Abstract
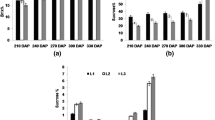
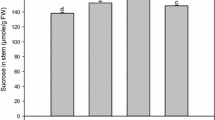
Sugarcane accumulates high amount of sucrose, thus making it one of the important cash crops worldwide. The final destination of sucrose accumulation in sugarcane is sink tissue, i.e., stalk, supplied by the source, i.e., leaf, to fulfill the need of plant growth, respiration, storage, and other metabolic activities. Signals between sink and source tissues regulate sucrose accumulation in sink and possibly the negative feedback from the sink restrains further accumulation in the stalk. However, perturbation of this negative feedback may help to improve sugar yield. This can be achieved by the application of GA3 (Gibberellic acid), a plant growth regulator, known to excite physiological responses and modify the source–sink metabolism through their effect on photosynthesis, which in turn improves sink strength by redistribution of the photoassimilates. In the present study, GA3 applied canes showed prominent increase in invertase activity, at early stage of the application, to provide hexoses. This in turn helped increase the internodal length and cane capacity for additional accumulation of sucrose, thereby increasing sink strength. At maturity, sucrose% and brix% were found higher in middle and top portions of the GA3-applied canes. Expression analysis of various sucrose metabolising genes viz., sucrose phosphate synthase (SPS), sucrose synthase (SuSy), soluble acid invertase, neutral invertase, and cell wall invertase (CWI) was carried out at different growth stages, using quantitative RT-PCR. CWI, which plays key role in phloem unloading in sink tissues, exhibited higher expression in GA3 samples at the elongation stage which decreased with maturity, whereas both SuSy and SPS, involved in regulation of sucrose accumulation, showed a variable level of expression. Thus, GA3 application on cane may improve the sucrose content in stalk and thus assuage maneuvering source–sink dynamics in sugarcane.









Similar content being viewed by others
Abbreviations
- CWI:
-
Cell wall invertase
- GA3 :
-
Gibberellic acid
- NI:
-
Neutral invertase
- SAI:
-
Soluble acid invertase
- SPS:
-
Sucrose phosphate synthase
- SuSy:
-
Sucrose synthase
- qRT-PCR:
-
Quantitative reverse transcriptase PCR
References
Aloni B, Daie J, Wyse RE (1986) Enhancement of [14C] sucrose export from source leaves of Vicia faba by gibberellic acid. Plant Physiol 82:962–966
Batta SK, Singh R (1986) Sucrose metabolism in sugarcane grown under varying climatic conditions: synthesis and storage of sucrose in relation to the activities of sucrose synthase, sucrose phosphate synthase and invertase. Phytochemistry 25:2431–2437
Bosch S, Grof CPL, Botha FC (2004) Expression of neutral invertase in sugarcane. Plant Sci 166:1125–1133
Botha FC, Black KG (2000) Sucrose phosphate synthase and sucrose synthase activity during maturation of intermodal tissue in sugarcane. Aust J Plant Physiol 27:81–85
Botha FC, Sawyer BJB, Birch RG (2001) Sucrose metabolism in the culm of transgenic sugarcane with reduced soluble acid invertase activity. Proc Intl Soc Sugar Cane Technol 24:588–591
Botha FC, Lakshmanan P, O’Connell A, Moore PH (2013) Hormones and growth regulators. In: Moore PH, Botha FC (eds) Sugarcane: physiology, biochemistry, and functional biology. Wiley, Chichester, pp 331–377. doi:10.1002/9781118771280
Buczynski SR, Thom M, Chourey P, Maretzki A (1993) Tissue distribution and characterization of sucrose synthase isozymes in sugarcane. J Plant Physiol 142:641–646
