Abstract
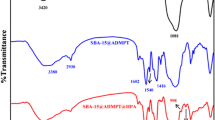
The hexagonal mesoporous organic–inorganic hybrid as a new nanocatalyst was prepared by the treatment of SBA-15 with (3-chloropropyl)triethoxysilane, the 2,4,6-triamino pyrimidine ligand, and then PdCl2 to obtain the SBA-15-propyl-triamino pyrimidine@Pd called as SBA-Pr-3AP@Pd, which was examined through Suzuki–Miyaura cross-coupling reaction by several aryl halides and phenylboronic acid under mild conditions in high yield.













Similar content being viewed by others
Change history
26 December 2022
This article has been retracted. Please see the Retraction Notice for more detail: https://doi.org/10.1007/s11164-022-04937-w
References
C. Deraedt, L. Salmon, D. Astruc, Adv. Synth. Catal. 356, 2525 (2014)
S. Rohani, G. Mohammadi Ziarani, A. Badiei, A. Ziarati, M. Jafari, A. Shayesteh, Appl. Organomet. Chem. 32, e4397 (2018)
S. Lee, C. Jo, R. Ryoo, J. Mater. Chem. A Mater. 5, 11086 (2017)
G. Mohammadi Ziarani, N. Lashgari, A. Badiei, Curr. Org. Chem. 21, 674 (2017)
S.H. Petrosko, R. Johnson, H. White, C.A. Mirkin, J. Am. Chem. Soc. 138, 7443 (2016)
M. Karimi, A. Badiei, G. Mohammadi Ziarani, RSC Adv. 5, 36530 (2015)
G. Mohammadi Ziarani, P. Gholamzadeh, A. Badiei, V. Fathi Vavsari, Res. Chem. Intermed. 44, 277 (2018)
B. Jiang, Y. Guo, J. Kim, A.E. Whitten, K. Wood, K. Kani, A.E. Rowan, J. Henzie, Y. Yamauchi, J. Am. Chem. Soc. 140, 12434 (2018)
T. Zhou, C.-L. Ji, X. Hong, M. Szostak, Chem. Sci. 10, 9865 (2019)
J. Buchspies, M. Szostak, Catalysts 9, 53 (2019)
S. Shi, S.P. Nolan, M. Szostak, Acc. Chem. Res. 51, 2589 (2018)
C. Li, M. Iqbal, B. Jiang, Z. Wang, J. Kim, A.K. Nanjundan, A.E. Whitten, K. Wood, Y. Yamauchi, Chem. Sci. 10, 4054 (2019)
C. Li, H. Tan, J. Lin, X. Luo, S. Wang, J. You, Y.-M. Kang, Y. Bando, Y. Yamauchi, J. Kim, Nano Today 21, 91 (2018)
D. Astruc, F. Lu, J.R. Aranzaes, Angew. Chem. 44, 7852 (2005)
L.A. Bivona, F. Giacalone, L. Vaccaro, C. Aprile, M. Gruttadauria, Chem. Cat. Chem. 7, 2526 (2015)
C. Yang, H. Wustefeld, M. Kalwei, F. Schiith, Studies in Surface Science and Catalysis (Elsevier, Amsterdam, 2004), p. 2574
G. Mohammadi Ziarani, S. Rohani, A. Ziarati, A. Badiei, RSC Adv. 8, 41048 (2018)
D. Zhao, P. Yang, N. Melosh, J. Feng, B.F. Chmelka, G.D. Stucky, Adv. Mater. 10, 1380 (1998)
S. Rostamnia, H. Xin, Appl. Organomet. Chem. 27, 348 (2013)
N. Miyaura, A. Suzuki, Chem. Rev. 95, 2457 (1995)
B. Abbas Khakiani, K. Pourshamsian, H. Veisi, Appl. Organomet. Chem. 29, 259 (2015)
H. Veisi, M. Pirhayati, A. Kakanejadifard, Tetrahedron Lett. 58, 4269 (2017)
N. Aayasha, S.M. Prachi, D. Negi, Int. J. Sci. Basic Appl. Res. 9, 491 (2019)
