Abstract
Purpose
The fuel reduction prescribed burning and biochar application can have significant impacts on water and nitrogen (N) use efficiency of understory acacia species as well as soil carbon (C) and N pools in a suburban native forest subject to N deposition in Southeast Queensland, Australia.
Methods
We evaluated the impact of biochar application rates (0, 5.0 and 10.0 t biochar per hectare) and prescribed burning on soil-plant interactions in carbon (C) and N cycling in a suburban native forest in the first two years of biochar application or three and half years of the recently prescribed burning.
Results
Anthropogenic N deposition not only enhanced N losses caused by N leaching and denitrification, but also inhibited biological N fixation (BNF) by increasing N availability in forest systems. The Acacia leiocalyx with higher water use efficiency was more inclined to utilize easily available N resources (from N deposition), compared with A. disparismma. In this study, biochar application could indeed reduce N loss in forest soil and improve soil fertility by improving plant water and N use efficiency. Meanwhile, soil moisture content affected by biochar application also influenced soil N transformations by affecting soil microbial activity.
Conclusion
For urban forest soils, the high N availability caused by N deposition could inhibit the BNF in a suburban native forest ecosystem. The high-porosity physical structure of biochar applied increased the soil water content and soil N retention capacity.
Similar content being viewed by others
Introduction
Since the industrial revolution and rapid development in the 150 years, we have dramatically altered the global nitrogen (N) cycle, with widespread and often divergent effects on ecological systems and environmental quality (Galloway et al. 2008; Xu et al. 2009; Succarie et al. 2022). The N cycling in terrestrial ecosystems is mainly driven by three primary sources: biological N fixation (BNF), N mineralization, and atmospheric N deposition. Among them, the potential threat of atmospheric N deposition to forest health (Binkley and Högberg 1997) may cause N saturation, which increases the risk of N leaching, soil acidification and N denitrification (Conley et al. 2009; Huang et al. 2015; Zhang et al. 2018a, b, c), which is causing concern about significant impacts on global forest health and plant biodiversity (Vitousek et al. 1997; Sala et al. 2000; Phoenix et al. 2006; Cecchini et al. 2021). At the same time, forest N dynamics interact closely with other biogeochemical cycles and greater N emissions contribute to a litany of well-documented and undesirable environmental changes, including loss of biodiversity (Bobbink et al. 2010; Aryal et al. 2022), soil and surface water acidification (Moldan and Wright 2011; Zhou et al. 2023) and reductions in air quality (Wolfe and Patz 2002; Wang et al. 2023).
Biochar is formed by heating organic material under low oxygen concentrations in a process known as pyrolysis (Lehmann et al. 2011; Rady et al. 2016; Haldar and Purkait 2021). Biochar is enriched in C, considered relatively stable in the soil, and has better nutrient retention than other forms of organic matter (Woolf et al. 2010; Nguyen et al. 2017; Farrar et al. 2022). Hence, adding biochar to the soil becomes an option for carbon (C) sequestration, potentially improving soil quality and reducing environmental pollutant release (Lehmann 2007; Gurwick et al. 2013; Das et al. 2023). Moreover, biochar can act as a soil amendment to affect nutrient cycling and plant growth by affecting microbial community composition and activity, water holding capacity and pH, as well as improving root growth (Kuzyakov et al. 2009; Liang et al. 2010; Robertson et al. 2012; Biederman and Harpole 2013; Thomas and Gale 2015; Liu et al. 2021). In addition, labile C fractions of biochar may also accelerate the decomposition of old soil organic matter through the priming effect (Cross and Sohi 2011; Zimmerman et al. 2011; Wang et al. 2016; Rasul et al. 2022).
