Abstract
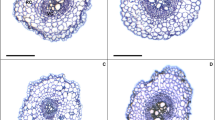
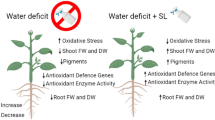
Water deficit is a type of abiotic stress that often limits seed germination, causing oxidative stress, cellular damage and physiological quality reduction. However, brassinosteroids regulate several biochemical and physiological processes in plants, reducing the oxidative stress caused by abiotic stresses, such as water deficit. The present study aimed to evaluate the contributions of 24-epibrassinolide (EBR) on the root anatomy and antioxidant enzymes as well as the possible interference of EBR on the oxidative stress, germination and biomass of soybean seedlings subjected to water deficit. The experiment had a factorial design, with the factors completely randomized, and two osmotic potentials (0.0 and − 0.3 MPa, as the control and water deficit, respectively) and three levels of brassinosteroids (0, 50 and 100 nM EBR). Soybean seedlings subjected to water deficit presented reductions of the root epidermis (RET), root endodermis (RDT), root cortex (RCT), vascular cylinder (VCD) and root metaxylem (RMD); however, the application of 100 nM brassinosteroids promoted increases in these values of 21, 12, 15, 38 and 15%, respectively, compared with water deficit + 0 nM brassinosteroids. In relation to antioxidant enzymes, the treatment with 100 nM brassinosteroids induced an increase in all of the enzymes evaluated. This study revealed that brassinosteroids promoted an increase in the germination, length and dry matter of soybean seedlings, and these results were explained by the increments in the root anatomy, particularly of the RET, RDT, RCT, VCD and RMD. Reductions in the concentrations of reactive oxygen species (superoxide and hydrogen peroxide) and membrane damage (malondialdehyde and electrolyte leakage) were intrinsically related to the higher activities of antioxidant enzymes (superoxide dismutase, catalase, ascorbate peroxidase and peroxidase), confirming the benefits of brassinosteroids on the antioxidant system. Moreover, 100 nM brassinosteroids resulted in a better dose–response in soybean seedlings exposed to water deficit.




Similar content being viewed by others
Abbreviations
- APX:
-
Ascorbate peroxidase
- ATP:
-
Adenosine triphosphate
- BRs:
-
Brassinosteroids
- CAT:
-
Catalase
- EBR:
-
24-Epibrassinolide
- EL:
-
Electrolyte leakage
- FCG:
-
First count to germination
- H2O2 :
-
Hydrogen peroxide
- HDM:
-
Hypocotyl dry matter
- LH:
-
Hypocotyl length
- MDA:
-
Malondialdehyde
- MTG:
-
Mean time to germination
- O2 − :
-
Superoxide
- PEG:
-
Polyethylene glycol
- POX:
-
Peroxidase
- RCT:
-
Root cortex thickness
- RDM:
-
Root dry matter
- RDT:
-
Root endodermis thickness
- RET:
-
Root epidermis thickness
- RL:
-
Root length
- RMD:
-
Root metaxylem diameter
- ROS:
-
Reactive oxygen species
- SOD:
-
Superoxide dismutase
- TDM:
-
Total dry matter
- TG:
-
Total germination
- TL:
-
Total length
- VCD:
-
Vascular cylinder diameter
References
Abedi T, Pakniyat H (2010) Antioxidant enzymes changes in response to drought stress in ten cultivars of oilseed rape (Brassica napus L.). Czech J Genet Plant Breed 46:27–34. https://doi.org/10.17221/67/2009-CJGPB
Ahammed GJ, Choudhary SP, Chen S et al (2013) Role of brassinosteroids in alleviation of phenanthrene–cadmium co-contamination-induced photosynthetic inhibition and oxidative stress in tomato. J Exp Bot 64:199–213. https://doi.org/10.1093/jxb/ers323
