Abstract
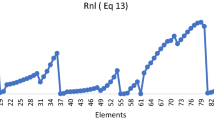
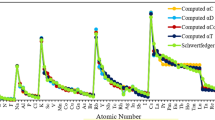
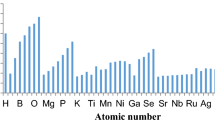
Atomic radius is an important periodic descriptor used in understanding a variety of physico-chemical and bio-chemical processes. Numerous scales are suggested to define atomic radii. The aim of the current study is to find out the most reliable and universal scale among different (experimental and theoretical) scales of radii. For this, we have used different types of radii to compute some size-dependent physico-chemical atomic descriptors, i.e. electronegativity, global hardness, polarizability, and a real-world molecular descriptor, i.e. internuclear bond distance for some diatomic molecules. The computed properties are compared with available experimental values. Important periodic trends and the presence of relativistic effects are also verified for each set of atomic radii. This comparative study is valuable to get an idea about the most effective atomic radii.
Similar content being viewed by others
Data availability
All data generated or analysed during this study are included in this published article and its supplementary information file.
References
Cotton FA, Wilkinson G, Murillo CA, Bochmann M (2009) Advanced inorganic chemistry. Wiley-New York
Rahm M, Hoffmann R, Ashcroft NW (2016) Atomic and ionic radii of elements 1–96. Chem 22:14625–14632
Chakraborty T, Gazi K, Ghosh DC (2010) Computation of the atomic radii through the conjoint action of the effective nuclear charge and the ionization energy. Mol Phys 108:2081–2092
Alvarez S (2013) A cartography of the van der Waals territories. Dalton Trans 42:8617–8636
Meyer L (1870) Justus Liebigs Ann Chem 354
Bragg WL (1920) The arrangement of atoms in crystals. The London, Edinburgh, and Dublin Philosophical Magazine and Journal of Science 40:169–189
Pauling LC (1960) The nature of the chemical bond and the structure of molecules and crystals. An introduction to modern structural chemistry, 3rd ed., Cornell University Press, Ithaca, N.Y.
Slater JC (1930) Atomic shielding constants. Phys Rev 36:57
Froese C (1966) Hartree - Fock parameters for the atoms helium to radon. J Chem Phys 45:1417–1420
Clementi E, Raimondi DL, Reinhardt WP (1967) Atomic screening constants from SCF functions. II. Atoms with 37 to 86 electrons. J Chem Phys 47:1300–1307
Fisk C, Fraga S (1969) Atomic Radii Anal Fis 65:135
Larson AC, Waber JT (1969) Self-consistent field Hartree calculations for atoms and ions (Report LA-4297). Los Alamos Scientific Lab, N. Mex
Fischer CF (1972) Average-energy-of-configuration Hartree-Fock results for the atoms helium to radon. Atomic Data 4:301–399
Kammeyer CW, Whitman DR (1972) Quantum mechanical calculation of molecular radii. I. Hydrides of elements of periodic groups IV through VII. J Chem Phys 56:4419–4421
Fraga S, Karwowski J, Saxena KMS (1973) Hartree-Fock values of coupling constants, polarizabilities, susceptibilities, and radii for the neutral atoms, helium to nobelium. Atom Data Nucl Data Tables 12:467–477
Fischer CF (1973) Average–energy of configuration Hartree-Fock results for the atoms helium to radon. Atom Data Nucl Data Tables 12:87–99
Desclaux JP (1973) Relativistic Dirac-Fock expectation values for atoms with Z= 1 to Z= 120. Atom Data Nucl Data Tables 12:311–406
Boyd RJ (1977) The relative sizes of atoms. J Phys B: At Mol Phys 10:2283
Deb BM, Singh R, Sukumar N (1992) A universal density criterion for correlating the radii and other properties of atoms and ions. J Mol Struct: THEOCHEM 259:121–139
Nath S, Bhattacharya S, Chattaraj PK (1995) Density functional calculation of a characteristic atomic radius. J Mol Struct: THEOCHEM 331:267–279
Ghosh DC, Biswas R (2002) Theoretical calculation of absolute radii of atoms and ions. Part 1. The atomic radii. Int J Mol Sci 3:87–113
Putz MV, Russo N, Sicilia E (2003) Atomic radii scale and related size properties from density functional electronegativity formulation. J Phys Chem A 107:5461–5465