Recently, although people have been interested in biochar as a soil amendment to improve soil quality, improve and maintain soil fertility, and increase soil C sequestration (Glaser et al. 2002; Das et al. 2021; Luo et al. 2023), there is still a lack of clear understanding of the characteristics of biochar produced from different raw materials and under different pyrolysis conditions and its interaction with soil. In addition, some researchers believe that biochar can be used as a fertilizer to provide N for plant growth (Wu et al. 2016, 2017). By improving the pH, cation exchange capacity (CEC), organic carbon and ash content of the soil (Rahim et al. 2020; Tomczyk et al. 2020), biochar may further reduce N loss from N leaching and ammonium volatilization (Deluca et al. 2015). In addition to helping the soil retain water and nutrients, the oxygen-containing functional groups on the biochar surface also improve the soil’s water holding capacity (WHC) and nitrogen use efficiency (NUE) (Yu et al. 2018), promoting plant productivity and improving soil fertility (Palansooriya et al. 2019; Bai et al. 2022). The ability of biochar to directly supply nutrients is limited, but its application to soil can improve soil fertility by changing the availability of soil N (Zhu et al. 2017; Asadyar et al. 2021). The high-temperature biochar (600 °C) used in this study usually has a higher surface area, porosity and alkalinity, thereby enhancing the soil water holding capacity and nutrient use efficiency of the soil and improving soil nutrient uptake by plants (Zhang et al. 2015; Burrell et al. 2016; Ding et al. 2016; Zornoza et al. 2016). However, the optimal application rate of high-temperature biochar and its relationship with plant-soil interactions have not been studied well (Zhang et al. 2015; Lan et al. 2018).
Prescribed burning has been widely used as a forest management tool to reduce the risk of wildfire spread (May and Attiwill 2003; Reverchon et al. 2011; Bai et al. 2012; Francos and Úbeda 2021). Prescribed burning may cause N losses through volatilization and release terrestrial carbon (C) into the atmosphere (Thonicke et al. 2010; Muqaddas et al. 2016). Acacia species can help soil nutrient recovery by biological N fixation (BNF) and increasing carbon (C) sequestration, especially in forests after prescribed burning and fire (Guinto et al. 2000; Bai et al. 2014; Witt et al. 2017; Reverchon et al. 2020). Most forest soil N is in organic form, but N mineralization rates are low, and tree growth is N-limited (Sponseller et al. 2016). Biochar application has been shown to stimulate soil organic matter decomposition by increasing net N mineralization and nitrification rates (Ameloot et al. 2015; Case et al. 2015; Gundale et al. 2016).
Based on previous studies, plant-soil δ13C and δ15N values are related to soil N and water availability and are closely related to C and N cycling of soil-to-plant (Werth and Kuzyakov 2010; Nogués et al. 2023). There is a significant correlation between plant δ13C and soil moisture content, which makes the δ13C a powerful tool to evaluate plant water use efficiency (Klaus et al. 2016). By analysing δ15N in soil and plants, we can better understand the retention and use efficiency of N in forest ecosystems (Schlesinger 2009; Hietz et al. 2011; Ibell et al. 2013; Mao et al. 2022). According to the reports, in the process of microbial transformation, the soil substrate is usually rich in 15N due to the rapid movement of the substrate in the process of N loss (Kuzyakov and Xu 2013; Nogués et al. 2023). However, the altered global N cycle will have important consequences. The N deposition will increase the N losses via denitrification and leaching of nitrate and the production of reactive N, adversely affecting the environment, climate, biodiversity and human health (Robertson and Vitousek 2009; Fowler et al. 2013; Cheng et al. 2020; Song et al. 2020). This study aimed to quantify the impact of biochar application rates (5.0 and 10.0 t biochar per hectare) on soil-plant interactions in C and N cycling in a suburban native forest in the first two years of biochar application or three and half years of the recent prescribed burning. Generally, biochar always causes a short-term limited positive priming effect, but long-term field experiments about the impacts of biochar in urban forest ecosystems subjected to prescribed burning are rare (Bruckman et al. 2015; Mitchell et al. 2015; Page-Dumroese et al. 2016).
