Abstract
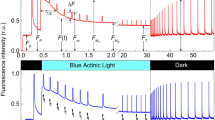
Low light (LL) and high light (HL)-acclimated plants of A. thaliana were exposed to blue (BB) or red (RR) light or to a mixture of blue and red light (BR) of incrementally increasing intensities. The light response of photosystem II was measured by pulse amplitude-modulated chlorophyll fluorescence and that of photosystem I by near infrared difference spectroscopy. The LL but not HL leaves exhibited blue light-specific responses which were assigned to relocation of chloroplasts from the dark to the light-avoidance arrangement. Blue light (BB and BR) decreased the minimum fluorescence (\(F_{0}^{\prime }\)) more than RR light. This extra reduction of the \(F_{0}^{\prime }\) was stronger than theoretically predicted for \(F_{0}^{\prime }\) quenching by energy dissipation but actual measurement and theory agreed in RR treatments. The extra \(F_{0}^{\prime }\) reduction was assigned to decreased light absorption of chloroplasts in the avoidance position. A maximum reduction of 30% was calculated. Increasing intensities of blue light affected the fluorescence parameters NPQ and qP to a lesser degree than red light. After correcting for the optical effects of chloroplast relocation, the NPQ responded similarly to blue and red light. The same correction method diminished the color-specific variations in qP but did not abolish it; thus strongly indicating the presence of another blue light effect which also moderates excitation pressure in PSII but cannot be ascribed to absorption variations. Only after RR exposure, a post-illumination overshoot of \(F_{0}^{\prime }\) and fast oxidation of PSI electron acceptors occurred, thus, suggesting an electron flow from stromal reductants to the plastoquinone pool.









Similar content being viewed by others
References
Allen JF (1992) Protein phosphorylation in regulation of photosynthesis. Biochim Biophys Acta 1098:275–335. https://doi.org/10.1016/S0005-2728(09)91014-3
Allen JF, de Paula WBM, Puthiyaveetil S, Nield J (2011) A structural phylogenetic map for chloroplast photosynthesis. Trends Plant Sci 16:645–655. https://doi.org/10.1016/j.tplants.2011.10.004
Anderson JM, Chow WS, Park Y-I (1995) The grand design of photosynthesis: acclimation of the photosynthetic apparatus to environmental cues. Photosynth Res 46:129–139
Antal TK, Kovalenko IB, Rubin AB, Tyystjärvi E (2013) Photosynthesis-related quantities for education and modeling. Photosynth Res 117:1–30. https://doi.org/10.1007/BF00020423
Armbruster U, Correa GV, Kunz H-H, Strand DD (2017) The regulation of the chloroplast proton motive force plays a key role for photosynthesis in fluctuating light. Curr Opin Plant Biol 37:56–62. https://doi.org/10.1016/j.pbi.2017.03.012
Bailey A, Walters RG, Jansson S, Horton P (2001) Acclimation of Arabidopsis thaliana to the light environment: the existence of separate low light and high light responses. Planta 213:794–801. https://doi.org/10.1007/s004250100556
Ballottari M, Dall’Osto L, Morosinotto T, Bassi R (2007) Contrasting behavior of higher plant photosystem I and II antenna systems during acclimation. J Biol Chem 282:8947–8958. https://doi.org/10.1074/jbc.M606417200
Ballottari M, Alcocer MJP, D’Andrea C, Viola D, Ahn TK, Petrozza A, Polli D, Fleming GR, Cerullo G, Bassi R (2014) Regulation of photosystem I light harvesting by zeaxanthin. Proc Natl Acad Sci USA 111:E2431–E2438. https://doi.org/10.1073/pnas.1404377111
Banaś AK, Aggarwal C, Łabuz J, Sztatelman O, Gabryś H (2012) Blue light signalling in chloroplast movements. J Exp Bot 63:1559–1574. https://doi.org/10.1093/jxb/err429
