sung JH, Kim DH et al (2019) Application of mesenchymal stem cell-derived extracellular vesicles for stroke: biodistribution and microRNA study. Transl Stroke Res 10:509–521. https://doi.org/10.1007/s12975-018-0668-1
Kim BJ, Jin HK, Bae JS (2011) Bone marrow-derived mesenchymal stem cells improve the functioning of neurotrophic factors in a mouse model of diabetic neuropathy. Lab Anim Res 27:171–176. https://doi.org/10.5625/lar.2011.27.2.171
Gazdic M, Volarevic V, Harrell CR et al (2018) Stem cells therapy for spinal cord injury. Int J Mol Sci 19:1039. https://doi.org/10.3390/ijms19041039
Allahdadi KJ, de Santana TA, Santos GC et al (2019) IGF-1 overexpression improves mesenchymal stem cell survival and promotes neurological recovery after spinal cord injury. Stem Cell Res Ther 10:146. https://doi.org/10.1186/s13287-019-1223-z
Gama KB, Santos DS, Evangelista AF et al (2018) Conditioned medium of bone marrow-derived mesenchymal stromal cells as a therapeutic approach to neuropathic pain: a preclinical evaluation. Stem Cells Int 2018:8179013. https://doi.org/10.1155/2018/8179013
Ul Hassan A, Hassan G, Rasool Z (2009) Role of stem cells in treatment of neurological disorder. Int J Health Sci 3:227–233
Venkatesh K, Sen D (2017) Mesenchymal stem cells as a source of dopaminergic neurons: a potential cell based therapy for Parkinson’s disease. Curr Stem Cell Res Ther 12:326–347. https://doi.org/10.2174/1574888X12666161114122059
Puertas-Neyra K, Usategui-Martín R, Coco RM, Fernandez-Bueno I (2020) Intravitreal stem cell paracrine properties as a potential neuroprotective therapy for retinal photoreceptor neurodegenerative diseases. Neural Regen Res 15:1631–1638. https://doi.org/10.4103/1673-5374.276324
Sadatpoor SO, Salehi Z, Rahban D, Salimi A (2020) Manipulated mesenchymal stem cells applications in neurodegenerative diseases. Int J Stem Cells 13:24–45. https://doi.org/10.15283/ijsc19031
Di Cesare ML, Tenci B, Micheli L et al (2018) Adipose-derived stem cells decrease pain in a rat model of oxaliplatin-induced neuropathy: role of VEGF-A modulation. Neuropharmacology 131:166–175. https://doi.org/10.1016/j.neuropharm.2017.12.020
Mendonça MV, Larocca TF, de Freitas Souza BS et al (2014) Safety and neurological assessments after autologous transplantation of bone marrow mesenchymal stem cells in subjects with chronic spinal cord injury. Stem Cell Res Ther 5:126. https://doi.org/10.1186/scrt516
Vaquero J, Zurita M, Rico MA et al (2018) Intrathecal administration of autologous mesenchymal stromal cells for spinal cord injury: safety and efficacy of the 100/3 guideline. Cytotherapy 20:806–819. https://doi.org/10.1016/j.jcyt.2018.03.032
Eleuteri S, Fierabracci A (2019) Insights into the secretome of mesenchymal stem cells and its potential applications. Int J Mol Sci 20:4597. https://doi.org/10.3390/ijms20184597
Quertainmont R, Cantinieaux D, Botman O, Sid S, Schoenen J, Franzen R (2012) Mesenchymal stem cell graft improves recovery after spinal cord injury in adult rats through neurotrophic and pro-angiogenic actions. PLoS One 7:e39500. https://doi.org/10.1371/journal.pone.0039500
Hsieh JY, Wang HW, Chang SJ et al (2013) Mesenchymal stem cells from human umbilical cord express preferentially secreted factors related to neuroprotection, neurogenesis, and angiogenesis. PLoS One 8:e72604. https://doi.org/10.1371/journal.pone.0072604