Materials and methods
Study area and experiment design
The study site was located in Toohey Forest, Brisbane, South Queensland, Australia (27032′53”S; 153003′21″E). This area is one of Australia’s native forests dominated by Eucalyptus species. Toohey forest is a subtropical climate area with an average annual temperature of 27 °C and an average annual rainfall of 1350 mm (Bai et al. 2012). Toohey forest has been subject to prescribed burning since 1993, and the risk of wildfire spread has been reduced by dividing areas for prescribed burning (Catterall et al. 2001; Wang et al. 2020a). The experimental site in this study was last burned in August 2017, and a field biochar application trial was established in this area in May 2019 after 20 months of prescribed burning. The test area was randomly placed in a site, which included a mixture of understorey legumes Acacia leiocalyx, Acacia disparismma and overstorey such as Eucalyptus psammitica, which is typical of the area.
In brief, the experimental design is a random complete block design, with 4 plots as 4 replicates. These four circular plots with a radius of 12.62 m, giving an internal area of 500 m2, were randomly located within the site, offering a total sampling area of one-fifth of a hectare. Each plot has 13 sub-samples, including two understorey legume species of Acacia with Eucalyptus planchoniana as a reference plant. Each plant area is defined by four steel pegs providing an area of 4 m2 (2 m × 2 m). Biochar was applied to each acacia plant at three rates of 0, 5 and 10 t ha−1. In each plot, there are 3 different treatment methods: 1) control (no biochar), 2) biochar 5 t ha−1 (2 kg/plant), and 3) biochar 10 t ha−1 (4 kg/plant), respectively. Biochar was manually scattered on the soil surface in May 2019. First, the grass in each plant area was manually removed, and the biochar was homogeneously distributed and mixed manually on the soil surface to minimise biochar loss. Soil samples were collected from the central area of each plant area after the removal of the litter layer to avoid edge effects.
Characterization and application of biochar
Biochar material used in this experiment was produced from pine wood (Pinus radiata) through slow pyrolysis at 600 °C introduced in Western Australia. The N isotope composition (δ15N) and C isotope composition (δ13C) of the chosen high-temperature biochar were 2.3‰ and − 27.5‰ respectively in this study. It is pertinent to note that we chose high-temperature biochar that was made above 500 °C because this study region is in a natural forest with high N deposition pollution (Bai et al. 2012). In terms of sustainability, the two biochar rates were selected in this study, as it has been confirmed and reported that 5 t ha−1 and 10 t ha−1 are ideal and economically feasible application rates (Williams and Arnott 2010; Bruckman et al. 2016). The biochar properties are summarized in Table S1.
Soil and foliage sample collection
After 24 months of the field establishment, soil samples were collected from three different depths of 0–5, 5–10 and 10–20 cm using a soil auger with a diameter of 7.5 cm at various points across each plant area in May 2021. After collecting soil and plant samples, the soil samples were sieved by using a 2 mm sieve and mixed thoroughly, then sub-sampled were collected for the following analyses: 1) air drying a part of the soil samples to analyse the soil pH value; 2) oven drying (60 °C) a part of soil samples for mass spectrometry analysis and determination of soil moisture content.
Similarly, plant samples (foliage) were oven-dried to a constant weight for 72 hours at 60 °C to a constant weight. After that, the dried foliage samples were ground into a fine powder using the Rocklabs™ ring grinder and weighted for mass spectrometry analyses. For foliage samples and oven-dried soil samples, we have evaluated the total C, total N, and their isotope composition (e.g., δ13C and δ15N) (Xu et al. 2000).
