Abstract
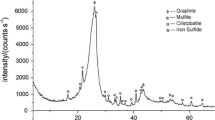
Magnesium production by carbothermic reduction of magnesia with CaF2 in vacuum was investigated experimentally by X-ray diffraction (XRD), scanning electron microscopy (SEM), energy-dispersive X-ray spectroscopy (EDS), and thermodynamic analysis. Thermodynamic calculations indicate that magnesium was generated by a carbothermic reduction among MgO-C system, which should be above 1500 K (1227 °C) (50 Pa). According to the carbothermic reduction analysis, the CaF2 does not participate in the carbothermic process. The experimental results demonstrated that the mass loss increased along with increasing CaF2. The percentage was up to 92 pct with 5 pct CaF2.The reduction degree increased with CaF2 more than it without CaF2 obviously. Considering the reduction degree and economic benefit, 5 pct CaF2 was the optimal choice. The purity of metal magnesium reached 95.59 wt pct, which has perfect crystallization and lamellar structure. CaF2 did not participate in magnesia carbothermic reduction in vacuum; instead, it played a catalytic role during the process.








Similar content being viewed by others
References
R.E. Brown: Light Metal Age, 1996, vol. 8.
W. Xian-Fei and X. Shou-Mei: Trans. Nonferrous Met. Soc. China, 2011, vol. 21, pp. 807–13.
L.I. Rong-Ti, P. Wei, and M. Sano: Metall. Mater. Trans. B, 2003, vol. 8B, pp. 433–37.
Z. Ding-Fei, Q. Fu-Gang, L. Wei, S. Guo-Liang, and Z. Xia-Bing: Trans. Nonferrous Met Soc. China, 2011, vol. 21, pp. 703–09.
B.L. Mordike and T. Ebert: Mater. Sci. Eng. A, 2001, vol. 302, pp. 37–45.
A. Froats: Light Metal, TMS-AIME, New York, NY, 1980, pp. 969–79.
S. Ramakrishnan and P. Koltun: Resour. Conservat. Recycl., 2004, vol. 42, pp. 49–64.
L. Zhi-Hua, X. Huai-Sheng, and D. Yong-Nian: Energy for Metallurgical Industry, 2004, vol. 23, pp. 20–22.
R. Winand: Trans. Inst. Min. Metall. Sect. C: Miner. Process. Extract. Metall., 1990, vol. 99, pp.105–12.
D.J. Min, J.W. Han, and W.S. Chung: Metall. Mater. Trans. B, 1999, vol. 30B, pp. 215–21.
L. Hong, K. Okumura, and M. Sano: Metall. Mater. Trans. B, 1999, vol. 30B, pp. 1003–08.
Y.U. Qing-Chun, Y. Bin, X. Bao-Qiang, L. Da-Chun, L. Zhi-Hua, and D. Yong-Nian: Proc. 9 th Vacuum Metallurgy and Surface Engineering Conference, Shenyang, China, 2009, pp. 428–33.
L. Zhi-Hua, D. Yong-Nian, and X. Huai-Sheng: Youse Jinshu, 2005, vol. 57, no. 1, pp. 56–59.
G. Brooks, S. Trang, and P. Witt: JOM, 2006, vol. 58, no. 5, pp. 51–55.
M.W. Chase: NIST-JANAF Thermochemical Tables[M],4th ed., American Chemical Society and the American Institute of Physics for the National Institute of Standards and Technology, New York, NY, 1998, pp. 98–143.
I. Brain, M. Cheng, Nai-Liang, N. Si-Tong, and X. Gui-Ying: Thermochemical Data of Pure Substances, 3rd ed., Science Press, Beijing, China, 2003.
X. Ri-Yao: Magnesium Production Process, Central South University Press, Changsha, China, 2003, pp. 12–15.
Acknowledgment
This work was supported financially by the Science Research Foundation of Education Department of Yunnan Province, China (No. 2010C257)
Author information
Authors and Affiliations
Corresponding author
Additional information
Manuscript submitted September 21, 2011.
Rights and permissions
About this article
Cite this article
Tian, Y., Qu, T., Yang, B. et al. Behavior Analysis of CaF2 in Magnesia Carbothermic Reduction Process in Vacuum. Metall Mater Trans B 43, 657–661 (2012). https://doi.org/10.1007/s11663-011-9622-2
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11663-011-9622-2