Abstract
Purpose
The roles of endothelial nitric oxide synthase (eNOS) gene polymorphisms in diabetes mellitus (DM) were intensively analyzed, but the results of these studies were inconsistent. Therefore, we performed this study to better assess the relationship between eNOS genetic variations and DM.
Methods
Eligible studies were searched in PubMed, Medline, Embase, and Web of Science. Odds ratios (ORs) with 95% confidence intervals (CIs) were used to assess correlations between eNOS polymorphisms and DM.
Results
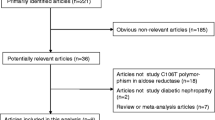
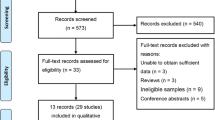
A total of 91 studies were finally included in our analyses. Significant associations with the susceptibility to DM were detected for the rs891512, rs1799983, rs2070744, and rs869109213 polymorphisms. As for vascular complications in DM, significant associations with the susceptibility to diabetic nephropathy were detected for the rs1799983 and rs2070744 polymorphisms. In addition, we also found that the rs1799983 polymorphism was significantly associated with the susceptibility to peripheral artery disease, whereas the rs2070744 polymorphism was significantly associated with the susceptibility to coronary artery disease in DM patients. Further subgroup analyses on the basis of type of disease and ethnicity of participants showed similar positive results.
Conclusions
In conclusion, our findings indicate that rs891512, rs1799983, rs2070744, and rs869109213 polymorphisms may serve as genetic biomarkers of DM, while rs1799983, rs2070744, and rs869109213 polymorphisms may contribute to the development of vascular complications in DM.

Similar content being viewed by others
References
D.J. Moore, J.M. Gregory, Y.A. Kumah-Crystal, J.H. Simmons, Mitigating micro-and macro-vascular complications of diabetes beginning in adolescence. Vasc. Health Risk. Manag. 5, 1015–1031 (2009)
American Diabetes Association, Diagnosis and classification of diabetes mellitus. Diabetes Care 37(Suppl 1), S81–S90 (2014)
A.K. Papazafiropoulou, N. Papanas, A. Melidonis, E. Maltezos, Family History of type 2 diabetes: Does having a diabetic parent increase the risk? Curr. Diabetes Rev. 13, 19–25 (2017)
K.J. Gaulton, Mechanisms of type 2 diabetes risk loci. Curr. Diab. Rep. 17, 72 (2017)
S. Jamwal, S. Sharma S, Vascular endothelium dysfunction: a conservative target in metabolic disorders. Inflamm. Res. (2018). https://doi.org/10.1007/s00011-018-1129-8
M. Tsutsui, H. Shimokawa, T. Morishita, Y. Nakashima, N. Yanagihara, Development of genetically engineered mice lacking all three nitric oxide synthases. J. Pharmacol. Sci. 102, 147–154 (2006)
H. Li, T. Wallerath, T. Münzel, U. Förstermann, Regulation of endothelial-type NO synthase expression in pathophysiology and in response to drugs. Nitric Oxide 7, 149–164 (2002)
D. Moher, A. Liberati, J. Tetzlaff, D.G. Altman, PRISMA group, Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. Ann. Intern. Med. 151, 264–269 (2009)
A. Stang, Critical evaluation of the Newcastle-Ottawa scale for the assessment of the quality of nonrandomized studies in meta-analyses. Eur. J. Epidemiol. 25, 603–605 (2010)
D.M. Nathan, Diabetes: advances in diagnosis and treatment. JAMA 314, 1052–1062 (2015)
GBD 2013 Mortality and Causes of Death Collaborators, Global, regional, and national age-sex specific all-cause and cause-specific mortality for 240 causes of death, 1990-2013: a systematic analysis for the Global Burden of Disease Study 2013. Lancet 385, 117–171 (2015)
C.J. Murray, C. Atkinson, K. Bhalla, G. Birbeck, R. Burstein, D. Chou et al.The state of US health, 1990-2010: burden of diseases, injuries, and risk factors. JAMA 310, 591–608 (2013)
K. Antosik, M. Borowiec, Genetic factors of diabetes. Arch. Immunol. Ther. Exp. (Warsz.) 64(Suppl 1), 157–160 (2016)
H.H. Hoang, S.V. Padgham, C.J. Meininger, L-arginine, tetrahydrobiopterin, nitric oxide and diabetes. Curr. Opin. Clin. Nutr. Metab. Care 16, 76–82 (2016)
T. Nakagawa, R.J. Johnson, Endothelial nitric oxide synthase. Contrib. Nephrol. 170, 93–101 (2011)
B.S. Dellamea, L.C. Pinto, C.B. Leitão, K.G. Santos, L.H. Canani, Endothelial nitric oxide synthase gene polymorphisms and risk of diabetic nephropathy: a systematic review and meta-analysis. BMC Med. Genet. 15, 9 (2014)
Z. Jia, X. Zhang, S. Kang, Y. Wu, Association of endothelial nitric oxide synthase gene polymorphisms with type 2 diabetes mellitus: a meta-analysis. Endocr. J. 60, 893–901 (2013)
S. Zhao, T. Li, B. Zheng, Z. Zheng, Nitric oxide synthase 3 (NOS3) 4b/a, T-786C and G894T polymorphisms in association with diabetic retinopathy susceptibility: a meta-analysis. Ophthalmic Genet. 33, 200–207 (2012)
Y. He, Z. Fan, J. Zhang, Q. Zhang, M. Zheng, Y. Li, D. Zhang, S. Gu, H. Yang, Polymorphisms of eNOS gene are associated with diabetic nephropathy: a meta-analysis. Mutagenesis 26, 339–349 (2011)
Authors contributions
J.D. and Y.Z. conceived of the study, participated in its design. J.D. and P.Y. conducted the systematic literature review. J.D., P.Y., and Y.W. performed data analyses. J.D. and Y.Z. drafted the manuscript. All authors have read and approved the final manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
These authors contributed equally: Jinnan Dong, Ping Yu.
Rights and permissions
About this article
Cite this article
Dong, J., Ping, Y., Wang, Y. et al. The roles of endothelial nitric oxide synthase gene polymorphisms in diabetes mellitus and its associated vascular complications: a systematic review and meta-analysis. Endocrine 62, 412–422 (2018). https://doi.org/10.1007/s12020-018-1683-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12020-018-1683-4