Data collection and measurements
Measurements of soil physiochemical properties
After collecting and processing soil samples, some initial soil characteristics were determined, such as soil moisture content and pH value. Soil moisture content (SMC) was determined by drying the field moist soil at 105 °C for 24 hours. The value of SMC was determined using the following equation (Voroney 2019):
The ratio of soil and water mass was 1:5 to determine soil pH. The 5 g of air-dried soil was taken into a 50 ml falcon and 25 ml of deionized water was added. The samples were then shaken in an end-to-end shaker for an hour, and then they were allowed to stand to settle down the solution for 20 minutes. After calibrating the machine with two buffer solutions, the soil pH was measured with a pH electrode.
Oven-dried soil samples were ground into fine powder by using the Rocklabs™ ring grinder. Then approximately 40–50 mg of soil (0–5 and 5–10 cm), about 50–60 mg (10–20 cm) and about 6–7 mg foliage samples were transferred into tin capsules for total C total N, δ13C and δ15N analyses by using the isotope ratio mass spectrometer (IRMS, Elementar, Langenselbold, Hesse, Germany).
The value of δ13C and δ15N were determined using the following two equations:
where,
- R:
-
the isotope ratio
- Rsample:
-
the ratio of 13C/12C and 15N/14N of sample respectively
- RVPDB:
-
the ratio of 13C/12C of the international standard (Vienna Pee Dee. Belemnite (VPDB))
- Rair:
-
the ratio of 15N/14N of the international standard (atmospheric N2)
The results for δ13C and δ15N were expressed as parts per thousand (‰).
Measurements of plant growth and physiochemical properties
Plant height and diameter at ground level (DGL) were measured in May 2021 at four plots by using diameter tape and height measurements hypsometer (Vertex IV), respectively. All understory plants, such as Acacia and Eucalyptus species for all plots, were included for growth measurements. Four plots contain an average of 52 plants, 48 plants for each Acacia spp. (A. leiocalyx = 24; A. disparimma = 24) and 4 reference plants of E. psammitica.
The percentage of N derived from atmospheric N2 (%Ndfa) was calculated using the following equation (Shearer and Kohl 1986).
where:
- δ15Nref:
-
The δ15N value of the reference plants
- δ15Nacacia:
-
The δ15N value of the Acacia spp.
- B value:
-
The relative isotopic abundance of Acacia spp. growing under an N-free nutrient condition
The B values reported from a large array of sources vary from −2.9‰ to 1.0‰ for woody plants (Shearer and Kohl 1986; Boddey et al. 2000). In the present study, using a B value of −1.3‰ resulted in an estimation of %Ndfa that was greater than 100%. Assuming B = −1.3‰ resulted in 75.0% of all cases having the %Ndfa greater than 100% (n = 30 out of 40), while this percentage dropped to 5.0% when B was assumed to be −0.8‰ (n = 2 out of 40). None of the cases were greater than 100% when B values were either −0.3‰ or 1.0‰. Therefore, different B values varying from −1.3‰ to 1.0‰ were examined to determine an applicable B value for Acacia spp. under the experimental condition. In our study, we chose a B value of −0.3‰ to report the results of BNF determination based on the 15N natural abundance method.
Statistical analyses
A general linear model was used to analyse treatment effects (SPSS 19.0, IBM Crop., Armonk, NY, US) (Bruckman et al. 2016). All data were tested for homogeneity of variance and normality of distribution. A three-way ANOVA was performed to test for these three factors (i.e., soil depths, biochar rates and plant species) and their interactions on each measured parameter. Pearson’s correlation analysis was used to assess relationships between soil properties across all data of three biochar rates at different soil depths. Statistical significance was set at α = 0.05.
Results
Chemical and physical properties of plant samples
The results from Table 1 show that after biochar application at the Toohey forest for 2 years, the foliar N isotope compositions (δ15N) and C isotope composition (δ13C) were significantly different between the two understorey acacia species (P < 0.05). The foliar δ15N of A. disparimma (−1.46‰) was significantly higher than that of A. leiocalyx (−2.01‰), and foliar δ13C of A. disparimma (−32.1‰) was also significantly higher than that of A. leiocalyx (−32.8‰) (Table 1). The foliar total N was also significantly increased by these two biochar application rates. The %Ndfa of A. disparimma was 58.0%, which was significantly higher than the %Ndfa of A. leiocalyx (37.4%) (P < 0.05) (Table 1).