Kato Y, Iwata T, Morikawa S, Yamato M, Okano T, Uchigata Y (2015) Allogeneic transplantation of an adipose-derived stem cell sheet combined with artificial skin accelerates wound healing in a rat wound model of type 2 diabetes and obesity. Diabetes 64:2723–2734. https://doi.org/10.2337/db14-1133
Zhang B, Wang M, Gong A et al (2015) HucMSC-exosome mediated-Wnt4 signaling is required for cutaneous wound healing. Stem Cells 33:2158–2168. https://doi.org/10.1002/stem.1771
Meldolesi J (2018) Exosomes and ectosomes in intercellular communication. Curr Biol 28:R435–R444. https://doi.org/10.1016/j.cub.2018.01.059
Samanta S, Rajasingh S, Drosos N, Zhou Z, Dawn B, Rajasingh J (2018) Exosomes: new molecular targets of diseases. Acta Pharmacol Sin 39:501–513. https://doi.org/10.1038/aps.2017.162
Théry C, Zitvogel L, Amigorena S (2002) Exosomes: composition, biogenesis and function. Nat Rev Immunol 2:569–579. https://doi.org/10.1038/nri855
Marcus ME, Leonard JN (2013) FedExosomes: engineering therapeutic biological nanoparticles that truly deliver. Pharmaceuticals 6:659–680. https://doi.org/10.3390/ph6050659
Zhang Y, Chopp M, Liu XS et al (2017) Exosomes derived from mesenchymal stromal cells promote axonal growth of cortical neurons. Mol Neurobiol 54:2659–2673. https://doi.org/10.1007/s12035-016-9851-0
Wang L, Chopp M, Szalad A et al (2020) Exosomes derived from Schwann cells ameliorate peripheral neuropathy in type 2 diabetic mice. Diabetes 69:749–759. https://doi.org/10.2337/db19-0432
Mathieu M, Martin-Jaular L, Lavieu G, Théry C (2019) Specificities of secretion and uptake of exosomes and other extracellular vesicles for cell-to-cell communication. Nat Cell Biol 21:9–17. https://doi.org/10.1038/s41556-018-0250-9
Yáñez-Mó M, Siljander PR, Andreu Z et al (2015) Biological properties of extracellular vesicles and their physiological functions. J Extracell Vesicles 4:27066. https://doi.org/10.3402/jev.v4.27066
Zhao Y (2018) Nanomedicine in China. Adv Healthc Mater 7:e1801051. https://doi.org/10.1002/adhm.201801051
Zhang Y, Li C, Qin Y et al (2021) Small extracellular vesicles ameliorate peripheral neuropathy and enhance chemotherapy of oxaliplatin on ovarian cancer. J Extracell Vesicles 10:e12073. https://doi.org/10.1002/jev2.12073
Fan B, Li C, Szalad A et al (2020) Mesenchymal stromal cell-derived exosomes ameliorate peripheral neuropathy in a mouse model of diabetes. Diabetologia 63:431–443. https://doi.org/10.1007/s00125-019-05043-0
Zhang YU, Ye G, Zhao J et al (2022) Exosomes carried miR-181c-5p alleviates neuropathic pain in CCI rat models. An Acad Bras Cienc 94:e20210564. https://doi.org/10.1590/0001-3765202220210564
Alvarez-Erviti L, Seow Y, Yin H, Betts C, Lakhal S, Wood MJ (2011) Delivery of siRNA to the mouse brain by systemic injection of targeted exosomes. Nat Biotechnol 29:341–345. https://doi.org/10.1038/nbt.1807
Zhang Y, Zhang Y, Chopp M, Zhang ZG, Mahmood A, Xiong Y (2020) Mesenchymal stem cell-derived exosomes improve functional recovery in rats after traumatic brain injury: a dose-response and therapeutic window study. Neurorehabil Neural Repair. 34:616–626. https://doi.org/10.1177/1545968320926164
Ebrahim N, Ahmed IA, Hussien NI et al (2018) Mesenchymal stem cell-derived exosomes ameliorated diabetic nephropathy by autophagy induction through the mTOR signaling pathway. Cells 7:226. https://doi.org/10.3390/cells7120226
