Abstract
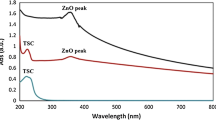
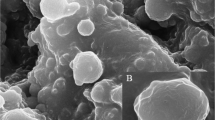
The antimicrobial efficacy of rhamnolipid is well established against a wide range of pathogens. However little is known about the enhancement of antimicrobial efficacy of rhamnolipid in the form of nanoparticles. With a curiosity of enhancing antimicrobial activity, a study has been carried out to evaluate the antimicrobial efficacy of rhamnolipid-coated zinc oxide nanoparticles. The zinc oxide nanoparticles were synthesized with rhamnolipid, produced by Pseudomonas aeruginosa JS29. The rhamnolipid-coated zinc oxide nanoparticles were characterized by FTIR, XRD, TGA, TEM, and SAED. The antimicrobial and antibiofilm efficacy of the nanoparticles was evaluated against Staphylococcus aureus MTCC 96. FTIR, XRD, TEM, and SAED analyses confirmed that the nanoparticles contain both rhamnolipid and zinc as constituents and are polycrystalline with sizes ranging from 40 to 50 nm. At a concentration of 250 µg/ml, rhamnolipid-coated zinc oxide nanoparticles exhibited 80% growth inhibition of the pathogen. Again, at the same concentration, the nanoparticle was observed to inhibit 78% of biofilm formation while disrupting 100% of preformed biofilm. The nanoparticles demonstrated an enhanced inhibitory and antibiofilm efficacy against the pathogen compared to the individual effect of both rhamnolipid and zinc oxide nanoparticles. With the established non-toxicity of rhamnolipid-coated zinc oxide nanoparticles in fibroblast cell lines, the nanoparticles could be a promising pharmaceutical alternative.
Graphic abstract






Similar content being viewed by others
Data availability
The manuscript doesn’t contain any data that are required to be submitted in any data repository.
References
Agarwal H, Kumar SV, Rajeshkumar S (2017) A review on green synthesis of zinc oxide nanoparticles—an eco-friendly approach. Resour Eff Technol 3(4):406–413. https://doi.org/10.1016/j.reffit.2017.03.002
Aleaghil SA, Fattahy E, Baei B, Saghali M, Bagheri H, Javid N, Ghaemi EA (2016) Antibacterial activity of zinc oxide nanoparticles on Staphylococcus aureus. Int J Adv Biotechnol Res 7(3):1569–1575
Barsaiya M, Singh DP (2018) Green synthesis of zinc oxide nanoparticles by Pseudomonas aeruginosa and their broad-spectrum antimicrobial effects. J Pure Appl Microbiol 12(4):2123–2134. https://doi.org/10.22207/JPAM.12.4.50
Bhattacharyya P, Agarwal B, Goswami M, Maiti D, Baruah S, Tribedi P (2018) Zinc oxide nanoparticle inhibits the biofilm formation of Streptococcus pneumoniae. Antonie Van Leeuwenhoek 111(1):89–99. https://doi.org/10.1007/s10482-017-0930-7
Borah SN, Sen S, Goswami L, Bora A, Pakshirajan K, Deka S (2019) Rice based distillers dried grains with solubles as a low cost substrate for the production of a novel rhamnolipid biosurfactant having anti-biofilm activity against Candida tropicalis. Colloids Surf B Biointerfaces 182:110358. https://doi.org/10.1016/j.colsurfb.2019.110358
Brown SD, Traczewski MM (2009) In vitro antimicrobial activity of a new cephalosporin, ceftaroline, and determination of quality control ranges for MIC testing. Antimicrob Agents Chemother 53(3):1271–1274
Cao D, Gong S, Shu X, Zhu D, Liang S (2019) Preparation of ZnO nanoparticles with high dispersibility based on oriented attachment (OA) process. Nanoscale Res Lett 14(1):1–11. https://doi.org/10.1186/s11671-019-3038-3
Das M, Patowary K, Vidya R, Malipeddi H (2016) Microemulsion synthesis of silver nanoparticles using biosurfactant extracted from Pseudomonas aeruginosa MKVIT3 strain and comparison of their antimicrobial and cytotoxic activities. IET nano-biotechnology 10(6):411–418. https://doi.org/10.1049/iet-nbt.2015.0119
de J Cortes-Sanchez, Hernandez-Sanchez A, Jaramillo-Flores H ME (2013) Biological activity of glycolipids produced by microorganisms: new trends and possible therapeutic alternatives. Microbiol Res 168(1):22–32. https://doi.org/10.1016/j.micres.2012.07.002
FDA e-CFR data (2020) Title 21: food and drugs part 182—substances generally recognized as safe, subpart i—nutrients Subpart I → §182.8991: Zinc oxide. https://www.ecfr.gov/cgi-bin/text-idx?SID=4c1314d72ae1950efd9cd6a05db06410&mc=true&node=se21.3.182_18991&rgn=div8
