Abstract
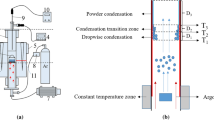
This article describes the influence of temperature profile on the morphology and impurity distributions of magnesium vapor condensation during silicothermic reduction of calcined dolomite. The formation of magnesium crystals was comprehensively analyzed by optical microscopy, scanning electron microscopy (SEM), and inductively coupled plasma (ICP). The results showed that the temperature changed the morphology and purity of condensation products in the condenser. When the temperature of the condenser zone was lower than 250 °C, the magnesium vapor condensation was fine and noncompacted. The crystal growth of magnesium occurred in the range of 250 °C and 380 °C. It was found that crystal magnesium became coarser and denser at higher temperatures. Based on the analysis of crystallized magnesium, the impurity distributions decreased at temperatures higher than those of the condenser. Furthermore, this indicates that the impurity contents with less than 0.04 pct were distributed randomly through the condenser.











Similar content being viewed by others
References
Y. Tian, T. Qu, B. Yang, Y.N. Dai, B.Q. Xu, and S. Geng: Metall. Mater. Trans. B, 2012, vol. 43B, pp. 657–61.
X.J. Wang, D.K. Xu, R.Z. Wu, X.B. Chen, Q.M. Peng, L. Jin, Y.C. Xin, Z.Q. Zhang, Y. Liu, X.H. Chen, G. Chen, K.K. Deng, and H.Y. Wang: J. Mater. Sci. Technol., 2017, in press.
F. Habashi: Handbook of Extractive Metallurgy, Wiley, New York, NY, 1997, pp. 1160–62.
L. Rongti, P. Wei, and M. Sano: Metall. Mater. Trans. B, 2003, vol. 34B, pp. 433–37.
Y. Tian, B.Q. Xu, C.B. Yang, B. Yang, T. Qu, H.X. Liu, Y.N. Dai, and D.C. Liu: Metall. Mater. Trans. B, 2014, vol. 45B, pp. 1936–41.
A. Krishnan, X.G. Lu, and U.B. Pal: Metall. Mater. Trans. B, 2005, vol. 36B, pp. 463–73.
K.N. Solanki, D. Orlov, A. Singh, and N.R. Neelameggham: Magnesium Technology 2017, Springer, Switzerland, 2017, pp. 107–11, 199–202, and 107–11.
R.B. Li, S.J. Zhang, L.J. Guo, and J.J. Wei: Int. J. Heat Mass Transfer, 2013, vol. 59, pp. 328–37.
D. Fu, Y. Wang, J. Peng, Y. Di, S. Tao, and N. Feng: Adv. Chem. Eng. II, 2012, vol. 550–553, pp. 1779–83.
W. Wulandari, A. Rhamdhani, G. Brooks, and B.J. Monaghan: Eur. Metall. Conf. on GDMB Society for Mining, EMC, Austria, 2009, pp. 1401–15.
I.M. Morsi and H.H. Ali: Int. J. Miner. Process., 2014, vol. 127, pp. 37–43.
D.X. Fu, T.A. Zhang, L.K. Guan, Z.H. Dou, and M. Wen: JOM, 2016, vol. 68, pp. 3208–13.
B. Mehrabi, M. Abdellatif, and F. Masoudi: Int. J. Mater. Sci. Eng., 2011, vol. 8, pp. 18–24.
C. Wang, C. Zhang, S.J. Zhang, and L.J. Guo: Int. J. Miner. Process., 2015, vol. 142, pp. 147–53.
Y. Aviezer, L. Birnhack, A. Leon, E. Aghion, and O. Lahav: Hydrometallurgy, 2017, vol. 169, pp. 520–33.
M. Bugdayci, A. Turan, M. Alkan, and O. Yucel: High Temp. Mater. Proc., 2018, vol. 37, pp. 1–8.
H. Halmann, A. Frei, and A. Steinfeld: Ind. Eng. Chem. Res., 2008, vol. 47, pp. 2146–54.
C.B. Yang, Y. Tian, T. Qu, B. Yang, B.Q. Xu, and Y.N. Dai: J. Magn. Alloys, 2014, vol. 2, pp. 50–58.
C.B. Yang, Y. Tian, T. Qu, B. Yang, B.Q. Xu, and Y.N. Dai: Trans. Nonferrous Met. Soc. China, 2014, vol. 24, pp. 561–69.
C.B. Yang, Y. Tian, T. Qu, B. Yang, B.Q. Xu, and Y.N. Dai: J. Magn. Alloys, 2013, vol. 1, pp. 323–29.
A.K. Mahapatra, J. Wang, H. Zhang, and M. Han: Lett. J. Explor. Front. Phys., 2016, vol. 115, pp. 1–6.
M. Cho and J.J. Fawcett: Am. Mineral., 1986, vol. 71, pp. 78–84.
I. Sunagawa: Crystals Growth, Morphology and Perfection, 1st ed., Cambridge University Press, New York, NY, 2005, pp. 45–47.
Author information
Authors and Affiliations
Corresponding author
Additional information
Manuscript submitted August 25, 2017.
Rights and permissions
About this article
Cite this article
Gorji, A., Panjepour, M. & Ahmadian, M. Study of Morphology and Magnesium Purity, Formed by Vapor Phase Through Silicothermic Reduction. Metall Mater Trans B 49, 2435–2443 (2018). https://doi.org/10.1007/s11663-018-1357-x
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11663-018-1357-x