Fan B, Chopp M, Zhang ZG, Liu XS (2021) Treatment of diabetic peripheral neuropathy with engineered mesenchymal stromal cell-derived exosomes enriched with microRNA-146a provide amplified therapeutic efficacy. Exp Neurol 341:113694. https://doi.org/10.1016/j.expneurol.2021.113694
Singh A, Raghav A, Shiekh PA, Kumar A (2021) Transplantation of engineered exosomes derived from bone marrow mesenchymal stromal cells ameliorate diabetic peripheral neuropathy under electrical stimulation. Bioact Mater 6:2231–2249. https://doi.org/10.1016/j.bioactmat.2021.01.008
Singh A, Shiekh PA, Qayoom I, Srivastava E, Kumar A (2021) Evaluation of polymeric aligned NGCs and exosomes in nerve injury models in diabetic peripheral neuropathy condition. Eur Polym J 146:110256. https://doi.org/10.1016/j.eurpolymj.2020.110256
Arnold LM, McCarberg BH, Clair AG et al (2017) Dose-response of pregabalin for diabetic peripheral neuropathy, postherpetic neuralgia, and fibromyalgia. Postgrad Med 129:921–933. https://doi.org/10.1080/00325481.2017.1384691
Ghanavatian S, Wie CS, Low RS et al (2019) Premedication with gabapentin significantly reduces the risk of postherpetic neuralgia in patients with neuropathy. Mayo Clin Proc 94:484–489. https://doi.org/10.1016/j.mayocp.2018.11.004
Rao RD, Michalak JC, Sloan JA et al (2007) Efficacy of gabapentin in the management of chemotherapy-induced peripheral neuropathy: a phase 3 randomized, double-blind, placebo-controlled, crossover trial (N00C3). Cancer 110:2110–2118. https://doi.org/10.1002/cncr.23008
Moore J, Gaines C (2019) Gabapentin for chronic neuropathic pain in adults. Br J Community Nurs 24:608–609. https://doi.org/10.12968/bjcn.2019.24.12.608
Kyurkchiev D, Bochev I, Ivanova-Todorova E et al (2014) Secretion of immunoregulatory cytokines by mesenchymal stem cells. World J Stem Cells 6:552–570. https://doi.org/10.4252/wjsc.v6.i5.552
Polydoro M, Dzhala VI, Pooler AM et al (2014) Soluble pathological tau in the entorhinal cortex leads to presynaptic deficits in an early Alzheimer’s disease model. Acta Neuropathol 127:257–270. https://doi.org/10.1007/s00401-013-1215-5
Gebler A, Zabel O, Seliger B (2012) The immunomodulatory capacity of mesenchymal stem cells. Trends Mol Med 18:128–134. https://doi.org/10.1016/j.molmed.2011.10.004
Mukhamedshina YO, Gracheva OA, Mukhutdinova DM, Chelyshev YA, Rizvanov AA (2019) Mesenchymal stem cells and the neuronal microenvironment in the area of spinal cord injury. Neural Regen Res 14:227–237. https://doi.org/10.4103/1673-5374.244778
Li G, Yu F, Lei T et al (2016) Bone marrow mesenchymal stem cell therapy in ischemic stroke: mechanisms of action and treatment optimization strategies. Neural Regen Res 11:1015–1024. https://doi.org/10.4103/1673-5374.184506
Shiue SJ, Rau RH, Shiue HS et al (2019) Mesenchymal stem cell exosomes as a cell-free therapy for nerve injury-induced pain in rats. Pain. 160:210–223. https://doi.org/10.1097/j.pain.0000000000001395
Zhang Y, Zhang ZG, Chopp M, Li C (2019) Exosomes from cerebral endothelial cells suppress chemotherapyinduced peripheral neuropathy and sensitize anti-tumour effects of platinum drugs. J Extracell Vesicles 8:142
Kalvala AK, Bagde A, Arthur P, Kulkarni T, Bhattacharya S, Surapaneni S et al (2023) Cannabidiol-loaded extracellular vesicles from human umbilical cord mesenchymal stem cells alleviate paclitaxel-induced peripheral neuropathy. Pharmaceutics 15:554. https://doi.org/10.3390/pharmaceutics15020554