Akitha Devi MK, Giridhar P (2013) Variations in physiological response, lipid peroxidation, antioxidant enzyme activities, proline and isoflavones content in soybean varieties subjected to drought stress. Proc Natl Acad Sci India Sect B Biol Sci 85:35–44. https://doi.org/10.1007/s40011-013-0244-0
Ali B, Hasan SA, Hayat S et al (2008) A role for brassinosteroids in the amelioration of aluminium stress through antioxidant system in mung bean (Vigna radiata L. Wilczek). Environ Exp Bot 62:153–159. https://doi.org/10.1016/j.envexpbot.2007.07.014
Amzallag GN, Vaisman J (2006) Influence of brassinosteroids on initiation of the root gravitropic response in Pisum sativum seedlings. Biol Plantarum 50:283–286. https://doi.org/10.1007/s10535-006-0021-5
Ansari WA, Atri N, Singh B et al (2018) Morpho-physiological and biochemical responses of muskmelon genotypes to different degree of water deficit. Photosynthetica 56:1–12. https://doi.org/10.1007/s11099-018-0821-9
Anuradha S, Rao S (2001) Effect of brassinosteroids on salinity stress induced inhibition of seed germination and seedling growth of rice (Oryza sativa L.). Plant Growth Regul 1:151–153. https://doi.org/10.1023/A:1017590108484
Azhar N, Su N, Shabala L, Shabala S (2017) Exogenously applied 24-epibrassinolide (EBL) ameliorates detrimental effects of salinity by reducing K+ efflux via depolarization-activated K++ channels. Plant Cell Physiol 58:802–810. https://doi.org/10.1093/pcp/pcx026
Badawi GH, Yamauchi Y, Shimada E et al (2004) Enhanced tolerance to salt stress and water deficit by overexpressing superoxide dismutase in tobacco (Nicotiana tabacum) chloroplasts. Plant Sci 166:919–928. https://doi.org/10.1016/j.plantsci.2003.12.007
Bajguz A, Tretyn A (2003) The chemical characteristic and distribution of brassinosteroids in plants. Phytochemistry 62:1027–1046. https://doi.org/10.1016/S0031-9422(02)00656-8
Behnamnia M, Kalantari KM, Ziaie J (2009) The effects of brassinosteroid on the induction of biochemical changes in Lycopersicon esculentum under drought stress. Turk J Botany 33:417–428. https://doi.org/10.3906/bot-0806-12
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254. https://doi.org/10.1016/0003-2697(76)90527-3
Cakmak I, Horst WJ (1991) Effect of aluminium on lipid peroxidation, superoxide dismutase, catalase, and peroxidase activities in root tips of soybean (Glycine max). Physiol Plant 83:463–468. https://doi.org/10.1111/j.1399-3054.1991.tb00121.x
Cakmak I, Marschner H (1992) Magnesium deficiency and high light intensity enhance activities of superoxide dismutase, ascorbate peroxidase, and glutathione reductase in bean leaves. Plant Physiol 98:1222–1227. https://doi.org/10.1104/pp.98.4.1222
Choudhary SP, Yu J-Q, Yamaguchi-Shinozaki K et al (2012) Benefits of brassinosteroid crosstalk. Trends Plant Sci 17:594–605. https://doi.org/10.1016/j.tplants.2012.05.012
Cruz de Carvalho MH (2008) Drought stress and reactive oxygen species. Plant Signal Behav 3:156–165. https://doi.org/10.4161/psb.3.3.5536
Elstner EF, Heupel A (1976) Inhibition of nitrite formation from hydroxylammoniumchloride: a simple assay for superoxide dismutase. Anal Biochem 70:616–620. https://doi.org/10.1016/0003-2697(76)90488-7
FAO (2017) Food and Agriculture Organization of the United Nations. In: FAO statistical databases, 2014. http://faostat.fao.org/
Farooq M, Wahid A, Basra SMA, Islam-ud-Din (2009) Improving water relations and gas exchange with brassinosteroids in rice under drought stress. J Agron Crop Sci 195:262–269. https://doi.org/10.1111/j.1439-037X.2009.00368.x
Giannopolitis CN, Ries SK (1977) Superoxide dismutases: I. Occurrence in higher plants. Plant Physiol 59:309–314