Bao H, Melnicki MR, Kerfeld CA (2017) Structure and functions of Orange Carotenoid Protein homologs in cyanobacteria. Curr Opin Plant Biol 37:1–9. https://doi.org/10.1016/j.pbi.2017.03.010
Bassi R, Dainese P (1992) A supramolecular light-harvesting complex from chloroplast photosystem-II membranes. Eur J Biochem 204:317–326. https://doi.org/10.1111/j.1432-1033.1992.tb16640.x
Bilger W, Björkman O (1990) Role of the xanthophyll cycle in photoprotection elucidated by measurements of light-induced absorbance changes, fluorescence and photosynthesis in leaves of Hedera canariensis. Photosynth Res 25:173–185. https://doi.org/10.1007/BF00033159
Boardman NK (1977) Comparative photosynthesis of sun and shade plants. Annu Rev Plant Physiol 28:355–377. https://doi.org/10.1146/annurev.pp.28.060177.002035
Boichenko VA (1998) Action spectra and functional antenna sizes of Photosystems I and II in relation to the thylakoid membrane organization and pigment composition. Photosynth Res 58:163–174. https://doi.org/10.1023/A:1006187425058
Briggs WR, Christie JM (2002) Phototropins 1 and 2: versatile plant blue-light receptors. Trends Plant Sci 7:204–210. https://doi.org/10.1016/S1360-1385(02)02245-8
Brugnoli E, Björkman O (1992) Chloroplast movements in leaves: Influence on chlorophyll fluorescence and measurements of light-induced absorbance changes related to ∆pH and zeaxanthin formation. Photosynth Res 32:23–35. https://doi.org/10.1007/BF00028795
Büchel C (2015) Evolution and function of light harvesting proteins. J Plant Physiol 172:62–75. https://doi.org/10.1016/j.jplph.2014.04.018
Buer CS, Imin N, Djordjevic MA (2010) Flavonoids: new roles for old molecules. J Integr Plant Biol 52:98–111. https://doi.org/10.1111/j.1744-7909.2010.00905.x
Byrdin M, Rimke I, Schlodder E, Stehlik D, Roelofs TA (2000) Decay kinetics and quantum yields of fluorescence in Photosystem I from Synechococcus elongatus with P700 in the reduced and oxidized state: are the kinetics of excited state decay trap-limited or transfer-limited? Biophys J 79:992–1007. https://doi.org/10.1016/S0006-3495(00)76353-3
Cazzaniga S, Dall’ Osto L, Kong S-G, Wada M, Bassi R (2013) Interaction between avoidance of photon absorption, excess energy dissipation and zeaxanthin synthesis against photooxidative stress in Arabidopsis. Plant J 76:568–579. https://doi.org/10.1111/tpj.12314
Cerovic ZG, Samson G, Morales F, Tremblay N, Moya I (1999) Ultraviolet-induced fluorescence for plant monitoring: present state and prospects. Agronomie 19:543–578
Cerovic ZG, Masdoumier G, Ben Ghozlen N, Latouche G (2012) A new optical leaf-clip meter for simultaneous non-destructive assessment of leaf chlorophyll and epidermal flavonoids. Physiol Plant 146:251–260. https://doi.org/10.1111/j.1399-3054.2012.01639.x
Chory J (2010) Light signal transduction: an infinite spectrum of possibilities. Plant J 61:982–991. https://doi.org/10.1111/j.1365-313X.2009.04105.x
Christie JM (2007) Phototropin blue-light receptors. Annu Rev Plant Biol 58:21–45. https://doi.org/10.1146/annurev.arplant.58.032806.103951
Comayras F, Jungas C, Lavergne J (2005) Functional consequences of the organization of the photosynthetic apparatus in Rhodobacter sphaeroides. I. Quinone domains and excitation transfer in chromatophores and reaction center-antenna complexes. J Biol Chem 280:11203–11213. https://doi.org/10.1074/jbc.M412088200
Dall’Osto L, Cazzaniga S, Wada M, Bassi R (2014) On the origin of a slowly reversible fluorescence decay component in the Arabidopsis npq4 mutant. Phil Trans R Soc Lond B 369:1–11. https://doi.org/10.1098/rstb.2013.0221
Davis PA, Hangarter RP (2012) Chloroplast movement provides photoprotection to plants by redistributing PSII damage within leaves. Photosynth Res 112:153–161. https://doi.org/10.1007/s11120-012-9755-4