Bull TA, Glasziou KT (1963) The evolutionary significance of sugar accumulation in Saccharum. Aust J Biol Sci 16:737–742
Buren LL, Moore PH, Yamasaki Y (1979) Gibberellin studies with sugarcane. II. Hand-sampled field trials. Crop Sci 19:425–428
Burg SP, Bieleski RL (1962) The physiology of sugarcane. V. Kinetics of sugar accumulation. Aust J Biol Sci 15:429–444
Chandra A, Jain R, Solomon S (2012) Complexities of invertases controlling sucrose accumulation and retention in sugarcane: the way forward. Curr Sci 102:857–866
Chandra A, Verma PK, Islam MN, Grisham MP, Jain R, Sharma A, Roopendra K, Singh K, Singh P, Verma I, Solomon S (2015) Expression analysis of genes associated with sucrose accumulation in sugarcane (Saccharum spp. hybrids) varieties differing in content and time of peak sucrose storage. Plant Biol 17:608–617
Cheikh N, Brenner ML, Huber JL, Huber SC (1992) Regulation of sucrose phosphate synthase by gibberellins in soybean and spinach plants. Plant Physiol 100:1238–1242
Chen WS, Liu ZH, Yang L, Chen WH (1994) Gibberellin and temperature influence carbohydrate content and flowering in Phalaenopsis. Physiol Plant 90:391–395
Cheng WH, Taliercio EW, Chourey PS (1996) The Miniaturel seed locus of maize encodes a cell wall invertase required for normal development of endosperm and maternal cells in the pedicel. Plant Cell 8:971–983
Chiou TJ, Bush DR (1998) Sucrose is a signal molecule in assimilate partitioning. Proc Natl Acad Sci 95:4784–4788
Clowes M (1980) Growth stimulation from ethrel and the effects of gibberellic acid when applied to sugarcane. Proc South Afr Sugar Technol Assoc 49:146–150
Cole DF, Dobrenz AK, Massengale MA (1972) Effect of gibberellic acid on alfalfa (Medicago sativa L.). Crop Sci 12:674–676
Davies PJ (1995) Plant hormones, physiology, biochemistry and molecular biology. Kluwer, Dordrecht
Doehlert DC, Huber SC (1985) The role of sulphahydryl groups in the regulation of spinach leaf sucrose phosphate synthase. Biochem Biophys Acta 93:353–355
Eschrich W (1980) Free space invertase, its possible role in phloem unloading. Berichte der Deutschen Botanischem Gesellschaft 93:363–378
Farrar JF (1993) Sink strength: what is it and how do we measure it? Introduction. Plant Cell Environ 16:10–15
Farrar J (1996) Regulation of shoot-root ratio is mediated by sucrose. Plant Soil 185:13–19
Gayler KR, Glasziou KT (1972) Storage of sugars in stalks of sugarcane. Bot Rev 38:471–490
Geigenberger P, Stitt M (1993) Sucrose synthase catalyses a readily reversible reaction in vivo in developing potato tubers and other plant tissues. Planta 189:329–339
Giaquinta RT (1979) Sucrose translocation and storage in the sugar beet. Plant Physiol 63:828–832
Gibson SI (2005) Control of plant development and gene expression by sugar signaling. Curr Opin Plant Biol 8:93–102
Goldner W, Thom M, Maretzki A (1991) Sucrose metabolism in sugarcane cell suspension cultures. Plant Sci 73:143–147
Groenewald JH, Botha FC (2007) Down-regulation of pyrophosphate: fructose 6- phosphate1-phosphotransferase (PFP) activity in sugarcane enhances sucrose accumulation in immature internodes. Transg Res 17:85–92
Grof CPL, Albertson PL, Bursle J, Perroux JM, Bonnett GD, Manners JM (2007) Sucrose-phosphate synthase, a biochemical marker of high sucrose accumulation in sugarcane. Crop Sci 47:1530–1539
Hänggi E, Fleming AJ (2001) Sucrose synthase expression pattern in young maize leaves: implications for phloem transport. Planta 214:326–329
Hatch MD, Glasziou KT (1963) Sugar accumulation cycle in sugarcane. II. Relationship of invertase activity to sugar content and growth rate in storage tissue of plants growth in controlled environments. Plant Physiol 38:344–348