Gill SS, Tuteja N (2010) Reactive oxygen species and antioxidant machinery in abiotic stress tolerance in crop plants. Plant Physiol Biochem 48:909–930. https://doi.org/10.1016/j.plaphy.2010.08.016
Gong M, Li Y-J, Chen S-Z (1998) Abscisic acid-induced thermotolerance in maize seedlings is mediated by calcium and associated with antioxidant systems. J Plant Physiol 153:488–496. https://doi.org/10.1016/S0176-1617(98)80179-X
Hameed M, Ashraf M, Naz N (2009) Anatomical adaptations to salinity in cogon grass [Imperata cylindrica (L.) Raeuschel] from the Salt Range, Pakistan. Plant Soil 322:229–238. https://doi.org/10.1007/s11104-009-9911-6
Havir EA, McHale NA (1987) Biochemical and developmental characterization of multiple forms of catalase in tobacco leaves. Plant Physiol 84:450–455. https://doi.org/10.1104/pp.84.2.450
Hosamani J, Dadlani M, Santha IM et al (2013) Biochemical phenotyping of soybean [Glycine max (L.) Merill] genotypes to establish the role of lipid peroxidation and antioxidant enzymes in seed longevity. Agric Res 2:119–126. https://doi.org/10.1007/s40003-013-0058-6
Hose E, Clarkson DT, Steudle E et al (2001) The exodermis: a variable apoplastic barrier. J Exp Bot 52:2245–2264. https://doi.org/10.1093/jexbot/52.365.2245
Jiang M, Zhang J (2001) Effect of abscisic acid on active oxygen species, antioxidative defence system and oxidative damage in leaves of maize seedlings. Plant Cell Physiol 42:1265–1273. https://doi.org/10.1016/j.plantsci.2005.04.004
Jin H, Do J, Shin S-J et al (2014) Exogenously applied 24-epi brassinolide reduces lignification and alters cell wall carbohydrate biosynthesis in the secondary xylem of Liriodendron tulipifera. Phytochemistry 101:40–51. https://doi.org/10.1016/j.phytochem.2014.02.003
Kadam NN, Yin X, Bindraban PS et al (2015) Does morphological and anatomical plasticity during the vegetative stage make wheat more tolerant of water deficit stress than rice? Plant Physiol 167:1389–1401. https://doi.org/10.1104/pp.114.253328
Kadam NN, Tamilselvan A, Lawas LMF et al (2017) Genetic control of plasticity in root morphology and anatomy of rice in response to water deficit. Plant Physiol 174:2302–2315. https://doi.org/10.1104/pp.17.00500
Kagale S, Divi UK, Krochko JE et al (2007) Brassinosteroid confers tolerance in Arabidopsis thaliana and Brassica napus to a range of abiotic stresses. Planta 225:353–364. https://doi.org/10.1007/s00425-006-0361-6
Kanwar MK, Bajguz A, Zhou J, Bhardwaj R (2017) Analysis of brassinosteroids in plants. J Plant Growth Regul 36:1002–1030. https://doi.org/10.1007/s00344-017-9732-4
Kartal G, Temel A, Arican E, Gozukirmizi N (2009) Effects of brassinosteroids on barley root growth, antioxidant system and cell division. Plant Growth Regul 58:261–267. https://doi.org/10.1007/s10725-009-9374-z
Li KR, Feng CH (2011) Effects of brassinolide on drought resistance of Xanthoceras sorbifolia seedlings under water stress. Acta Physiol Plant 33:1293–1300. https://doi.org/10.1007/s11738-010-0661-0
Li YH, Liu YJ, Xu XL et al (2012) Effect of 24-epibrassinolide on drought stress-induced changes in Chorispora bungeana. Biol Plant 56:192–196
Lima JV, Lobato AKS (2017) Brassinosteroids improve photosystem II efficiency, gas exchange, antioxidant enzymes and growth of cowpea plants exposed to water deficit. Physiol Mol Biol Plants 23:59–72. https://doi.org/10.1007/s12298-016-0410-y
Líška D, Martinka M, Kohanová J, Lux A (2016) Asymmetrical development of root endodermis and exodermis in reaction to abiotic stresses. Ann Bot 118:667–674. https://doi.org/10.1093/aob/mcw047