Demmig-Adams B, Winter K, Krüger A, Czygan F-C (1989) Light response of CO2 assimilation, dissipation of excess excitation energy, and zeaxanthin content of sun and shade leaves. Plant Physiol 90:881–886. https://doi.org/10.1104/pp.90.3.881
Diner BA (1977) Dependence of the deactivation reactions of photosystem II on the redox state of plastoquinone pool A varied under anaerobic conditions. Equilibria on the acceptor side of photosystem II. Biochim Biophys Acta 460:247–258. https://doi.org/10.1016/0005-2728(77)90211-0
Dutta S, Cruz JA, Jiao Y, Chen J, Kramer DM, Osteryoung KW (2015) Non-invasive, whole-plant imaging of chloroplast movement and chlorophyll fluorescence reveals photosynthetic phenotypes independent of chloroplast photorelocation defects in chloroplast division mutants. Plant J 84:428–442. https://doi.org/10.1111/tpj.13009
Endo T, Shikanai T, Takabayashi A, Asada K, Sato F (1999) The role of chloroplastic NAD(P)H dehydrogenase in photoprotection. FEBS Lett 457:5–8. https://doi.org/10.1016/S0014-5793(99)00989-8
Endo T, Kawase D, Sato F (2005) Stromal over-reduction by high-light stress as measured by decreases in P700 oxidation by far-red light and its physiological relevance. Plant Cell Physiol 46:775–781. https://doi.org/10.1093/pcp/pci084
Foyer CH, Lelandais M, Harbinson J (1992) Control of the quantum efficiencies of photosystems I and II, electron flow, and enzyme activation following dark-to-light transitions in pea leaves. Relationship between NADP/NADPH ratios and NADP-malate dehydrogenase activation state. Plant Physiol 99:979–986. https://doi.org/10.1104/pp.99.3.979
Galvão VC, Fankhauser C (2015) Sensing the light environment in plants: photoreceptors and early signaling steps. Curr Opin Neurobiol 34:46–53. https://doi.org/10.1016/j.conb.2015.01.013
Genty B, Briantais J-M, Baker NR (1989) The relationship between the quantum yield of photosynthetic electron transport and quenching of chlorophyll fluorescence. Biochim Biophys Acta 990:87–92. https://doi.org/10.1016/S0304-4165(89)80016-9
Gilmore AM, Yamamoto HY (1991) Zeaxanthin formation and energy-dependent fluorescence quenching in pea chloroplasts under artificially mediated linear and cyclic electron transport. Plant Physiol 96:635–643. https://doi.org/10.1104/pp.96.2.635
Giovagnetti V, Ware MA, Ruban AV (2015) Assessment of the impact of photosystem I chlorophyll fluorescence on the pulse-amplitude modulated quenching analysis in leaves of Arabidopsis thaliana. Photosynth Res 125:179–189. https://doi.org/10.1007/s11120-015-0087-z
Gorton HL, Williams WE, Vogelmann TC (1999) Chloroplast movement in Alocasia macrorrhiza. Physiol Plant 106:421–428. https://doi.org/10.1034/j.1399-3054.1999.106410.x
Gorton HL, Herbert SK, Vogelmann TC (2003) Photoacoustic analysis indicates that chloroplast movement does not alter liquid-phase CO2 diffusion in leaves of Alocasia brisbanensis. Plant Physiol 132:1529–1539. https://doi.org/10.1104/pp.102.019612
Goss R, Jakob T (2010) Regulation and function of xanthophyll cycle-dependent photoprotection in algae. Photosynth Res 106:103–122. https://doi.org/10.1007/s11120-010-9536-x
Goss R, Lepetit B (2015) Biodiversity of NPQ. J Plant Physiol 172:13–32. https://doi.org/10.1016/j.jplph.2014.03.004
Haldrup A, Jensen PE, Lunde C, Scheller HV (2001) Balance of power: a view of the mechanism of photosynthetic state transitions. Trends Plant Sci 6:301–305. https://doi.org/10.1016/S1360-1385(01)01953-7
Harbinson J, Genty B, Baker NR (1989) Relationship between the quantum efficiencies of photosystems I and II in pea leaves. Plant Physiol 90:1029–1034. https://doi.org/10.1104/pp.90.3.1029