Pyykkö P, Riedel S, Patzschke M (2005) Triple–bond covalent radii. Chem A Euro J 11:3511–3520
Ghosh DC, Biswas R, Chakraborty T, Islam N, Rajak SK (2008) The wave mechanical evaluation of the absolute radii of atoms. J Mol Struct: THEOCHEM 865:60–67
Pyykkö P, Atsumi M (2009) Molecular single–bond covalent radii for elements 1–118. Chem A Euro J 15:186–197
Mande C, Deshmukh P (1977) A new scale of electronegativity on the basis of calculations of effective nuclear charges from X-ray spectroscopic data. J Phys B: At Mol Phys 10:2293
Mande C, Chattopadhyay S, Deshmukh PC, Padma R, Deshmukh PC (1990) Spectroscopically determined electronegativity values for heavy elements. Pramana 35:397–403
Miller IJ (1987) The quantisation of the screening constant. Austr J Phys 40:329–346
Reed JL (1999) The genius of Slater’s rules. J Chem edu 76:802
Szarek P, Grochala W (2014) Most probable distance between the nucleus and HOMO electron: the latent meaning of atomic radius from the product of chemical hardness and polarizability. J Phys Chem A 118:10281–10287
Tandon H, Ranjan P, Chakraborty T, Suhag V (2020) Computation of absolute radii of 103 elements of the periodic table in terms of nucleophilicity index. J Math Chem 58:1025–1040
Tandon H, Chakraborty T, Suhag V (2021) A scale of absolute radii derived from electrophilicity index. Mol Phys 119:e1820594
Prasanna KG, Sunil S, Kumar A, Joseph J (2021) Theoretical atomic radii of elements (H-Cm): a non-relativistic study with Gaussian basis set using HF, post-HF and DFT methods. ChemRxiv. Preprint. https://doi.org/10.26434/chemrxiv.13663337.v1
Coulson CA (1951) Critical survey of the method of ionic-homopolar resonance. Proceedings of the Royal Society of London. Series A. Math Phys Sci 207: 63–73
Fukui K (1982) Role of frontier orbitals in chemical reactions. Sci 218:747–754
Pauling L (1932) The nature of the chemical bond. IV. The energy of single bonds and the relative electronegativity of atoms. J Am Chem Soc 54:3570–3582
Mulliken RS (1934) A new electroaffinity scale; together with data on valence states and on valence ionization potentials and electron affinities. J Chem Phys 2:782–793
Gordy W (1946) A new method of determining electronegativity from other atomic properties. Phys Rev 69:604
Allred AL, Rochow EG (1958) A scale of electronegativity based on electrostatic force. J Inorg Nucl Chem 5:264–268
Simons G, Zandler ME, Talaty ER (1976) Nonempirical electronegativity scale. J Am Chem Soc 98:7869–7870
Nagle JK (1990) Atomic polarizability and electronegativity. J Am Chem Soc 112:4741–4747
Ghosh DC, Chakraborty T (2009) Gordy’s electrostatic scale of electronegativity revisited. J Mol Struct: THEOCHEM 906:87–93
Tandon H, Labarca M, Chakraborty T (2021) A scale of atomic electronegativity based on floating spherical gaussian orbital approach. ChemistrySelect 6:5622–5627
Allen LC (1989) Electronegativity is the average one-electron energy of the valence-shell electrons in ground-state free atoms. J Am Chem Soc 111:9003–9014
Islam N, Ghosh DC (2011) Spectroscopic evaluation of the global hardness of the atoms. Mol Phys 109:1533–1544
Pearson RG (1997) Chemical hardness. Wiley-VCH, Weinheim
Mulliken RS (1952) Molecular compounds and their spectra. II J Am Chem Soc 74:811–824
Cárdenas C, Heidar-Zadeh F, Ayers PW (2016) Benchmark values of chemical potential and chemical hardness for atoms and atomic ions (including unstable anions) from the energies of isoelectronic series. Phys Chem Chem Phys 18:25721–25734
Pearson RG (1988) Absolute electronegativity and hardness: application to inorganic chemistry. Inorg Chem 27:734–740
Robles J, Bartolotti LJ (1984) Electronegativities, electron affinities, ionization potentials, and hardnesses of the elements within spin polarized density functional theory. J Am Chem Soc 106:3723–3727
Ghosh DC, Islam N (2010) Semiempirical evaluation of the global hardness of the atoms of 103 elements of the periodic table using the most probable radii as their size descriptors. Int J Quantum Chem 110:1206–1213
