Abstract
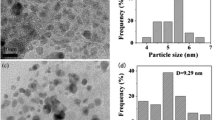
Core-crosslinked shelled-core microspheres of poly(styrene-co-methyl acrylic acid) (PS-co-PMAA), with cores rich in PS and the shell rich in PMAA, were synthesized by one-stage soap-free emulsion polymerization. A palladium (Pd)-iminodiacetic acid (IDA) complex catalyst is loaded on the shell of the PS-co-PMAA microsphere, which results in the advantage of high dispersion degree and, therefore, high activity. The resultant polymeric microspheres catalyst systems are then applied to catalyze the Suzuki reaction of aryl halides with phenylboronic acid in an ionic liquid of 1-butyl-3-methylimidazolium tetrafluoroborate ([bmim][BF4]). Our catalyst systems are proved to be efficient and active for both aryl bromides and aryl iodides. Compared to traditional Pd(Ph3)4 catalyst, the PS-co-PMAA-IDA-Pd catalyst used here affords higher yield of Suzuki reaction at even lower catalyst concentration. In addition, our polymeric-microsphere based catalytic system can be easily recycled at least four times with high activity in ionic [bmim][BF4] liquid.




Similar content being viewed by others
References
N. Miyaura, T. Yanagigand, A. Suzuki, Synth. Commun. 11, 513 (1981)
A. Suzuki, J. Organomet. Chem. 576, 147 (1999)
J. Hassan, M. Sevignon, C. Gozzi, E. Schulz, M. Lemaire, Chem. Rev. 102, 1359 (2002)
L. Yin, J. Liebscher, Chem. Rev. 107, 133 (2007)
Z.C. Xiong, N.D. Wang, M. Dai, A. Li, J.H. Chen, Z. Yang, Org. Lett. 6, 3337 (2004)
L.L. Zhang, Y.L. Guo, A. Iqbal, B. Li, M. Deng, D.Y. Gong, W.S. Liu, W.W. Qin, J. Nanopart. Res. 19, 150 (2017)
R.B. Bedford, S.J.C. Cazin, S.L. Hazelwood, Angew. Chem. Int. Ed. 114, 4294 (2002)
J.P. Stambuli, R. Kuwano, J.F. Hartwig, Angew. Chem. Int. Ed. 41, 4746 (2002)
R.B. DeVasher, L.R. Moore, K.H. Shaughnessy, J. Org. Chem. 69, 7919 (2004)
J.H. Kirchhoff, C. Dai, G.C. Fu, Angew. Chem. Int. Ed. 114, 2025 (2002)
K.N. Sharma, N. Satrawala, R.K. Joshi, Eur. J. Inorg. Chem. 16, 1743 (2018)
M. Ibrahim, I. Malik, W. Mansour, M. Sharif, M. Fettouhi, B.E. Ali, J. Organomet. Chem. 859, 44 (2018)
O. Navarro, R.A. Kelly, S.P. Nolan, J. Am. Chem. Soc. 125, 16194 (2003)
O. Bárta, I. Císařová, P. Štěpnička, Eur. J. Inorg. Chem. 2, 489 (2016)
R. Maity, A. Verma, M. van der Meer, S. Hohloch, B. Sarkar, Eur. J. Inorg. Chem. 1, 111 (2016)
H.Y. Liu, X.S. Li, F. Liu, Y. Tan, Y.Y. Jiang, J. Organomet. Chem. 794, 27 (2015)
A. Avila-Sorrosa, H.A. Jiménez-Vázquez, A. Reyes-Arellanoa, J.R. Pioquinto-Mendoza, R.A. Toscano, L. González-Sebastiánb, D. Morales-Morales, J. Organomet. Chem. 819, 69 (2016)
M. Khajehzadeh, M. Moghadam, J. Organomet. Chem. 863, 60 (2018)
H.H. Zhang, J. Han, F. Tian, Q.Z. Chen, C.Z. Wang, H. Jin, G.Y. Bai, Res. Chem. Intermed. 41, 6731 (2014)
S. Borah, S. Mishra, L. Cardenas, G. Nayanmoni, J. Inorg. Chem. 6, 751 (2018)
Y.L. Huang, Q. Wei, Y.Y. Wang, L.Y. Dai, Carbon 136, 150 (2018)
S. RoyKula, K.K. Senapati, P. Phukan, Res. Chem. Intermed. 41, 5753 (2014)
C. Biglione, A.L. Cappelletti, M.C. Strumia, S.E. MartínPaula, M. Uberman, J. Nanopart. Res. 20, 127 (2018)
J. Chen, J. Zhang, D.J. Zhu, T. Li, J. Porous Mater. 24, 847 (2017)
X. Liu, X.H. Zhao, M. Lu, J. Organomet. Chem. 768, 23 (2014)
B.H. Zhang, Y.G. Xue, A.N. Jiang, Z.M. Xue, Z.H. Li, J.C. Hao, A.C.S. Appl, Mater. Interfaces 9, 7217 (2017)
Y.C. Hu, N. Li, G.Y. Li, A.Q. Wang, Y. Cong, X.D. Wang, T. Zhang, ACS Catal. 7, 2576 (2017)
T. Itoh, Chem. Rev. 117, 10567 (2017)
A. Mondal, A. Das, B. Adhikary, D.K. Mukherjee, J. Nanopart. Res. 16, 2366 (2014)
P.W. Zheng, W.Q. Zhang, J. Catal. 250, 324 (2007)
X.W. Jiang, G.W. Wei, X. Zhang, W.Q. Zhang, P.W. Zheng, F. Wen, L.Q. Shi, J. Mol. Catal. A: Chem. 277, 102 (2007)
X.W. Jiang, Y. Wang, W.Q. Zhang, P.W. Zheng, L.Q. Shi, Macromol. Rapid Commun. 27, 1833 (2006)
S.T. Handy, X. Zhang, Org. Lett. 3, 233 (2001)
J.Z. Zhang, W.Q. Zhang, Y. Wang, M.C. Zhang, Adv. Synth. Catal. 350, 2065 (2008)
C.J. Mathews, P.J. Smith, T. Welton, Chem. Commum. 1249 (2000)
M. Pesavento, R. Biesuz, M. Gallorini, A. Profumo, Anal. Chem. 65, 2522 (1993)
C. Pan, M. Liu, L. Zhang, H. Wu, J. Ding, J. Cheng, Catal. Commun. 9, 508 (2008)
A. Fihri, M. Bouhrara, B. Nekoueishahraki, J.-M. Basset, V. Polshottiwar, Chem. Soc. Rev. 40, 5181 (2011)
Acknowledgements
This work was supported by the National Natural Science Foundation of China (Grant Nos. 21706002), Natural Science Foundation of Anhui Province (1808085QB53), and the Research Fund of School of Chemistry and Chemical Engineering (Anhui University).
Author information
Authors and Affiliations
Corresponding authors
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Zhang, J., Chen, J., Zhang, Q. et al. Polymeric microsphere-loaded palladium-iminodiacetic acid complex as an efficient and easily recycled catalyst for Suzuki reaction in ionic liquid. Res Chem Intermed 45, 2503–2514 (2019). https://doi.org/10.1007/s11164-019-03738-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11164-019-03738-y