Hoffmann-Thoma G, Hinkel K, Nicolay P, Willenbrink J (1996) Sucrose accumulation in sweet sorghum stem internodes in relation to growth. Physiol Plant 97:277–284
Huang D, Qin C, Gui Y, Zhao L, Chen Z, Wang M, Sun Y, Liao Q, Li Y, Lakshmanan P (2017) Role of the SPS gene families in the regulation of sucrose accumulation in sugarcane. Sugar Tech 19:117–124
Huber SC, Akazawa T (1986) A novel sucrose synthase pathway for sucrose degradation in cultured sycamore cells. Plant Physiol 81:1008–1013
Huber SC, Huber JL (1996) Role and regulation of sucrose phosphate synthase in higher plants. Ann Rev Plant Physiol Plant Mol Biol 47:431–444
Iqbal N, Nazar R, Khan MIR, Masood A, Khan NA (2011) Role of gibberellins in regulation of source–sink relations under optimal and limiting environmental conditions. Curr Sci 100:998–1007
Iskandar HM, Simpson RS, Casu RE, Bonnett GD, Maclean DJ, Manners JM (2004) Comparison of reference genes for quantitative real-time polymerase chain reaction analysis of gene expression in sugarcane. Plant Mol Biol Rep 22:325–337
Jackson PA (2005) Breeding for improved sugar content in sugarcane. Field Crops Resh 92:277–290
Jain R, Chandra A, Solomon S (2013) Impact of exogenously applied enzyme effectors on sucrose metabolizing enzymes (SPS, SS and SAI) and sucrose content in sugarcane. Sugar Tech 5:370–378
Kaufman PB, Ghosheh NS, LaCroix JD, Soni SL, Ikuma H (1973) Regulation of invertase levels in Avena stem segments by gibberellic acid, sucrose, glucose, and fructose. Plant Physiol 52:221–228
Khan NA, Singh S, Nazar R, Lone PM (2007) The source–sink relationship in mustard. Asian Aust J Plant Sci Biotech 1:10–18
Kozłowska M, Rybus-Zając M, Stachowiak J, Janowska B (2007) Changes in carbohydrate contents of Zantedeschia leaves under gibberellin-stimulated flowering. Acta Physiol Plant 29:27–32
Krapp A, Hofman B, Schäfer C, Stitt M (1993) Regulation of the expression of rbcS and other photosynthetic genes by carbohydrates:mechanism for the sink regulation of photosynthesis? Plant J 3:817–828
Lakshmanan P, Geijskes RJ, Aitken KS, Grof CLP, Bonnet GD, Smith GR (2005) Sugarcane biotechnology: the challenges and opportunities. In Vitro Cell Dev Biol Plant 41:345–363
Lalonde S, Tegeder M, Throne-Holst M, Frommer WB, Patrick JW (2003) Phloem loading and unloading of sugar and amino acids. Plant Cell Environ 26:37–56
Lingle SE, Smith RC (1991) Sucrose metabolism related to growth and ripening in sugarcane internodes. Crop Sci 31:172–177
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ (1951) Protein measurement with the Folin phenol reagent. J Biol Chem 193:265–275
Lunn JE, Furbank RT (1999) Sucrose biosynthesis in C4 plants. New Phytol 143:221–237
McCormick AJ, Cramer MD, Watt D (2006) Sink strength regulates photosynthesis in sugarcane. New Phytol 171:759–770
McCormick AJ, Watt DA, Cramer MD (2009) Supply and demand: sink regulation of sugar accumulation in sugarcane. J Exp Bot 60:357–364
Miller EM, Chourey PS (1992) The maize invertase-deficient miniature-l seed mutant is associated with aberrant pedicel and endosperm development. Plant Cell 4:297–305
Moore PH (1977) Use of gibberellic acid to increase sugarcane yields in Hawaii. Proc Plant Growth Regul Work Group 4:173–180
Moore PH (1995) Temporal and spatial regulation of sucrose metabolism in the sugarcane stem. Aust J Plant Physiol 22:661–679
Moore PH, Buren LL (1978) Gibberellin studies with sugarcane. I. Cultivar differences in growth responses to gibberellic acid. Crop Sci 17:443–446
Moore PH, Ginoza H (1980) Gibberellin studies with sugarcane. III. Effects of rate and frequency of gibberellic acid applications on stalk length and fresh weight. Crop Sci 20:78–82