Ho QT, Berghuijs HNC, Watté R, Verboven P, Herremans E, Yin X, Retta MA, Aernouts B, Saeys W, Helfen L, Farquhar GD, Struik PC, Nicolaï BM (2016) Three-dimensional microscale modelling of CO2 transport and light propagation in tomato leaves enlightens photosynthesis. Plant Cell Environ 39:50–61. https://doi.org/10.1111/pce.12590
Hogewoning SW, Wientjes E, Douwstra P, Trouwborst G, van Ieperen W, Croce R, Harbinson J (2012) Photosynthetic quantum yield dynamics:from photosystems to leaves. Plant Cell 24:1921–1935. https://doi.org/10.1105/tpc.112.097972
Horton P, Black MT (1981) Light-dependent quenching of chlorophyll fluorescence in pea chloroplasts induced by adenosine 5′-triphosphate. Biochim Biophys Acta 635:53–62. https://doi.org/10.1016/0005-2728(81)90006-2
Hüner NPA, Bode R, Dahal K, Hollis L, Rosso D, Krol M, Ivanov AG (2012) Chloroplast redox imbalance governs phenotypic plasticity: the “grand design of photosynthesis” revisited. Front Plant Sci 255:1. https://doi.org/10.3389/fpls.2012.00255
Inada K (1980) Spectral absorption property of pigments in living leaves and its contribution to photosynthesis. Japan J Crop Sci 49:286–294. https://doi.org/10.1626/jcs.49.286
Jarillo JA, Gabrys H, Capel J, Alonso JM, Ecker JR, Cashmore AR (2001) Phototropin-related NPL1 controls chloroplast relocation induced by blue light. Nature 410:952–954. https://doi.org/10.1038/35073622
Jenkins GI (2017) Photomorphogenic responses to ultraviolet-B light. Plant Cell Environ. https://doi.org/10.1111/pce.12934
Kadota A, Yamada N, Suetsugu N, Hirose M, Saito C, Shoda K, Ichikawa S, Kagawa T, Nakano A, Wada M (2009) Short actin-based mechanism for light-directed chloroplast movement in Arabidopsis. Proc Natl Acad Sci USA 106:13106–13111. https://doi.org/10.1073/pnas.0906250106
Kagawa T, Wada M (2000) Blue light-induced chloroplast relocation in Arabidopsis thaliana as analyzed by microbeam irradiation. Plant Cell Physiol 41:84–93. https://doi.org/10.1093/pcp/41.1.84
Kagawa T, Sakai T, Suetsugu N, Oikawa K, Ishiguro S, Kato T, Tabata S, Okada K, Wada M (2001) Arabidopsis NPL1: a phototropin homolog controlling the chloroplast high-light avoidance response. Science 291:2138–2141. https://doi.org/10.1126/science.291.5511.2138
Kasahara M, Kagava T, Oikava K, Suetsugu N, Miyao M, Wada M (2002a) Chloroplast avoidance movement reduces photodamage in plants. Nature 420:829–832. https://doi.org/10.1038/nature01213
Kasahara M, Swartz TE, Olney MA, Onodera A, Mochizuki N, Fukuzawa H, Asamizu E, Tabata S, Kanegae H, Takano M, Christie JM, Nagatani A, Briggs WR (2002b) Photochemical properties of the flavin mononucleotide-binding domains of the phototropins from Arabidopsis, rice, and Chlamydomonas reinhardtii. Plant Physiol 129:762–773. https://doi.org/10.1104/pp.002410
Keren N, Krieger-Liszkay A (2011) Photoinhibition: molecular mechanisms and its physiological significance. Physiol Plant 142:1–5. https://doi.org/10.1111/j.1399-3054.2011.01467.x
Kim G-T, Yano S, Kozuka T, Tsukaya H (2005) Photomorphogenesis of leaves: shade-avoidance and differentiation of sun and shade leaves. Photochem Photobiol Sci 4:770–774. https://doi.org/10.1039/B418440H
Klughammer C, Schreiber U (1994) An improved method, using saturating light pulses, for the determination of photosystem I quantum yield via P700+-absorbance changes at 830 nm. Planta 192:261–268. https://doi.org/10.1007/BF01089043
Knapp A, Carter G (1998) Variability in leaf optical properties among 26 species from a broad range of habitats. Am J Bot 85:940–946
Kolb C, Käser M, Kopecký J, Zotz G, Riederer M, Pfündel EE (2001) Effects of natural intensities of visible and UV radiation on epidermal UV-screening and photosynthesis in grape leaves (Vitis vinifera cv. Silvaner). Plant Physiol 127:863–875. https://doi.org/10.1104/pp.010373
