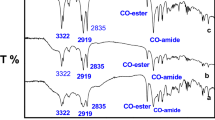
Abstract
Two-membered cationic surfactants namely: N,N-bis(hydroxymethyl)-N-[(2-mercaptoacetoxy)methyl)dodecane ammonium bromide (C12Q) and N,N-bis(hydroxymethyl)-N-[(2-mercaptoacetoxy)methyl]hexadecane ammonium bromide (C16Q) and their cobalt, tin and zinc complexes (C12QCo, C12QSn, C12QZn, C16QCo, C16QSn and C16QZn) were synthesized. The chemical structures of the synthesized compounds were confirmed using different spectroscopy tools. The surface activities of the synthesized surfactants were influenced by their chemical structures and the type of the transition metals. The synthesized cationic surfactants and their metal complexes were evaluated as biocides against Gram- positive bacteria (Bacillus subtilis and Micrococcus luteus), Gram-negative bacteria (Bordetella pertussis and Escherichia coli) and fungi (Candida albicans and Aspergillus niger), while the sulfate-reducing bacteria were tested against Desulfomonas pigra. The biological activity results showed that the cationic surfactants exhibit moderate to high efficiency against the tested microorganisms. The antimicrobial activity was strongly increased by complexation of these cationic surfactants with Co(II), Sn(II) and Zn(II) ions. The antimicrobial activity of the synthesized compounds against sulfate-reducing bacteria showed promising results in the field of biocides application.







Similar content being viewed by others
References
X. Zhang, C. Wang, Chem. Soc. Rev. 40, 94–101 (2011)
R.L. Grant, C. Yao, D. Gabaldon, D. Acosta, Toxicology 76, 153–176 (1992)
N. Vlachy, D. Touraud, J. Heilmann, W. Kunz, Colloids Surf. B Biointerfaces 70, 278–280 (2009)
T. Kosuri, K. Hemalatha, R. Karuna, B.S. Rao, Mycotoxin Res. 26, 155–170 (2010)
J. Hrenovic, T. Ivankovic, Cent. Eur. J. Biol. 2, 405–414 (2007)
M.T. Garcia, E. Campos, J. Sanchez-Leal, I. Ribosa, Chemosphere 38, 3473–3483 (1999)
D.B. Vieira, A.M. Carmona-Ribeiro, J. Antimicrob. Chemother. 58, 760–767 (2006)
T. Hamouda, J.R. Baker, J. Appl. Microbiol. 89, 397–403 (2000)
J.M. Kofonow, N.D. Adappa, Relat. Spec. 74, 179–184 (2012)
G. Viscardi, P. Quagliotto, C. Barolo, P. Savarino, E. Barni, E. Fisicaro, J. Org. Chem. 65, 8197–8203 (2000)
T. Wu, A.G. Xie, S.Z. Tan, X. Cai, Colloids Surf. B Biointerfaces 86, 232–236 (2011)
S. He, B. Wang, H. Chen, C. Tang, Y. Feng, Appl. Mater. Interfaces 4, 2116–2123 (2012)
N. Simonetti, F.D. D‘Auria, V. Strippoli, Chemotherapy 37, 32–37 (1991)
A. Mandal, V. Meda, W.J. Zhang, K.M. Farhan, A. Gnanamani, Colloids Surf. B Biointerfaces 90, 191–196 (2012)
D.H. Dusane, V.S. Pawar, Y.V. Nancharaiah, V.P. Venugopalan, A.R. Kumar, S.S. Zinjarde, Biofouling 27, 645–654 (2011)
R.D. Rufino, J.M. Luna, L.A. Sarubbo, L.R.M. Rodrigues, J.A.C. Teixeira, G.M. Campos-Takaki, Colloids Surf. B Biointerfaces 84, 1–5 (2011)
M.I. Sriram, K. Kalishwaralal, V. Deepak, R. Gracerosepat, K. Srisakthi, S. Gurunathan, Colloids Surf. B Biointerfaces 85, 174–181 (2011)
S.M. Simoes, L.C. Simoes, S. Cleto, M.O. Pereira, M.J. Vieira, Int. J. Food Microbiol. 121, 335–341 (2008)
D. Enjalbert, C. Bassilana, V. Krier, S. Szönyi, A. Cambon, Res. Chem. Intermed. 24, 667–678 (1998)
M. Nazari, M. Kurdi, H. Heerklotz, Biophys. J. 102, 498–506 (2012)
K. Nakata, T. Tsuchido, Y. Matsumura, J. Appl. Microbiol. 110, 568–579 (2011)
K. Nakata, M.M. Koh, T. Tsuchido, Y. Matsumura, Appl. Microbiol. Biotechnol. 87, 1895–1905 (2010)
N.A. Negm, S.M. Tawfik, J. Ind. Eng. Chem. (2014). doi:10.1016/j.jiec.2014.02.018
F.A. Bassyouni, S.M. Abu-Bakr, K.H. Hegab, W. El-Eraky, A.A. El Beih, M.E. Abdel Rehim, Res. Chem. Intermed. 38, 1527–1550 (2012)
A.A. Al-Amiery, Res. Chem. Intermed. 38, 745–759 (2012)
A.S. Megahed, M.S. Al-Amoudi, M.S. Refat, Res. Chem. Intermed. 40, 1425–1439 (2014)
M.M. Abd-Elzaher, A.A. Labib, H.A. Mousa, S.A. Oustafa, M.M. Abdallah, Res. Chem. Intermed. 40, 1923–1936 (2014)
