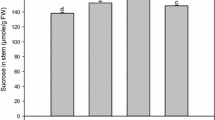
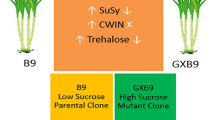
Abstract
Sucrose synthesis/accumulation in sugarcane depends on the source–sink communication wherein source responds to sink demand for photoassimilate supply. Sucrose in stalk (sink) acts as signal, and sends feedback to restrain further synthesis of sucrose by regulating photosynthetic efficiency of leaves (source). Hence sucrose synthesis/accumulation is controlled by many genes and regulatory sequences including 3 invertases (SAI, CWI, NI), sucrose synthase (SuSy) and sucrose phosphate synthase (SPS). SPS and invertase play key role in enhancing sink strength which ultimately promotes greater sucrose accumulation in the sink tissues. In present study, a significant positive correlation was found between sucrose% of source and sink tissues which was greater in the top (R2= 0.679) than middle (R2= 0.580) and bottom (R2= 0.518) internodes, depicting that sucrose accumulation in the stalk bears a direct relation with sucrose translocation efficiency from source. Results indicated an increased sucrose% with maturity, while reducing sugar content decreased with crop growth. qRT-PCR results exhibited an elevated expression of invertase in immature sink tissues depicting increased sink requirement, which declined with maturity. Similarly, increased PEP carboxylase gene expression as observed supported the fact that higher sink demand results in enhanced photosynthetic rate and thus influences the source activity. SPS was found active at initial stage of cane development indicating its role in sucrose synthesis. Thus by studying expression patterns of the different genes both, in source and sink tissues, a better understanding of the sucrose accumulation pathway in sugarcane is possible, which in turn can help in elucidating ways to enhance sucrose concentration in sink.









Similar content being viewed by others
References
Albertson PL, Peters KF, Grof CPL (2001) An improved method for the measurement of cell wall invertase activity in sugarcane tissue. Funct Plant Biol 28:323–328
Ap Rees T (1987) Compartmentation of plant metabolism. In: Davies DD (ed) The biochemistry of plants, 12th edn. Academic Press, San Diego, pp 87–115
Barratt DHP, Barber L, Kruger NJ, Smith AM, Wang TL, Martin C (2001) Multiple distinct isoforms of sucrose synthase in pea. Plant Physiol 127:655–664
Berding N (1997) Clonal improvement of sugarcane based on selection for moisture content: fact or fiction. Proc Aust Soc Sugarcane Technol 19:245–253
Bosch S, Grof CPL, Botha FC (2004) Expression of neutral invertase in sugarcane. Plant Sci 166:1125–1133
Buczynski SR, Thom M, Chourey P, Maretzki A (1993) Tissue distribution and characterization of sucrose synthase isozymes in sugarcane. J Plant Physiol 142:641–646
Bull TA, Glasziou KT (1963) The evolutionary significance of sugar accumulation in Saccharum. Aust J Biol Sci 16:737–742
Chandra A (2011) Physio-biochemical and molecular approaches to enhance sucrose content in sugarcane: Indian initiatives. Sugar Tech 13:315–321
Chandra A, Jain R, Rai RK, Solomon S (2011) Revisiting the source–sink paradigm in sugarcane. Curr Sci 100:978–980
Chandra A, Jain R, Solomon S (2012) Complexities of invertases controlling sucrose accumulation and retention in sugarcane: the way forward. Curr Sci 102:857–866
Chandra A, Verma PK, Islam MN, Grisham MP, Jain R, Sharma A, Roopendra K, Singh K, Singh P, Verma I, Solomon S (2015) Expression analysis of genes associated with sucrose accumulation in sugarcane (Saccharum spp. hybrids) varieties differing in content and time of peak sucrose storage. Plant Biol 17:608–617
Franck N, Vaast P, Ge´nard M, Dauzat J (2006) Soluble sugars mediate sink feedback down-regulation of leaf photosynthesis in field-grown Coffea arabica. Tree Physiol 26:517–525
Gayler KR, Glasziou KT (1972) Storage of sugars in stalks of sugarcane. Bot Rev 38:471–490