Kong S-G, Wada M (2016) Molecular basis of chloroplast photorelocation movement. J Plant Res 129:159–166. https://doi.org/10.1007/s10265-016-0788-1
Königer M, Delamaide JA, Marlow ED, Harris GC (2008) Arabidopsis thaliana leaves with altered chloroplast numbers and chloroplast movement exhibit impaired adjustments to both low and high light. J Exp Bot 59:2285–2297. https://doi.org/10.1093/jxb/ern099
Le Quiniou C, Tian L, Drop B, Wientjes E, van Stokkum IHM, van Oort B, Croce R (2015) PSI-LHCI of Chlamydomonas reinhardtii: increasing the absorption cross section without losing efficiency. Biochim Biophys Acta 1847:458–467. https://doi.org/10.1016/j.bbabio.2015.02.001
Lepetit B, Dietzel L (2015) Light signaling in photosynthetic eukaryotes with ‘green’ and ‘red’ chloroplasts. Environ Exp Bot 114:30–47. https://doi.org/10.1016/j.envexpbot.2014.07.007
Li FW, Rothfels CJ, Melkonian M, Villarreal JC, Stevenson DW, Graham SW, Wong GK, Mathews S, Pryer KM (2015) The origin and evolution of phototropins. Front Plant Sci 6:637. https://doi.org/10.3389/fpls.2015.00637
Lichtenthaler HK, Buschmann C, Döll M, Fietz H-J, Bach T, Kozel U, Meier D, Rahmsdorf U (1981) Photosynthetic activity, chloroplast ultrastructure, and leaf characteristics of high-light and low-light plants and of sun and shade leaves. Photosynth Res 2:115–141. https://doi.org/10.1007/BF00028752
Loreto F, Tsonev T, Centritto M (2009) The impact of blue light on leaf mesophyll conductance. J Exp Bot 60:2283–2290. https://doi.org/10.1093/jxb/erp112
Louis J, Cerovic ZG, Moya I (2006) Quantitative study of fluorescence excitation and emission spectra of bean leaves. J Photochem Photobiol 85:65–71. https://doi.org/10.1016/j.jphotobiol.2006.03.009
Luesse DR, DeBlasio SL, Hangarter RP (2010) Integration of phot1, phot2, and PhyB signalling in light-induced chloroplast movements. J Exp Bot 61:4387–4397. https://doi.org/10.1093/jxb/erq242
Mekala NR, Suorsa M, Rantala M, Aro E-M, Tikkanen M (2015) Plants actively avoid state-transitions upon changes in light intensity: role of light-harvesting complex II protein dephosphorylation in high light. Plant Physiol 168:721–734. https://doi.org/10.1104/pp.15.00488
Melis A (1991) Dynamics of photosynthetic membrane composition and function. Biochim Biophys Acta 1058:87–106. https://doi.org/10.1016/S0005-2728(05)80225-7
Merzlyak MN, Melø TB, Naqvi KR (2008) Effect of anthocyanins, carotenoids, and flavonols on chlorophyll fluorescence excitation spectra in apple fruit: signature analysis, assessment, modelling, and relevance to photoprotection. J Exp Bot 59:349–359. https://doi.org/10.1093/jxb/erm316
Munekage Y, Hashimoto M, Miyake C, Tomizawa K-I, Endo T, Tasaka M, Shikanai T (2004) Cyclic electron flow around photosystem I is essential for photosynthesis. Nature 429:579–582. https://doi.org/10.1038/nature02598
Nichelmann L, Schulze M, Herppich W, Bilger W (2016) A simple indicator for non-destructive estimation of the violaxanthin cycle pigment content in leaves. Photosynth Res 128:183–193. https://doi.org/10.1007/s11120-016-0218-1
Nishiyama Y, Murata N (2014) Revised scheme for the mechanism of photoinhibition and its application to enhance the abiotic stress tolerance of the photosynthetic machinery. Appl Microbiol Biotechnol 98:8777–8796. https://doi.org/10.1007/s00253-014-6020-0
Niyogi KK, Grossman AR, Björkman O (1998) Arabidopsis mutants define a central role for the xanthophyll cycle in the regulation of photosynthetic energy conversion. Plant Cell 10:1121–1134. https://doi.org/10.1105/tpc.10.7.1121