Liu EK, Mei XR, Yan CR et al (2016) Effects of water stress on photosynthetic characteristics, dry matter translocation and WUE in two winter wheat genotypes. Agric Water Manag 167:75–85. https://doi.org/10.1016/j.agwat.2015.12.026
Maia CF, Silva BRS, Lobato AKS (2018) Brassinosteroids positively modulate growth: physiological, biochemical and anatomical evidence using two tomato genotypes contrasting to dwarfism. J Plant Growth Regul 37:1–14. https://doi.org/10.1007/s00344-018-9802-2
Manivannan P, Jaleel CA, Kishorekumar A et al (2007) Changes in antioxidant metabolism of Vigna unguiculata (L.) Walp. by propiconazole under water deficit stress. Colloids Surfaces B Biointerfaces 57:69–74. https://doi.org/10.1016/j.colsurfb.2007.01.004
Meyer CJ, Peterson CA, Steudle E (2011) Permeability of Iris germanica’s multiseriate exodermis to water, NaCl, and ethanol. J Exp Bot 62:1911–1926. https://doi.org/10.1093/jxb/erq380
Michel BE, Kaufmann MR (1973) The osmotic potential of polyethylene glycol 6000. Plant Physiol 51:914–916. https://doi.org/10.1104/pp.51.5.914
Mujoo R, Trinh DT, Ng PKW (2003) Characterization of storage proteins in different soybean varieties and their relationship to tofu yield and texture. Food Chem 82:265–273. https://doi.org/10.1016/S0308-8146(02)00547-2
Nakano Y, Asada K (1981) Hydrogen peroxide is scavenged by ascorbate-specific peroxidase in spinach chloroplasts. Plant Cell Physiol 22:867–880
Nishinari K, Fang Y, Guo S, Phillips GO (2014) Soy proteins: a review on composition, aggregation and emulsification. Food Hydrocoll 39:301–318. https://doi.org/10.1016/j.foodhyd.2014.01.013
O’Brien TP, Feder N, McCully ME (1964) Polychromatic staining of plant cell walls by toluidine blue O. Protoplasma 59:368–373. https://doi.org/10.1007/BF01248568
Pandey P, Srivastava RK, Rajpoot R et al (2015) Water deficit and aluminum interactive effects on generation of reactive oxygen species and responses of antioxidative enzymes in the seedlings of two rice cultivars differing in stress tolerance. Environ Sci Pollut Res 23:1516–1528. https://doi.org/10.1007/s11356-015-5392-8
Peña-Valdivia CB, Sánchez-Urdaneta AB, Meza Rangel J et al (2010) Anatomical root variations in response to water deficit: wild and domesticated common bean (Phaseolus vulgaris L.). Biol Res 43:417–427. https://doi.org/10.4067/S0716-97602010000400006
Qin D, Wang H, Zhang C et al (2017) Effects of GA3 and ABA on the respiratory pathways during the secondary bud burst in black currants. J For Res 28:705–712. https://doi.org/10.1007/s11676-016-0354-7
Ramakrishna B, Rao SSR (2015) Foliar application of brassinosteroids alleviates adverse effects of zinc toxicity in radish (Raphanus sativus L.) plants. Protoplasma 252:665–677. https://doi.org/10.1007/s00709-014-0714-0
Ranathunge K, Steudle E, Lafitte R (2003) Control of water uptake by rice (Oryza sativa L.): role of the outer part of the root. Planta 217:193–205. https://doi.org/10.1007/s00425-003-0984-9
Ribas-Carbo M (2005) Effects of water stress on respiration in soybean leaves. Plant Physiol 139:466–473. https://doi.org/10.1104/pp.105.065565
Ribeiro ES, Pereira MP, Castro EM et al (2015) Relationship between root anatomy on the uptake, storage and tolerance to lead of Echinodorus grandiflorus. Rev Bras Eng Agríc Ambient 19:605–612. https://doi.org/10.1590/1807-1929/agriambi.v19n6p605-612
Sade B, Soylu S, Yetim E (2011) Drought and oxidative stress. Afr J Biotechnol 10:11102–11109. https://doi.org/10.5897/AJB11.1564
Sanjukta S, Rai AK (2016) Production of bioactive peptides during soybean fermentation and their potential health benefits. Trends Food Sci Technol 50:1–10. https://doi.org/10.1016/j.tifs.2016.01.010