Zhang W, Fang XX, Li QC, Pi W, Han N (2023) Reduced graphene oxide-embedded nerve conduits loaded with bone marrow mesenchymal stem cell-derived extracellular vesicles promote peripheral nerve regeneration. Neural Regen Res 18:200–206. https://doi.org/10.4103/1673-5374.343889
Noronha NC, Mizukami A, Caliári-Oliveira C et al (2019) Priming approaches to improve the efficacy of mesenchymal stromal cell-based therapies. Stem Cell Res Ther 10:131. https://doi.org/10.1186/s13287-019-1224-y
Park JS, Suryaprakash S, Lao YH, Leong KW (2015) Engineering mesenchymal stem cells for regenerative medicine and drug delivery. Methods 84:3–16. https://doi.org/10.1016/j.ymeth.2015.03.002
Liu W, Rong Y, Wang J et al (2020) Exosome-shuttled miR-216a-5p from hypoxic preconditioned mesenchymal stem cells repair traumatic spinal cord injury by shifting microglial M1/M2 polarization. J Neuroinflammation 17:47. https://doi.org/10.1186/s12974-020-1726-7
Zhang Y, Wang WT, Gong CR, Li C, Shi M (2020) Combination of olfactory ensheathing cells and human umbilical cord mesenchymal stem cell-derived exosomes promotes sciatic nerve regeneration. Neural Regen Res 15:1903–1911. https://doi.org/10.4103/1673-5374.280330
Jiang CM, Liu J, Zhao JY et al (2015) Effects of hypoxia on the immunomodulatory properties of human gingiva-derived mesenchymal stem cells. J Dent Res 94:69–77. https://doi.org/10.1177/0022034514557671
Cui X, Wang H, Guo H et al (2010) Transplantation of mesenchymal stem cells preconditioned with diazoxide, a mitochondrial ATP-sensitive potassium channel opener, promotes repair of myocardial infarction in rats. Tohoku J Exp Med 220:139–147. https://doi.org/10.1620/tjem.220.139
Liu J, Zhu P, Song P et al (2015) Pretreatment of adipose derived stem cells with curcumin facilitates myocardial recovery via antiapoptosis and angiogenesis. Stem Cells Int 2015:638153. https://doi.org/10.1155/2015/638153
Schepici G, Gugliandolo A, Mazzon E (2022) Mesenchymal stromal cells preconditioning: a new strategy to improve neuroprotective properties. Int J Mol Sci 23:2088. https://doi.org/10.3390/ijms23042088
Chiu CT, Wang Z, Hunsberger JG, Chuang DM (2013) Therapeutic potential of mood stabilizers lithium and valproic acid: beyond bipolar disorder. Pharmacol Rev 65:105–142. https://doi.org/10.1124/pr.111.005512
Oses C, Olivares B, Ezquer M et al (2017) Preconditioning of adipose tissue-derived mesenchymal stem cells with deferoxamine increases the production of pro-angiogenic, neuroprotective and anti-inflammatory factors: potential application in the treatment of diabetic neuropathy. PLoS One 12:e0178011. https://doi.org/10.1371/journal.pone.0178011
Adlerz K, Patel D, Rowley J, Ng K, Ahsan T (2020) Strategies for scalable manufacturing and translation of MSC-derived extracellular vesicles. Stem Cell Res 48:101978. https://doi.org/10.1016/j.scr.2020.101978
Wang X, Chen Y, Zhao Z et al (2018) Engineered exosomes with ischemic myocardium-targeting peptide for targeted therapy in myocardial infarction. J Am Heart Assoc 7:e008737. https://doi.org/10.1161/JAHA.118.008737
Wu P, Zhang B, Ocansey DKW, Xu W, Qian H (2021) Extracellular vesicles: a bright star of nanomedicine. Biomaterials 269:120467. https://doi.org/10.1016/j.biomaterials.2020.120467
Hassanzadeh A, Rahman HS, Markov A et al (2021) Mesenchymal stem/stromal cell-derived exosomes in regenerative medicine and cancer; overview of development, challenges, and opportunities. Stem Cell Res Ther 12:297. https://doi.org/10.1186/s13287-021-02378-7