Geigenberger P, Stitt M (1993) Sucrose synthase catalyzes a rapidly reversible reaction in vivo in developing potato tubers and other plant tissues. Planta 189:329–339
Grof CPL, Campbell JA (2001) Sugarcane sucrose metabolism: scope for molecular manipulation. Aust J Plant Physiol 28:1–12
Grof CPL, Albertson PL, Bursle J, Perroux JM, Bonnett GD, Manners JM (2007) Sucrose –Phosphate Synthase, a biochemical marker of high sucrose accumulation in Sugarcane. Crop Sci 47:1530–1539
Gutierrez-Miceli FA, Rodriguez-Mendiola MA, Ochoa-Alejo N, Mendez-Salas R, Dendooven L, Arias-Castro C (2002) Relationship between sucrose accumulation and activities of sucrose-phosphatase, sucrose synthase, neutral invertase and soluble acid invertase in micropropagated sugarcane plants. Acta Physiol Plant 24:441–446
Hammond JBW, Burton KS, Shaw AF, Ho LC (1984) Source—sink relationships and carbon metabolism in tomato leaves 2. carbohydrate pools and catabolic enzymes. Ann Bot 53:307–314
Hatch MD, Glasziou KT (1963) Sugar accumulation cycle in sugarcane. II. Relationship of invertase activity to sugar content and growth rate in storage tissue of plants growth in controlled environments. Plant Physiol 38:344–348
Hibberd JM, Covshoff S (2010) The regulation of gene expression required for C4 photosynthesis. Annu Rev Plant Biol 61:181–207
Hubbard NI, Huber SC, Pharr DM (1989) Sucrose phosphate synthase and acid invertase as determinants of sucrose concentration in developing muskmelon (Cucumis melo L.) fruits. Plant Physiol 91:1527–1534
Inman-Bamber NG, Bonnett GD, Spillman MF, Hewitt ML, Jackson J (2008) Increasing sucrose accumulation in sugarcane by manipulating leaf extension and photosynthesis with irrigation. Aust J Agric Res 59:13–26
Iskandar HM, Simpson RS, Casu RE, Bonnett GD, Maclean DJ, Manners JM (2004) Comparison of reference genes for quantitative real-time polymerase chain reaction analysis of gene expression in sugarcane. Plant Mol Biol Rep 22:325–337
Jackson PA (2005) Breeding for improved sugar content in sugarcane. Field Crops Res 92:277–290
Jacobsen KR, Fisher DG, Maretzki A, Moore PH (1992) Developmental changes in the anatomy of the sugarcane stem in relation to phloem unloading and sucrose storage. Plant Biol 105:70–80
Jeschke WD, Hilpert A (1997) Sink-stimulated photosynthesis and sink-dependent increase in nitrate uptake: nitrogen and carbon relations of the parasitic association Cuscutareflexa-Ricinus communis. Plant, Cell Environ 20:47–56
Lalonde S, Tegeder M, Throne-Holst M, Frommer WB, Patrick JW (2003) Phloem loading and unloading of sugars and amino acids. Plant, Cell Environ 26:37–56
Lemoine R, La Camera S, Atanassova R, Dédaldéchamp F, Allario T, Pourtau N, Bonnemain JL, Laloi M, Coutos-Thévenot P, Maurousset L, Faucher M (2013) Source-to-sink transport of sugar and regulation by environmental factors. Front Plant Sci 4:1–21
Lian L, Wang X, Zhu Y, He W, Cai Q, Xie H, Zhang M, Zhang J (2014) Physiological and photosynthetic characteristics of indica Hang2 expressing the sugarcane PEPC gene. Mol Biol Rep 41:2189–2197
Lingle SE (1999) Sugar metabolism during growth and development in sugarcane internodes. Crop Sci 39:480–486
Lingle SE, Smith RC (1991) Sucrose metabolism related to growth and ripening in sugarcane internodes. Crop Sci 31:172–177
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ (1951) Protein measurement with the Folin phenol reagent. J Biol Chem 193:265–275
Marcelis LFM (1996) Sink strength as a determinant of dry matter partitioning in the whole plant. J Exp Bot 47:1281–1291
McCormick AJ, Cramer MD, Watt DA (2006) Sink strength regulates photosynthesis in sugarcane. New Phytol 171:759–770
McCormick AJ, Cramer MD, Watt DA (2008) Culm sucrose accumulation promotes physiological decline of mature leaves in ripening sugarcane. Fields Crops Res 108:250–258
McCormick AJ, Watt DA, Cramer MD (2009) Supply and demand: sink regulation of sugar accumulation in sugarcane. J Exp Bot 60:357–364
Moore PH (1995) Temporal and spatial regulation of sucrose accumulation in the sugarcane stem. Aust J Plant Physiol 22:661–679