Oguchi R, Douwstra P, Fujita T, Chow WS, Terashima I (2011) Intra-leaf gradients of photoinhibition induced by different color lights: Implications for the dual mechanisms of photoinhibition and for the application of conventional chlorophyll fluorometers. New Phytol 191:146–159. https://doi.org/10.1111/j.1469-8137.2011.03669.x
Okajima K (2016) Molecular mechanism of phototropin light signaling. J Plant Res 129:149–157. https://doi.org/10.1007/s10265-016-0783-6
Omasa K, Takayama K (2003) Simultaneous measurement of stomatal conductance, non-photochemical quenching, and photochemical yield of photosystem II in intact leaves by thermal and chlorophyll fluorescence imaging. Plant Cell Physiol 44:1290–1300. https://doi.org/10.1093/pcp/pcg165
Oxborough K, Baker NR (1997) Resolving chlorophyll a fluorescence images of photosynthetic efficiency into photochemical and non-photochemical components — calculation of qP and Fv’/Fm’ without measuring Fo’. Photosynth Res 54:135–142. https://doi.org/10.1023/A:1005936823310
Park Y-I, Chow WS, Anderson JM (1995) Light inactivation of functional photosystem II in leaves of peas grown in moderate light depends on photon exposure. Planta 196:401–411. https://doi.org/10.1007/BF00203636
Pfündel EE (2003) Action of UV and visible radiation on chlorophyll fluorescence from dark-adapted grape leaves (Vitis vinifera L.). Photosynth Res 75:29–39. https://doi.org/10.1023/A:1022486925516
Pfündel EE (1998) Estimating the contribution of photosystem I to total leaf chlorophyll fluorescence. Photosynth Res 56:185–195. https://doi.org/10.1023/A:1006032804606
Pfündel EE (2009) Deriving room temperature excitation spectra for photosystem I and photosystem II fluorescence in intact leaves from the dependence of FV/FM on measuring wavelength. Photosynth Res 100:163–177. https://doi.org/10.1007/s11120-009-9453-z
Pfündel EE, Baake E (1990) A quantitative description of fluorescence excitation spectra in intact bean leaves greened under intermittent light. Photosynth Res 26:19–28. https://doi.org/10.1007/BF00048973
Pfündel EE, Agati G, Cerovic ZG (2008) Optical properties of plant surfaces. In: Riederer M, Müller C (eds) Biology of the plant cuticle. Annual plant reviews, vol 23. Blackwell Publishing, Oxford, pp 216–249
Pfündel EE, Klughammer C, Meister A, Cerovic ZG (2013) Deriving fluorometer-specific values of relative PSI fluorescence intensity from quenching of F0 fluorescence in leaves of Arabidopsis thaliana and Zea mays. Photosynth Res 114:189–206. https://doi.org/10.1007/s11120-012-9788-8
Pons TL (2012) Interaction of temperature and irradiance effects on photosynthetic acclimation in two accessions of Arabidopsis thaliana. Photosynth Res 113:207–219. https://doi.org/10.1007/s11120-012-9756-3
Quick WP, Stitt M (1989) An examination of factors contribution to non-photochemical quenching of chlorophyll fluorescence in barley leaves. Biochim Biophys Acta 977:287–296. https://doi.org/10.1016/S0005-2728(89)80082-9
Rintamäki E, Salonen M, Suoranta U-M, Carlberg I, Andersson B, Aro E-M (1997) Phosphorylation of light-harvesting complex II and photosystem II core proteins shows different irradiance-dependent regulation in vivo. J Biol Chem 272:30476–30482. https://doi.org/10.1074/jbc.272.48.30476
Rintamäki E, Martinsuo P, Pursiheimo S, Aro E-M (2000) Cooperative regulation of light-harvesting complex II phosphorylation via the plastoquinol and ferredoxin-thioredoxin system in chloroplasts. Proc Natl Acad Sci USA 97:11644–11649. https://doi.org/10.1073/pnas.180054297
Ruban AV (2009) Plants in light. Commun Integr Biol 2:50–55. https://doi.org/10.1074/jbc.272.48.30476
Ruban AV (2016) Nonphotochemical chlorophyll fluorescence quenching: mechanism and effectiveness in protecting plants from photodamage. Plant Physiol 170:1903–1916. https://doi.org/10.1104/pp.15.01935