Haque M, Sartelli M, McKimm J, Bakar MA (2018) Health care-associated infections–an overview. Infect Drug Resist 11:2321. https://doi.org/10.2147/IDR.S177247
ISO 10993–5 (2009) Biological evaluation of medical devices. Part 5: tests for in vitro cytotoxicity; International Organization for Standardization, Geneva
Jiang J, Pi J, Cai J (2018) The advancing of zinc oxide nanoparticles for biomedical applications. Bioinorg Chem Appl 2018. https://doi.org/10.1155/2018/1062562
Karanth NGK, Deo PG, Veenanadig NK (1999) Microbial production of biosurfactants and their importance. Curr Sci 77:116–126
Khalid HF, Tehseen B, Sarwar Y, Hussain SZ, Khan WS, Raza ZA, Bajwa SZ, Kanaras AG, Hussain I, Rehman A (2019) Biosurfactant coated silver and iron oxide nanoparticles with enhanced anti-biofilm and anti-adhesive properties. J Hazard Mater 364:441–448. https://doi.org/10.1016/j.jhazmat.2018.10.049
Kiran GS, Selvin J, Manilal A, Sujith S (2011) Biosurfactants as green stabilizers for the biological synthesis of nanoparticles. Crit Rev Biotechnol 31(4):354–364. https://doi.org/10.3109/07388551.2010.539971
Kumar CG, Mamidyala SK (2011) Extracellular synthesis of silver nanoparticles using culture supernatant of Pseudomonas aeruginosa. Colloids Surf B Biointerfaces 84(2):462–466. https://doi.org/10.1016/j.colsurfb.2011.01.042
Lahkar J, Borah SN, Deka S, Ahmed G (2015) Biosurfactant of Pseudomonas aeruginosa JS29 against Alternaria solani: the causal organism of early blight of tomato. Biocontrol 60(3):401–411. https://doi.org/10.1007/s10526-015-9650-y
Lopez Del Egido L, Navarro-Miró D, Martinez-Heredia V, Toorop PE, Iannetta PP (2017) A spectrophotometric assay for robust viability testing of seed batches using 2, 3, 5-triphenyl tetrazolium chloride: using Hordeum vulgare L. as a model. Frontiers in plant science 8:747. https://doi.org/10.3389/fpls.2017.00747
Luna JM, Rufino RD, Sarubbo LA, Rodrigues LR, Teixeira JA, de Campos-Takaki GM (2011) Evaluation antimicrobial and antiadhesive properties of the biosurfactant Lunasan produced by Candida sphaerica UCP 0995. Curr Microbiol 62(5):1527–1534. https://doi.org/10.1007/s00284-011-9889-1
Magalhães L, Nitschke M (2013) Antimicrobial activity of rhamnolipids against Listeria monocytogenes and their synergistic interaction with nisin. Food Control 29(1):138–142. https://doi.org/10.1016/j.foodcont.2012.06.009
McNamara K, Tofail SA (2017) Nanoparticles in biomedical applications. Adv Phys 2(1):54–88. https://doi.org/10.1080/23746149.2016.1254570
Mishra PK, Mishra H, Ekielski A, Talegaonkar S, Vaidya B (2017) Zinc oxide nanoparticles: a promising nanomaterial for biomedical applications. Drug Discov Today 22(12):1825–1834. https://doi.org/10.1016/j.drudis.2017.08.006
Nanda A, Saravanan M (2009) Biosynthesis of silver nanoparticles from Staphylococcus aureus and its antimicrobial activity against MRSA and MRSE. Nanomedicine 5(4):452–456. https://doi.org/10.1016/j.nano.2009.01.012
Narayanan J, Ramji R, Sahu H, Gautam P (2010) Synthesis, stabilisation and characterisation of rhamnolipid-capped ZnS nanoparticles in aqueous medium. IET Nano-Biotechnol 4(2):29–34. https://doi.org/10.1049/iet-nbt.2009.0010
Ndlovu T, Rautenbach M, Vosloo JA, Khan S, Khan W (2017) Characterisation and antimicrobial activity of biosurfactant extracts produced by Bacillus amyloliquefaciens and Pseudomonas aeruginosa isolated from a wastewater treatment plant. AMB Express 7(1):1–19. https://doi.org/10.1186/s13568-017-0363-8
Park SY, Kim CG (2018) A comparative study of three different viability tests for chemically or thermally inactivated Escherichia coli. Environ Eng Res 23(3):282–287. https://doi.org/10.4491/eer.2017.223
Patowary R, Patowary K, Kalita MC, Deka S (2016) Utilization of paneer whey waste for cost-effective production of rhamnolipid biosurfactant. Appl Biochem Biotechnol 180(3):383–399. https://doi.org/10.1007/s12010-016-2105-9
Patton T, Barrett J, Brennan J, Moran N (2006) Use of a spectrophotometric bioassay for determination of microbial sensitivity to manuka honey. J Microbiol Methods 64(1):84–95. https://doi.org/10.1016/j.mimet.2005.04.007
Płaza GA, Chojniak J, Banat IM (2014) Biosurfactant mediated biosynthesis of selected metallic nanoparticles. Int J Mol Sci 15(8):13720–13737. https://doi.org/10.3390/ijms150813720