J. Yu, A. Shen, Y. Cao, G. Lu, Catalysts 6, 181 (2016)
S.M. Sarkar, M.L. Rahman, M.M. Yusoff, New J. Chem. 39, 3564 (2015)
R. Ghorbani-Vaghei, S. Hemmati, H. Veisi, J. Mol. Catal. A. Chem. 393, 240 (2014)
B. Cornils, W.A. Herrmann, M. Beller, R. Paciello, Applied Homogeneous Catalysis with Organometallic Compounds: A Comprehensive Handbook in Four Volumes (Wiley, Hoboken, 2017)
A. Suzuki, J. Organomet. Chem. 576, 147 (1999)
R. Martin, S.L. Buchwald, Acc. Chem. Res. 41, 1461 (2008)
S. MacQuarrie, B. Nohair, J.H. Horton, S. Kaliaguine, C. Crudden, J. Phys. Chem. C 114, 57 (2009)
D.H. Lee, M. Choi, B.W. Yu, R. Ryoo, A. Taher, S. Hossain, M. Jin, Adv. Synth. Catal. 351, 2912 (2009)
Y. Jiang, Q. Gao, J. Am. Chem. Soc. 128, 716 (2006)
A. Corma, Chem. Rev. 97, 2373 (1997)
D. Zhao, J. Feng, Q. Huo, N. Melosh, G.H. Fredrickson, B.F. Chmelka, G.D. Stucky, Science 279, 548 (1998)
S. Rostamnia, E. Doustkhah, RSC Adv. 4, 28238 (2014)
L.K. Mannepalli, C. Gadipelly, G. Deshmukh, P. Likhar, S. Pottabathula, Bull. Chem. Soc. Jpn. 93, 355 (2020)
K. Mukhopadhyay, B.R. Sarkar, R.V. Chaudhari, J. Am. Chem. Soc. 124, 9692 (2002)
G. Mohammadi Ziarani, N. Hosseini Mohtasham, N. Lashgari, A. Badiei, Res. Chem. Intermed. 41, 7581 (2015)
V. Fathi Vavsari, G. Mohammadi Ziarani, S. Balalaie, A. Badiei, F. Golmohammadi, S. Ramezanpour, F. Rominger, ChemistrySelect 2, 3496 (2017)
E. Doustkhah, S. Rostamnia, H.G. Hossieni, R. Luque, ChemistrySelect 2, 329 (2017)
M. Niakan, Z. Asadi, M. Masteri-Farahani, ChemistrySelect 4, 1766 (2019)
K. Matos, J.A. Soderquist, J. Org. Chem. 63, 461 (1998)
D.E. Bergbreiter, P.L. Osburn, J.D. Frels, Adv. Synth. Catal. 347, 172 (2005)
L. Wu, Y. Long, J. Ma, G. Lu, Res. Chem. Intermed. 45, 3809 (2019)
J. Zhang, J. Chen, Q. Zhang, R. Wang, S. Wu, Res. Chem. Intermed. 45, 2503 (2019)
H. Ünver, Res. Chem. Intermed. 44, 7835 (2018)
E. Rafiee, M. Kahrizi, M. Joshaghani, P.G.S. Abadi, Res. Chem. Intermed. 42, 5573 (2016)
Acknowledgements
We are grateful for the Research Council support of Alzahra University.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflicts of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article has been retracted. Please see the retraction notice for more detail: https://doi.org/10.1007/s11164-022-04937-w"
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Mohajer, F., Mohammadi Ziarani, G. & Badiei, A. RETRACTED ARTICLE: The synthesis of SBA-Pr-3AP@Pd and its application as a highly dynamic, eco-friendly heterogeneous catalyst for Suzuki–Miyaura cross-coupling reaction. Res Chem Intermed 46, 4909–4922 (2020). https://doi.org/10.1007/s11164-020-04218-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11164-020-04218-4