Ruban AV, Johnson MP (2009) Dynamics of higher plant photosystem cross-section associated with state transitions. Photosynth Res 99:173–183. https://doi.org/10.1007/s111200089387x
Sakurai N, Domoto K, Takagi S (2005) Blue-light-induced reorganization of the actin cytoskeleton and the avoidance response of chloroplasts in epidermal cells of Vallisneria gigantea. Planta 221:66–74. https://doi.org/10.1007/s00425-004-1416-1
Sarvikas P, Hakala M, Pätsikkä E, Tyystjärvi T, Tyystjärvi E (2006) Action spectrum of photoinhibition in leaves of wild type and npq 1-2 and npq4-1 mutants of Arabidopsis thaliana. Plant Cell Physiol 47:391–400. https://doi.org/10.1093/pcp/pcj006
Schlodder E, Çetin M, Byrdin M, Terekhova I, Karapetyan NV (2005) P700+- and 3P700-induced quenching of the fluorescence at 760 nm in trimeric Photosystem I complexes from the cyanobacterium Arthrospira platensis. Biochim Biophys Acta 1706:53–67. https://doi.org/10.1016/j.bbabio.2004.08.009
Schöttler MA, Tóth SZ (2014) Photosynthetic complex stoichiometry dynamics in higher plants: environmental acclimation and photosynthetic flux control. Front Plant Sci 5:188. https://doi.org/10.3389/fpls.2014.00188
Schreiber U (2004) Pulse-amplitude-modulation (PAM) fluorometry and saturation pulse method: an overview. In: Papageorgiou GC, Govindjee (eds) Chlorophyll a fluorescence: a signature of photosynthesis. Advances in Photosynthesis and Respiration, vol 19. Springer, Dordrecht, pp 279–319
Schreiber U, Schliwa U, Bilger W (1986) Continuous recording of photochemical and non-photochemical chlorophyll fluorescence quenching with a new type of modulation fluorometer. Photosynth Res 10:51–62. https://doi.org/10.1007/BF00024185
Shikanai T, Yamamoto H (2017) Contribution of cyclic and pseudo-cyclic electron transport to the formation of proton motive force in chloroplasts. Mol Plant 10:20–29. https://doi.org/10.1016/j.molp.2016.08.004
Shimazaki K-I, Doi M, Assmann SM, Kinoshita T (2007) Light regulation of stomatal movement. Annu Rev Plant Biol 58:219–247. https://doi.org/10.1146/annurev.arplant.57.032905.105434
Solovchenko A (2010) Screening pigments: general questions. In: Solovchenko A (ed) Photoprotection in plants. Springer, Berlin, pp 9–31. https://doi.org/10.1007/978-3-642-13887-4
Sonoike K (2011) Photoinhibition of photosystem I. Physiol Plant 142:56–64. https://doi.org/10.1111/j.1399-3054.2010.01437.x
Suetsugu N, Wada M (2012) Chloroplast photorelocation movement: a sophisticated strategy for chloroplasts to perform efficient photosynthesis. In: Najafpour MM (ed) Advances in photosynthesis-fundamental aspects. Tech, Rijeka, Croatia, pp 215–234
Suetsugu N, Dolja VV, Wada M (2010) Why have chloroplasts developed a unique motility system? Plant Signal Behav 5:1190–1196. https://doi.org/10.4161/psb.5.10.12802
Sztatelman O, Waloszek A, Banaś AK, Gabryś H (2010) Photoprotective function of chloroplast avoidance movement: in vivo chlorophyll fluorescence study. J Plant Physiol 167:709–716. https://doi.org/10.1016/j.jplph.2009.12.015
Takahashi S, Milward SE, Yamori W, Evans JR, Hillier W, Badger MR (2010) The solar action spectrum of photosystem II damage. Plant Physiol 153:988–993. https://doi.org/10.1104/pp.110.155747 (supplemental data)
Takizawa K, Cruz JA, Kanazawa A, Kramer DM (2007) The thylakoid proton motive force in vivo. Quantitative, non-invasive probes, energetics, and regulatory consequences of light induced pmf. Biochim Biophys Acta 1767:1233–1244. https://doi.org/10.1016/j.bbabio.2007.07.006
Tholen D, Boom C, Noguchi KO, Ueda S, Katase T, Terashima I (2008) The chloroplast avoidance response decreases internal conductance to CO2 diffusion in Arabidopsis thaliana leaves. Plant Cell Environ 31:1688–1700. https://doi.org/10.1111/j.1365-3040.2008.01875.x
