Abstract
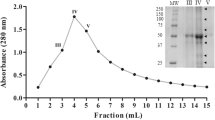
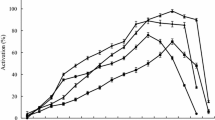
A plant protease named microcarpain was purified from the latex of Ficus microcarpa by acetonic (20–40 % saturation) precipitation, Sephadex G-75 filtration, and Mono Q-Sefinose FF chromatography. The protease was purified with a yield of 9.25 % and a purification factor of 8. The molecular weight of the microcarpain was estimated to be 20 kDa by sodium dodecyl sulfate polyacrylamide gel electrophoresis (SDS-PAGE). The purified enzyme showed maximum activity at pH 8.0 and at a temperature of 70 °C. Proteolytic activity was strongly inhibited by dithio-bis-nitrobenzoic acid (DTNB), Hg2+, and Cu2+. The N-terminal amino acid sequence of the purified microcarpain “VPETVDWRSKGAV” showed high homology with a protease from Arabidopsis thaliana. Inhibition studies and N-terminal sequence classified the enzyme as a member of the cysteine peptidases family.




Similar content being viewed by others
References
Turk, B. (2006). Nature Reviews Drug Discovery, 5, 785–798.
Frost and Sullivan. (2007). Feed enzymes: the global scenario. London, UK.
Schaller, A. (2004). Planta, 220, 183–197.
Boller, T. (1986), in Plant proteolytic enzymes, vol. 1: roles of proteolytic enzymes in interactions of plants with other organisms (Dalling, M.J., ed), Florida, USA, pp. 67–96.
Baker, E. N., & Drenth, J. (1987). Biological macromolecules and assemblies, vol. 3. In F. Jurnak & A. McPherson (Eds.), The cysteine proteinases structure and mechanism (pp. 313–368). New York: Wiley.
Caffini, N. O., Lopez, L. M. I., Natalucci, C. L., & Priolo, N. S. (1988). Acta Farmaceutica Bonaerense, 7, 195–213.
Van der Hoorn, R. A., & Jones, J. D. (2004). Current Opinion in Plant Biology, 7, 400–407.
Souza, D. P., Freitas, C. D. T., Pereira, D. A., Nogueira, F. C., Silva, F. D. A., Salas, C. E., & Ramos, M. V. (2011). Planta, 234, 183–193.
Shivaprasad, H. V., Rajesh, R., Nanda, B. L., Dharmappa, K. K., & Vishwanath, B. S. (2009). Journal of Ethnopharmacology, 123, 106–109.
Fonseca, K. C., Morais, N. C. G., Queiroz, M. R., Silva, M. C., Gomes, M. S., Costa, J. O., Mamede, C. C. N., Torres, F. S., Penha-Silva, N., Beletti, M. E., Canabrava, H. A. N., & Oliveiraa, F. (2010). Phytochemistry, 71, 708–715.
Freitas, C. D. T., Oliveira, J. S., Miranda, M. R. A., Macedo, N. M. R., Pereira Sales, M., Villas Boas, L. A., & Ramos, M. V. (2007). Plant Physiology and Biochemistry, 45, 781–789.
Vierstra, R. D. (1996). Plant Molecular Biology, 32, 275.
Ervatamia. (1952), in The wealth of India, vol. 3: Council of Scientific and Industrial Research, New Delhi, pp. 192–193.
Gunter, R., Hans, P. S., Friedrich, D., & Peter, L. (2002). British Journal of Haematology, 119, 1042–1051.
Badgujar, S. B. (2014). Journal of Ethnopharmacology, 151, 733–739.
Thankamma, L. (2003). Current Science, 84, 971–972.
Domsalla, A., & Melzig, M. F. (2008). Planta Medica, 74, 699–711.
Wagner, W. L., Herbst, D. R., & Sohmer, S. H. (1999). Manual of the flowering plants of Hawaii, revised edition. 2 vols, Bishop Museum Special Publication 97. Honolulu: University of Hawaii Press and Bishop Museum Press.
Bradford, M. (1976). Analytical Biochemistry, 72, 248–254.