D.A. Jaeger, V.B. Reddy, N. Arulsamy, D.S. Bohle, D.W. Grainger, B. Berggren, Langmuir 14, 2589 (1998)
F.M. Menger, L.H. Gan, E. Johnson, D.H. Durst, J. Am. Chem. Soc. 109, 2800 (1987)
Z. Oren, J. Hong, Y.J. Shai, Biol. Chem. 272, 14643 (1997)
S.M. Shaban, A. Sayed, S.M. Tawfik, A. Abd-Elaal, I. Aiad, J. Ind. Eng. Chem. 19, 2004–2009 (2013)
N. Negm, S.M. Tawfik, Chem. Today J. 30(6), 5–8 (2012)
I. Aiad, S.M. Tawfik, S.M. Shaban, A. Abd-Elaal, M. El-Shafie, J. Surfactants Deterg. 17, 391–401 (2014)
A. Abd-Elaal, I. Aiad, S.M. Shaban, S.M. Tawfik, A. Sayed, J. Surfactants Deterg. 17, 483–491 (2014)
N.A. Negm, Egypt. J. Chem. 45, 483 (2002)
N.A. Negm, A. El-Farargy, M.F. Zaki, S.A. Mahmoud, N. Abdel Rahman, Egypt. J. Pet. 17, 15 (2008)
R. Agarwal, L. Singh, D.K. Sharma, Bioinorg. Chem. Appl. 1, 34 (2006)
N.A. Negm, S.M.I. Morsy, A.M. Badawi, Egypt. J. Chem. 48, 645–652 (2005)
N.A. Negm, Y.M. Elkholy, F.M. Ghuiba, M.K. Zahran, S.A. Mahmoud, S.M. Tawfik, J. Adsorpt. Sci. Technol. 32, 512–518 (2011)
G.H. Sayed, F.M. Ghuiba, M.I. Abdou, E.A. Badr, S.M. Tawfik, N.A. Negm, Colloids Surf. A Physicochem. Eng. Asp. 393, 96 (2012)
R.E. Cooper, Analytical Microbiology (Academic Press, New York, 1972)
National Committee for Clinical Laboratory Standards, Methods for Dilution Antimicrobial Susceptibility Tests for Bacteria that Grow Aerobically. Approved Standard M7-A4, National Committee for Clinical Laboratory standards, Wayne (1997)
S.S. Shah, M.S. Khan, H. Ullah, M.A. Awan, J. Colloid Interface Sci. 186, 382–386 (1997)
D.N. Muanza, B.W. Kim, K.L. Euler, L. Williams, Int. J. Pharm. 32, 337–345 (1994)
N.A. Negm, I.A. Aiad, S.M. Tawfik, J. Surfactants Deterg. 13, 503–511 (2010)
N.A. Negm, F.M. Ghuiba, S.A. Mahmoud, S.M. Tawfik, Eng. Life Sci. 11(5), 496–510 (2011)
H.S. Galal, F.M. Ghuiba, M.I. Abdou, E.A. Badr, S.M. Tawfik, N.A. Negm, J. Surfactants Deterg. 15(6), 735–743 (2012)
N.A. Negm, Y.M. Elkholy, M.K. Zahran, S.M. Tawfik, Corros. Sci. 52, 3523–3536 (2010)
M.J. Rosen, Surfactants and Interfacial Phenomena, 2nd edn. (Wiley, New York, 1989)
Z. Feng, X. Guo, L. Jia, Y. Zhang, Res. Chem. Intermed. (2013). doi:10.1007/s11164-013-1492-6
N.A. Negm, F.M. Ghuiba, S.M. Tawfik, Corros. Sci. 53, 3566–3575 (2011)
R. Zana, J. Colloids Interface Sci. 248, 203–220 (2002)
H. Nagamune, T. Maeda, K. Ohkura, K. Yamamoto, M. Nakajima, H. Kourai, Toxicol. In Vitro 14, 139–147 (2000)
G. Viscardi, P. Quagliotto, C. Barolo, P. Savarino, E. Barni, E. Fisciaro, J. Agro Chem. 65, 8197–8203 (2000)
A. Timin, E. Rumyantsev, Res. Chem. Intermed. (2013). doi:10.1007/s11164-013-1358-y
C.R. Birnie, D. Malamud, R.L. Schnaare, Antimicrob. Agents Chemother. 44, 2514–2517 (2000)
C. Campanac, L. Pineau, A. Payard, G. Baziard-Mouysset, C. Roques, Antimicrob. Agents Chem. 46, 1469–1474 (2002)
C. Jamora, M.A. Theodoraki, V. Malhotra, E.A. Theodorakis, Bioorg. Med. Chem. 9(6), 1365 (2001)
M. Ganzle, Appl. Microbiol. Biotechnol. 64(3), 326 (2004)
I. Chernomordik, M.M. Kozlov, J. Zimmerberg, J. Membr. Biol. 146, 1 (1995)
M.H.J. Saier, Enzymes in Metabolic Pathways (Harper & Row, New York, 1987)
D.L. Jack, N.M. Yang, M.H. Saier, J. Eur. J. Biochem. 268, 3620 (2001)
A. Mobinikhaledi, M. Jabbarpour, Res. Chem. Intermed. (2013). doi:10.1007/s11164-013-1205-1
K. Medjahed, L. Tennouga, A. Mansri, O. Baba, Res. Chem. Intermed. 39, 4099–4107 (2013)
S.M. Tawfik, A. Sayed, I. Aiad, J. Surfactants Deterg. 15, 577 (2012)
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Tawfik, S.M., Zaky, M.F. Synthesis, characterization and antimicrobial activity of N,N-bis(hydroxymethyl)-N-[(2-mercaptoacetoxy)methyl]alkyl ammonium bromide surfactant and their Co(II), Zn(II) and Sn(II) complexes. Res Chem Intermed 41, 7925–7943 (2015). https://doi.org/10.1007/s11164-014-1867-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11164-014-1867-3