Sutaria DS, Badawi M, Phelps MA, Schmittgen TD (2017) Achieving the promise of therapeutic extracellular vesicles: the devil is in details of therapeutic loading. Pharm Res 34:1053–1066. https://doi.org/10.1007/s11095-017-2123-5
Shen H, Yao X, Li H et al (2018) Role of exosomes derived from miR-133b modified MSCs in an experimental rat model of intracerebral hemorrhage. J Mol Neurosci 64:421–430. https://doi.org/10.1007/s12031-018-1041-2
Xin H, Li Y, Liu Z et al (2013) MiR-133b promotes neural plasticity and functional recovery after treatment of stroke with multipotent mesenchymal stromal cells in rats via transfer of exosome-enriched extracellular particles. Stem Cells 31:2737–2746. https://doi.org/10.1002/stem.1409
Yuan B, Pan S, Dong Y, Zhang W, He X (2020) Effect of exosomes derived from mir-126-modified mesenchymal stem cells on the repair process of spinal cord injury in rats. Eur Rev Med Pharmacol Sci 24:483–490. https://doi.org/10.26355/eurrev_202001_20025
Chen Y, Tian Z, He L et al (2021) Exosomes derived from miR-26a-modified MSCs promote axonal regeneration via the PTEN/AKT/mTOR pathway following spinal cord injury. Stem Cell Res Ther 12:224. https://doi.org/10.1186/s13287-021-02282-0
Yang M, Chen J, Chen L (2022) The roles of mesenchymal stem cell-derived exosomes in diabetes mellitus and its related complications. Front Endocrinol (Lausanne) 13:1027686. https://doi.org/10.3389/fendo.2022.1027686
Gomari H, Forouzandeh Moghadam M, Soleimani M (2018) Targeted cancer therapy using engineered exosome as a natural drug delivery vehicle. Onco Targets Ther 11:5753–5762. https://doi.org/10.2147/OTT.S173110
Melzer C, Rehn V, Yang Y, Bähre H, von der Ohe J, Hass R (2019) Taxol-loaded MSC-derived exosomes provide a therapeutic vehicle to target metastatic breast cancer and other carcinoma cells. Cancers (Basel) 11:798. https://doi.org/10.3390/cancers11060798
Chen S, Sun F, Qian H, Xu W, Jiang J (2022) Preconditioning and engineering strategies for improving the efficacy of mesenchymal stem cell-derived exosomes in cell-free therapy. Stem Cells Int 2022:1779346. https://doi.org/10.1155/2022/1779346
Yang Y, Hong Y, Nam GH, Chung JH, Koh E, Kim IS (2017) Virus-mimetic fusogenic exosomes for direct delivery of integral membrane proteins to target cell membranes. Adv Mater 29:1605604. https://doi.org/10.1002/adma.201605604
Zhang N, Song Y, Huang Z et al (2020) Monocyte mimics improve mesenchymal stem cell-derived extracellular vesicle homing in a mouse MI/RI model. Biomaterials 255:120168. https://doi.org/10.1016/j.biomaterials.2020.120168
Newton WC, Kim JW, Luo JZQ, Luo L (2017) Stem cell-derived exosomes: a novel vector for tissue repair and diabetic therapy. J Mol Endocrinol 59:R155–R165. https://doi.org/10.1530/JME-17-0080
Sun B, Peng J, Wang S et al (2018) Applications of stem cell-derived exosomes in tissue engineering and neurological diseases. Rev Neurosci 29:531–546. https://doi.org/10.1515/revneuro-2017-0059
Petersen KE, Manangon E, Hood JL et al (2014) A review of exosome separation techniques and characterization of B16-F10 mouse melanoma exosomes with AF4-UV-MALS-DLS-TEM. Anal Bioanal Chem 406:7855–7866. https://doi.org/10.1007/s00216-014-8040-0
Tang Y, Zhou Y, Li HJ (2021) Advances in mesenchymal stem cell exosomes: a review Stem Cell Res Ther 12:71. https://doi.org/10.1186/s13287-021-02138-7
Elliott RO, He M (2021) Unlocking the power of exosomes for crossing biological barriers in drug delivery. Pharmaceutics 13:122. https://doi.org/10.3390/pharmaceutics13010122