Kembhavi, A. A., Kulkarni, A., & Pant, A. (1993). Applied Biochemistry and Biotechnology, 38, 83–92.
Laemmli, U. K. (1970). Nature, 227, 680–685.
Garcia-Carreno, F. L., Dimes, L. E., & Haard, N. F. (1993). Analytical Biochemistry, 214, 65–69.
Shivaprasad, H. V., Rajaiah, R., Frey, B. M., Frey, F. J., & Vishwanath, B. S. (2010). Thrombosis Research, 125, 100–105.
Liggieri, C., Obregon, W., Trejo, S., & Priolo, N. (2009). Acta Biochimica et Biophysica Sinca, 41, 154–162.
Singh, A. N., Shukla, A. K., Jagannadham, M. V., & Dubey, V. K. (2010). Process Biochemistry, 45, 399–406.
Torres, M. J., Trejo, S. A., Obregon, W. D., Aviles, F. X., Lopez, L. M. I., & Natalucci, C. L. (2012). Planta, 236, 1471–1484.
Hashim, M. M., Mingsheng, D., Iqbal, M. F., & Xiaohong, C. (2011). Phytochemistry, 72, 458–464.
Revell, D. F., Cummings, N. J., Baker, K. C., Collins, M. E., Taylor, M. A., Sumner, I. G., Pickersgill, R. W., Connerton, I. F., & Goodenough, P. W. (1993). Gene, 127, 221–225.
Vairo Cavalli, S. E., Arribere, M. C., Cortadi, A., Caffini, N. O., & Priolo, N. S. (2003). Journal of Protein Chemistry, 22, 15–22.
Morcelle del Valle, S. R., Trejo, S. A., Canals, F., Aviles, F. X., & Priolo, N. S. (2004). Protein Journal, 25, 205–215.
Vairo Cavalli, S. E., Cortadi, A., Arribere, M. C., Conforti, P., Caffini, N. O., & Priolo, N. S. (2001). Biological Chemistry, 382, 879–883.
Teixeira, R. D., Ribeiro, H. A. L., Gomes, M. T. R., Lopes, M. T. P., & Salas, C. E. (2008). Plant Physiology and Biochemistry, 46, 956–961.
Torres, M. J., Trejo, S. A., Natalucci, C. L., & Lopez, L. M. I. (2013). Planta, 237, 1651–1659.
Vasu, P., Savary, B. J., & Cameron, R. G. (2012). Food Chemistry, 133, 366–372.
Demir, Y., Güngör, A. A., Duran, E. D., & Demir, N. (2008). Food Technology and Biotechnology, 46, 286–291.
Chanda, I., Basu, S. K., Dutta, S. K., & Chanda Das, S. R. (2011). Tropical Journal of Pharmaceutical Research, 10, 705–711.
Dubey, V. K., & Jagannadham, M. V. (2003). Phytochemistry, 62, 1057–1071.
Fahmy, A. S., Ali, A. A., & Mohamed, S. A. (2004). Bioresource Technology, 91, 297–304.
Vallés, D., Furtado, S., & Cantera, A. M. B. (2007). Enzyme and Microbial Technology, 40, 409–413.
Male, R., Lorens, L. B., Smalas, A. O., & Torrissen, K. R. (1995). European Journal of Biochemistry, 232, 677–685.
Bezerra, R. S., Lins, E. J. F., Alencar, R. B., Paiva, P. M. G., Chaves, M. E. C., Coelho, L. C. B. B., & JrLB, C. (2005). Process Biochemistry, 40, 1829–1834.
Siala, R., Fakhfakh, N., Hamza-Mnif, I., Nasri, M., Vallaeys, T., & Sellami-Kamoun, A. (2012). Biotechnology and Bioprocess Engineering, 17, 556–564.
Acknowledgments
This work was funded by the Ministry of Higher Education and Scientific Research, Tunisia.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Mnif, I.H., Siala, R., Nasri, R. et al. A Cysteine Protease Isolated from the Latex of Ficus microcarpa: Purification and Biochemical Characterization. Appl Biochem Biotechnol 175, 1732–1744 (2015). https://doi.org/10.1007/s12010-014-1376-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12010-014-1376-2