Sew YS, Ströher E, Fenske R, Millar AH (2016) Loss of mitochondrial malate dehydrogenase activity alters seed metabolism impairing seed maturation and post-germination growth in arabidopsis. Plant Physiol 171:849–863. https://doi.org/10.1104/pp.16.01654
Soltani A, Gholipoor M, Zeinali E (2006) Seed reserve utilization and seedling growth of wheat as affected by drought and salinity. Environ Exp Bot 55:195–200. https://doi.org/10.1016/j.envexpbot.2004.10.012
Steel RG, Torrie JH, Dickey DA (2006) Principles and procedures of statistics: a biometrical approach, 3rd edn. Academic Internet Publishers, Moorpark
Talaat NB, Shawky BT, Ibrahim AS (2015) Alleviation of drought-induced oxidative stress in maize (Zea mays L.) plants by dual application of 24-epibrassinolide and spermine. Environ Exp Bot 113:47–58. https://doi.org/10.1016/j.envexpbot.2015.01.006
Tian F, Jia T, Yu B (2014) Physiological regulation of seed soaking with soybean isoflavones on drought tolerance of Glycine max and Glycine soja. Plant Growth Regul 74:229–237. https://doi.org/10.1007/s10725-014-9914-z
Velikova V, Yordanov I, Edreva A (2000) Oxidative stress and some antioxidant systems in acid rain-treated bean plants protective role of exogenous polyamines. Plant Sci 151:59–66. https://doi.org/10.1016/S0168-9452(99)00197-1
Wang B, Zhang J, Xia X, Zhang W-H (2011) Ameliorative effect of brassinosteroid and ethylene on germination of cucumber seeds in the presence of sodium chloride. Plant Growth Regul 65:407–413. https://doi.org/10.1007/s10725-011-9595-9
Wu Q-S, Xia R-X, Zou Y-N (2006) Reactive oxygen metabolism in mycorrhizal and non-mycorrhizal citrus (Poncirus trifoliata) seedlings subjected to water stress. J Plant Physiol 163:1101–1110. https://doi.org/10.1016/j.jplph.2005.09.001
Wu X, Yao X, Chen J et al (2014) Brassinosteroids protect photosynthesis and antioxidant system of eggplant seedlings from high-temperature stress. Acta Physiol Plant 36:251–261. https://doi.org/10.1007/s11738-013-1406-7
Xue Z-C, Gao H-Y, Zhang L-T (2013) Effects of cadmium on growth, photosynthetic rate and chlorophyll content in leaves of soybean seedlings. Biol Plant 57:587–590. https://doi.org/10.1007/s10535-013-0318-0
Zhang M, Zhai Z, Tian X et al (2008) Brassinolide alleviated the adverse effect of water deficits on photosynthesis and the antioxidant of soybean (Glycine max L.). Plant Growth Regul 56:257–264. https://doi.org/10.1007/s10725-008-9305-4
Zhang H, Jiao H, Jiang C-X et al (2010) Hydrogen sulfide protects soybean seedlings against drought-induced oxidative stress. Acta Physiol Plant 32:849–857. https://doi.org/10.1007/s11738-010-0469-y
Acknowledgements
This research received financial support from Fundação Amazônia de Amparo a Estudos e Pesquisas (FAPESPA/Brazil), Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq/Brazil) and Universidade Federal Rural da Amazônia (UFRA/Brazil) to AKSL, while DGSR was supported with scholarship from Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES/Brazil).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Additional information
Communicated by B. Zheng.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Ribeiro, D.G.d., Silva, B.R.S. & Lobato, A.K.d. Brassinosteroids induce tolerance to water deficit in soybean seedlings: contributions linked to root anatomy and antioxidant enzymes. Acta Physiol Plant 41, 82 (2019). https://doi.org/10.1007/s11738-019-2873-2
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11738-019-2873-2