Satpute SK, Mone NS, Das P, Banat IM, Banpurkar AG (2019) Inhibition of pathogenic bacterial biofilms on PDMS based implants by L. acidophilus derived biosurfactant. BMC Microbial 19(1):1–15. https://doi.org/10.1186/s12866-019-1412-z
Singh BN, Rawat AKS, Khan W, Naqvi AH, Singh BR (2014) Biosynthesis of stable antioxidant ZnO nanoparticles by Pseudomonas aeruginosa rhamnolipids. PLoS ONE 9(9):106937. https://doi.org/10.1371/journal.pone.0106937
Soemo AR (2013) Microenvironment of monorhamnolipid biosurfactant aggregates and monorhamnolipid effects on aqueous dispersion properties of metal oxide nanoparticles. Doctoral dissertation, The University of Arizona. https://repository.arizona.edu/handle/10150/293563
Swain PS, Rao SB, Rajendran D, Dominic G, Selvaraju S (2016) Nano zinc, an alternative to conventional zinc as animal feed supplement: a review. Anim Nutr 2(3):134–141. https://doi.org/10.1016/j.aninu.2016.06.003
Tong SY, Davis JS, Eichenberger E, Holland TL, Fowler VG (2015) Staphylococcus aureus infections: epidemiology, pathophysiology, clinical manifestations, and management. Clin Microbiol Rev 28(3):603–661. https://doi.org/10.1128/CMR.00134-14
Tran N, Mir A, Mallik D, Sinha A, Nayar S, Webster TJ (2010) Bactericidal effect of iron oxide nanoparticles on Staphylococcus aureus. Int J Nanomed 5:277. https://doi.org/10.2147/ijn.s9220
Tuli HS, Kashyap D, Bedi SK, Kumar P, Kumar G, Sandhu SS (2015) Molecular aspects of metal oxide nanoparticle (MO-NPs) mediated pharmacological effects. Life Sci 143:71–79. https://doi.org/10.1016/j.lfs.2015.10.021
Vallee BL, Falchuk KH (1993) The biochemical basis of zinc physiology. Physiol Rev 73(1):79–118. https://doi.org/10.1152/physrev.1993.73.1.79
World Health Organization (2015) Health care-associated infections fact sheet [Internet]. Geneva. https://www.who.int/gpsc/country_work/gpsc_ccisc_fact_sheet_en.pdf. Accessed 26 Feb 2021
Yusof HM, Mohamad R, Zaidan UH (2019) Microbial synthesis of zinc oxide nanoparticles and their potential application as an antimicrobial agent and a feed supplement in animal industry: a review. J Anim Sci Biotechnol 10(1):1–22. https://doi.org/10.1186/s40104-019-0368-z
Zak AK, Razali R, Abd Majid WH, Darroudi M (2011) Synthesis and characterization of a narrow size distribution of zinc oxide nanoparticles. Int J Nanomed 6:1399. https://doi.org/10.2147/ijn.s19693
Zhang L, Gu FX, Chan JM, Wang AZ, Langer RS, Farokhzad OC (2008) Nanoparticles in medicine: therapeutic applications and developments. Clin Pharm Therap 83(5):761–769. https://doi.org/10.1038/sj.clpt.6100400
Acknowledgements
The authors are thankful to the Director, Institute of Advanced Study in Science and Technology (IASST), Guwahati, India for providing laboratory facilities for the research. Chandana Malakar is thankful to Gauhati University for the registration of Ph.D. The authors are also thankful to Dr. Mojibur Rahman Khan, Associate Professor II, Life Science Division, IASST for providing a cell culture facility to carry out the cell line studies. Chandana Malakar would also like to acknowledge Madhurankhi Goswami, the Senior Research Fellow, Life Science Division, IASST for her help in improving the writing of the manuscript.
Funding
This work is supported by the ‘Department of Biotechnology (DBT), Govt. of India’ to Chandana Malakar as a Junior Research Fellow for carrying out Ph.D. research under the supervision of Prof. Suresh Deka vide letter No. DBT/JRF/BET-17/I/2017/AL/351 dated 01/06/2017.
Author information
Authors and Affiliations
Contributions
CM: Methodology, validation, investigation, formal analysis, writing—original draft, funding acquisition. KP: Writing—review & editing, validation, methodology. SD: Conceptualization, methodology, resources, writing—review & editing, supervision. MCK: Conceptualization, writing—review & editing.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Ethical approval
The study does not contain human or animal study.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Malakar, C., Patowary, K., Deka, S. et al. Synthesis, characterization, and evaluation of antibacterial efficacy of rhamnolipid-coated zinc oxide nanoparticles against Staphylococcus aureus. World J Microbiol Biotechnol 37, 193 (2021). https://doi.org/10.1007/s11274-021-03160-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11274-021-03160-w