Jia Y, Ni Z, Sun H, Wang C (2019) Microfluidic approaches toward the isolation and detection of exosome nanovesicles. Ieee Access 7:45080–45098. https://doi.org/10.1109/ACCESS.2019.2907123
Madrigal M, Rao KS, Riordan NH (2014) A review of therapeutic effects of mesenchymal stem cell secretions and induction of secretory modification by different culture methods. J Transl Med 12:260. https://doi.org/10.1186/s12967-014-0260-8
Joo HS, Suh JH, Lee HJ, Bang ES, Lee JM (2020) Current knowledge and future perspectives on mesenchymal stem cell-derived exosomes as a new therapeutic agent. Int J Mol Sci 21:727. https://doi.org/10.3390/ijms21030727
Gao W, Liang T, He R, Ren J, Yao H, Wang K, Zhu L, Xu Y (2020) Exosomes from 3D culture of marrow stem cells enhances endothelial cell proliferation, migration, and angiogenesis via activation of the HMGB1/AKT pathway. Stem Cell Res 50:102122. https://doi.org/10.1016/j.scr.2020.102122
Li Q, Xu Y, Lv K, Wang Y, Zhong Z, Xiao C et al (2021) Small extracellular vesicles containing miR-486-5p promote angiogenesis after myocardial infarction in mice and nonhuman primates. Sci Transl Med 13:eabb0202. https://doi.org/10.1126/scitranslmed.abb0202
Kwon S, Shin S, Do M, Oh BH, Song Y, Bui VD, Lee ES, Jo DG et al (2021) Engineering approaches for effective therapeutic applications based on extracellular vesicles. J Control Release 330:15–30. https://doi.org/10.1016/j.jconrel.2020.11.062
Zhang Y, Bi J, Huang J, Tang Y, Du S, Li P (2020) Exosome: a review of its classification, isolation techniques, storage, diagnostic and targeted therapy applications. Int J Nanomedicine 15:6917–6934. https://doi.org/10.2147/IJN.S264498
Zhang Z, Mugisha A, Fransisca S, Liu Q, Xie P, Hu Z (2021) Emerging role of exosomes in retinal diseases. Front Cell Dev Biol 9:643680. https://doi.org/10.3389/fcell.2021.643680
Vizoso FJ, Eiro N, Cid S, Schneider J, Perez-Fernandez R (2017) Mesenchymal stem cell secretome: toward cell-free therapeutic strategies in regenerative medicine. Int J Mol Sci. 18:1852. https://doi.org/10.3390/ijms18091852
Acknowledgements
All of the persons contributed to the manuscript were placed as author.
Funding
Open access funding provided by The Science, Technology & Innovation Funding Authority (STDF) in cooperation with The Egyptian Knowledge Bank (EKB). The authors declare that no funds, grants, or other support was received during the preparation of this manuscript.
Author information
Authors and Affiliations
Contributions
LA designed the original idea, developed it in detail, provided valuable feedback, and helped in writing and revising the draft.
KA-M wrote the original draft preparation and helped in the design of table and figures. Both authors participated in drafting, writing and editing the manuscript, and approving it for submission.
Corresponding author
Ethics declarations
Ethics Approval
There are no animal experiments carried out for this article.
Consent to Participate
Not applicable
Consent for Publication
Not applicable
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Ahmed, L.A., Al-Massri, K.F. Exploring the Role of Mesenchymal Stem Cell–Derived Exosomes in Diabetic and Chemotherapy-Induced Peripheral Neuropathy. Mol Neurobiol 61, 5916–5927 (2024). https://doi.org/10.1007/s12035-024-03916-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12035-024-03916-z