Tian L, Xu P, Chukhutsina VU, Holzwarth AR, Croce R (2017) Zeaxanthin-dependent nonphotochemical quenching does not occur in photosystem I in the higher plant Arabidopsis thaliana. Proc Natl Acad Sci USA 114:4828–4832. https://doi.org/10.1073/pnas.1621051114
Tikhonov AN (2014) The cytochrome b 6 f complex at the crossroad of photosynthetic electron transport pathways. Plant Physiol Biochem 81:163–183. https://doi.org/10.1016/j.plaphy.2013.12.011
Tikkanen M, Aro E-M (2014) Integrative regulatory network of plant thylakoid energy transduction. Trends Plant Sci 19:10–17. https://doi.org/10.1016/j.tplants.2013.09.003
Trissl HW (1997) Determination of the quenching efficiency of the oxidized primary donor of Photosystem I, P700+: Implications for the trapping mechanism. Photosynth Res 54:237–240. https://doi.org/10.1023/A:1005981016835
Trojan A, Gabryś H (1996) Chloroplast distribution in Arabidopsis thaliana (L.) depends on light conditions during growth. Plant Physiol 111:419–425. https://doi.org/10.1104/pp.111.2.419
Van Kooten O, Snel JFH (1990) The use of chlorophyll fluorescence nomenclature in plant stress physiology. Photosynth Res 2:147–150. https://doi.org/10.1007/BF00033156
Van Amerongen H, Van Grondelle R (2001) Understanding the energy transfer function of LHCII, the major light-harvesting complex of green plants. J Phys Chem 105:604–617. https://doi.org/10.1021/jp0028406
Vass I, Styring S, Hundal T, Koivuniemi A, Aro E-M, Andersson B (1992) Reversible and irreversible intermediates during photoinhibition of photosystem II: stable reduced QA species promote chlorophyll triplet formation. Proc Natl Acad Sci USA 89:1408–1412. https://doi.org/10.1073/pnas.89.4.1408
Vavilin DV, Tyystjärvi E, Aro E-M (1998) Model for the fluorescence induction curve of photoinhibited thylakoids. Biophys J 75:503–512. https://doi.org/10.1016/S0006-3495(98)77539-3
Wada M (2013) Chloroplast movement. Plant Sci 210:177–182. https://doi.org/10.1016/j.plantsci.2013.05.016
Walters RG (2005) Towards an understanding of photosynthetic acclimation. J Exp Bot 56:435–447. https://doi.org/10.1093/jxb/eri060
Walters RG, Horton P (1991) Resolution of components of non-photochemical chlorophyll fluorescence quenching in barley leaves. Photosynth Res 27(2):121-133
Ware MA, Belgio E, Ruban AV (2015) Photoprotective capacity of non-photochemical quenching in plants acclimated to different light intensities. Photosynth Res 126:261–274. https://doi.org/10.1007/s11120-015-0102-4
Wu S-H (2014) Gene expression regulation in photomorphogenesis from the perspective of the central dogma. Annu Rev Plant Biol 65:311–333. https://doi.org/10.1146/annurev-arplant-050213-040337
Zurzycki J (1955) Chloroplast arrangements as a factor in photosynthesis. Acta Soc Bot Pol 24:27–63. https://doi.org/10.5586/asbp.1955.003
Acknowledgements
We thank Frank Reichel and Thomas Simon for skillful technical assistance. We are grateful to Dr. Lars Nichelmann for providing seeds and instructions on plant growth. We thank Dr. Robert Porra for helping in preparing this manuscript. EP wishes to thank Marie-Françoise and Gabriel Cornic and the Heinz Walz GmbH for supporting his research stay at the Université Paris-Sud.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Pfündel, E.E., Latouche, G., Meister, A. et al. Linking chloroplast relocation to different responses of photosynthesis to blue and red radiation in low and high light-acclimated leaves of Arabidopsis thaliana (L.). Photosynth Res 137, 105–128 (2018). https://doi.org/10.1007/s11120-018-0482-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11120-018-0482-3