Moore PH, Botha FC, Furbank RT, Grof CPL (1997) Potential for overcoming physio-biochemical limits to sucrose accumulation. In: Keating BA, Wilson JR (eds) Intensive sugarcane production: meeting the challenges beyond 2000. CAB International, Wallingford, pp 141–155
Morris DA, Arthur ED (1985) Effects of gibberellic acid on patterns of carbohydrate distribution and acid invertase activity in Phaseolus vulgaris. Physiol Plant 65:257–262
Nelson N (1944) A photometric adaptation of Somogyi method for the determination of glucose. J Biol Chem 153:375–380
Nguyen-Quoc B, Foyer CH (2001) A role for “futile cycles” involving invertase and sucrose synthase metabolism of tomato fruit. J Exp Bot 52:881–889
Ranwala AP, Miller WB (2008) Gibberellin-mediated changes in carbohydrate metabolism during flower stalk elongation in tulips. Plant Growth Regul 55:241–248
Sacher JA, Hatch MD, Glasziou KT (1963) Sugar accumulation cycle in sugarcane. III. Physical and metabolic aspect of cycle in immature storage tissues. Plant Physiol 39:348–354
Schafer WE, Rohwer JM, Botha FC (2004) Protein level expression and localization of sucrose synthase in sugarcane culm. Physiol Plant 121:187–195
Uys L, Botha FC, Hofmeyr JHS, Rohwer JM (2007) Kinetic model of sucrose accumulation in maturing sugarcane culm tissue. Phytochemistry 68:2375–2392
Verma AK, Upadhyaya SK, Srivastava MK, Verma PC, Solomon S, Singh SB (2011) Transcript expression and soluble acid invertase during sucrose accumulation in sugarcane. Acta Physiol Plant 33:1749–1757
Walsh KB, Sky RC, Brown SM (2005) The anatomy of the pathway of sucrose unloading within the sugarcane stalk. Funct Plant Biol 32:367–374
Whittaker A, Botha FC (1997) Carbon partitioning during sucrose accumulation in sugarcane internodal tissue. Plant Physiol 115:1651–1659
Wu L, Birch RG (2007) Doubling sugar content in sugarcane plants modified to produce a sucrose isomer. Plant Biotech J 5:109–117
Wu L, Mitchell JP, Cohn NS, Kaufman PB (1993) Gibberellin (GA3) enhances cell wall invertase activity and mRNA levels in elongating dwarf pea (Pisum sativum) shoots. Int J Plant Sci 154:280–289
Yamaguchi S (2008) Gibberellin metabolism and its regulation. Annu Rev Plant Biol 59:225–251
Zhang C, Tanabe K, Tamura F, Itai A, Yoshida M (2007) Roles of gibberellins in increasing sink demand in Japanese pear fruit during rapid fruit growth. Plant Growth Regul 52:161–172
Zhu YJ, Albert HH, Moore PH (1997) Sucrose accumulation in the sugarcane stem is regulated by the difference between the activities of soluble acid invertase and sucrose phosphate synthase. Plant Physiol 115:609–616
Zrenner R, Salanoubat M, Willmitzer L, Sonnewald U (1995) Evidence of the crucial role of sucrose synthase for sink strength using transgenic potato plants (Solanum tuberosum L.). Plant J 7:97–107
Acknowledgements
This work was supported by the Science and Engineering Research Board (SERB/SR/SO/PS/36/2012), Department of Science and Technology, Government of India. Authors are also thankful to the Director for necessary facilities to carry out the work.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by O. Ferrarese-Filho.
Rights and permissions
About this article
Cite this article
Verma, I., Roopendra, K., Sharma, A. et al. Expression analysis of genes associated with sucrose accumulation in sugarcane under normal and GA3-induced source–sink perturbed conditions. Acta Physiol Plant 39, 133 (2017). https://doi.org/10.1007/s11738-017-2433-6
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11738-017-2433-6