Moore PH, Botha FC, Furbank RT (1997) Potential for overcoming physio-biochemical limits to sucrose accumulation. In: Keating BA, Wilson JR (eds) Intensive Sugarcane Production: Meeting the Challenges Beyond 2000. CAB International, Wallinford, UK
Nelson N (1944) A photometric adaptation of Somogyi method for the determination of glucose. J Biol Chem 153:375–380
Nonis A, Ruperti B, Falchi R, Casatta E, Thamasebi Enferadi S, Vizzotto G (2007) Differential expression and regulation of a neutral invertase encoding gene from peach (Prunus persica): evidence for a role in fruit development. Physiol Plant 129:436–446
Paul MJ, Foyer CH (2001) Sink regulation of photosynthesis. J Exp Bot 52:1381–1400
Rae AL, Perroux JM, Grof CPL (2005) Sucrose partitioning between vascular bundles and storage parenchyma in the sugarcane stem: a potential role for the ShSUT1 sucrose transporter. Planta 220:817–825
Roitsch T, Bittne M, Godt DE (1995) Induction of apoplastic invertase of Chenopodium rubrum by d-glucose and a glucose analog and tissue-specific expression suggest a role in sink-source regulation. Plant Physiol 108:285–294
Roitsch T, Balibrea ME, Hofmann M, Proels R, Singh AK (2003) Extracellular invertase: key metabolic enzyme and PR protein. J Exp Bot 54:513–524
Ruan YL, Chourey PS (2006) Carbon partitioning in developing seed. In: Basra AS (ed) Seed sciences and technology: trends and advances. The Haworth Press, New York, pp 125–152
Stitt M, Gerhardt R, Wilke I, Heldt HW (1987) The contribution of fructose 2, 6-bisphosphate to the regulation of sucrose synthesis during photosynthesis. Physiol Plant 69:377–386
Sturm A (1999) Invertases. Primary structures, functions, and roles in plant development and sucrose partitioning. Plant Physiol 121:1–8
Tang GQ, Lüscher M, Sturm A (1999) Antisense repression of vacuolar and cell wall invertase in transgenic carrot alters early plant development and sucrose partitioning. Plant Cell 11:177–189
Tejera NA, Rodés R, Ortega E, Campos R, Lluch C (2007) Comparative analysis of physiological characteristics and yield components in sugarcane cultivars. Field Crops Resh 102:64–72
Van Handel E (1968) Direct micro determination of sucrose. Anal Biochem 22:280–283
Verma I, Roopendra K, Sharma A, Jain R, Singh RK, Chandra A (2017) Expression analysis of genes associated with sucrose accumulation in sugarcane under normal and GA3-induced source–sink perturbed conditions. Acta Physiol Planta 39:133
Watt DA, McCormick AJ, Govender C, Carson DL, Cramer MD, Huckett BI, Botha FC (2005) Increasing the utility of genomics in unraveling sucrose accumulation. Field Crops Res 92:149–158
Welbaum GE, Meinzer FC, Grayson RL, Thornham KT (1992) Evidence for the consequences of a barrier to solute diffusion between the apoplast and vascular bundles in sugarcane stalk tissue. Funct Plant Biol 19:611–623
Zhu YJ, Albert HH, Moore PH (1997) Sucrose accumulation in the sugarcane stem is regulated by the difference between the activities of soluble acid invertase and sucrose phosphate synthase. Plant Physiol 115:609–616
Acknowledgements
Authors are thankful to ICAR-Indian Institute of Sugarcane Research, Lucknow and Integral University, Lucknow for providing necessary facilities and guidance to carry out this work. IV thankful to Integral University for providing manuscript ID (IU/R&D/2018-MCN000393). Authors are thankful to the anonymous reviewer for critical review and improvement.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All the authors declare that they have no conflicts of interest.
Rights and permissions
About this article
Cite this article
Verma, I., Roopendra, K., Sharma, A. et al. Expression analysis of genes associated with sucrose accumulation and its effect on source–sink relationship in high sucrose accumulating early maturing sugarcane variety. Physiol Mol Biol Plants 25, 207–220 (2019). https://doi.org/10.1007/s12298-018-0627-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12298-018-0627-z