Kaya S, Kaya C (2015) A new equation for calculation of chemical hardness of groups and molecules. Mol Phys 113:1311–1319
Yadav P, Tandon H, Malik B, Chakraborty T (2021) An alternative approach to compute atomic hardness. Theor Chem Acc 140:60
Dalgarno A (1962) Atomic polarizabilities and shielding factors. Adv Phys 11:281–315
Bonin KD, Kresin VV (1997) Electric-dipole polarizabilities of atoms, molecules, and clusters. World Scientific, Singapore
Teachout RR, Pack RT (1971) The static dipole polarizabilities of all the neutral atoms in their ground states. Atom Data Nucl Data Tables 3:195–214
Bonin KD, Kadar-Kallen MA (1994) Linear electric-dipole polarizabilities. Int J Modern Phys B 8:3313–3370
Chattaraj PK, Maiti B (2001) Electronic structure principles and atomic shell structure. J Chem Edu 78:811
Politzer P, Murray JS, Bulat FA (2010) Average local ionization energy: a review. J Mol Model 16:1731–1742
Safronova MS, Mitroy J, Clark CW, Kozlov MG (2015) Atomic polarizabilities AIP Conf Proc 1642:81–89
Schwerdtfeger P, Nagle JK (2019) Table of static dipole polarizabilities of the neutral elements in the periodic table. Mol Phys 117:1200–1225
Tandon H, Chakraborty T, Suhag V (2019) A new scale of atomic static dipole polarizability invoking other periodic descriptors. J Math Chem 57:2142–2153
Chakraborty T, Ghosh DC (2010) Computation of the internuclear distances of some heteronuclear diatomic molecules in terms of the revised electronegativity scale of Gordy. Eur Phys J D 59:183–192
Ray NK, Samuelsc L, Parr RG (1979) Studies of electronegativity equalization. J Chem Phys 70:3680–3684
Sanderson RT (1951) An interpretation of bond lengths and a classification of bonds. Science 114:670–672
Sanderson RT (1952a) An interpretation of bond lengths in alkali halide gas molecules. J Am Chem Soc 74:272–274
Sanderson RT (1952b) Electronegativities in inorganic chemistry. J Chem Edu 29:539
Sanderson RT (1955) Partial charges on atoms in organic compounds. Science 121:207–208
Pasternak A (1977) Electronegativity based on the simple bond charge model. Chem Phys 26:101–112
Parr RG, Borkman RF (1967) Chemical binding and potential-energy functions for molecules. J Chem Phys 46:3683–3685
Borkman RF, Parr RG (1968) Toward an understanding of potential-energy functions for diatomic molecules. J Chem Phys 48:1116–1126
Parr RG, Borkman RF (1968) Simple bond-charge model for potential-energy curves of homonuclear diatomic molecules. J Chem Phys 49:1055–1058
Borkman RF, Simons G, Parr RG (1969) Simple bond-charge model for potential-energy curves of heteronuclear diatomic molecules. J Chem Phys 50:58–65
Lovas FJ, Tiemann E (1974) Microwave spectral tables 1. Diatomic Molecules. J Phys Chem Ref Data 3:609–770
Pyykkö P (2012) Relativistic effects in chemistry: more common than you thought. Annu Rev Phys Chem 63:45–64
Balasubramanian K (1997a) Relativistic effects in chemistry: part A theory and techniques. John Wiley & Sons, New York
Balasubramanian K (1997b) Relativistic effects in chemistry: part B: applications. Wiley, New York
Emsley J (1991) The elements. Clarendon Press, Oxford
Acknowledgements
Dr. Tanmoy Chakraborty is thankful to Sharda University, and Dr. Hiteshi Tandon and Ms. PoonamYadav are thankful to Manipal University Jaipur for providing a research facility.
Author information
Authors and Affiliations
Contributions
Conceptualization: Tanmoy Chakraborty; data curation: Hiteshi Tandon; methodology: Tanmoy Chakraborty; formal analysis: Poonam Yadav; investigation: Poonam Yadav; visualization: Hiteshi Tandon, Poonam Yadav; writing — original draft: Poonam Yadav; writing — review and editing: Hiteshi Tandon, Tanmoy Chakraborty; resources: Hiteshi Tandon; supervision: Tanmoy Chakraborty, Babita Malik, Vandana Suhag.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Yadav, P., Tandon, H., Malik, B. et al. A quest for the universal atomic radii. Struct Chem 33, 389–394 (2022). https://doi.org/10.1007/s11224-021-01850-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11224-021-01850-7