There were no significant differences in plant height, diameter at ground level (DGL), basal area (BA) and volume between the three different biochar application rates and the two species (P > 0.05) (Table 2). Relative to the control, biochar addition increased plant growth in natural forests and biochar applied at a rate of 10 t ha−1 led to the greatest improvement in plant growth (0.05 < P < 0.10). Further details are documented in the Supplementary Material. Table S3 provides the interactive effects of the biochar application rates and species on plant height, diameter at ground level (DGL), basal area (BA) and volume.
Soil chemical properties
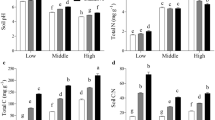
As the soil depth increased, soil total C was 6.81%, 4.77%, and 3.27% for 0–5, 5–10 and 10–20 cm respectively, while the corresponding soil total N was 0.250%, 0.168% and 0.106% respectively, significantly decreased as the soil depth increased (P < 0.05) (Table 3). Soil δ13C values were − 27.10‰, −25.86‰ and − 25.63‰ respectively for the 0–5, 5–10 and 10–20 cm soil depths (Table 3). For soil δ15N from 5 to 10 cm depth (1.044‰) was significantly higher than those of the other two soil depths (P < 0.05) (Table 3). The results of Table 3 also show that soil total C and total N were significantly higher only when the biochar application rate was 10 t/ha (P < 0.05) (Table 3).
After two years of biochar application, the soil moisture content of the biochar application rate of 10 t/ha was 14.3% and 13.8% higher than that under the control treatment at 0–5 cm and 5–10 cm soil depth (0.05 < P < 0.10) (Table 4). At the 0–5 cm soil, the results of Table 4 also showed that soil total C for the biochar application rate at 10 t/ha was significantly higher than that of the biochar application rate at 0 t/ha (P < 0.05). The soil total N at 5 and 10 t/ha biochar application rates (0.262% and 0.265%) were higher than that of without biochar application (0.230%) (Table 4).
Relationship plant physiological variables
Regression analysis showed that there was a significant positive relationship between foliar δ15N (‰) and plant BNF capacity, which depended on different acacia species. It has been shown from Fig. 1 that Acacia disparrima foliar δ15N was more strongly related to plant BNF capacity (R2 = 0.863, P < 0.001) than that of Acacia leiocalyx (R2 = 0.661, P < 0.001).
Linear relationship between foliar δ15N (‰) and biological nitrogen fixation rate (% Ndfa) of Acacia leiocalyx and Acacia disparrima. This relationship represents the variation of two acacia species and shows the contribution of foliar δ15N in changing the BNF rate of Acacia leiocalyx and Acacia disparrima respectively
Relationships between soil physiological variables
In the Toohey Forest, it has been shown from Fig. 2, soil δ13C was positively related to soil δ15N at soil depths of 5–10 cm, and soil δ13C was strongly related to soil δ15N on biochar rate of 10 t/ha (R2 = 0.641, P < 0.001) than those of biochar rates of 5 t/ha (R2 = 0.388, P = 0.01) (Fig. 2). There were strong correlations between soil total N and soil δ15N at soil depth of 0–5 and 5–10 cm, but the relationship depended on the biochar application rates (Fig. 3). At soil depth of 0–5 cm, there was a tight non-linear relationship between soil total N and soil δ15N (R2 = 0.424, P < 0.05) when the biochar application rate is 5 t/ha (Fig. 3a). In the 5–10 cm soil layer, the correlations between soil total N and soil δ15N became stronger and corresponding peaks also move backward as the biochar application rates increased (Fig. 3b).
In addition, a single regression analysis explained the non-linear relationship between soil moisture content and soil δ15N at soil depth of 5–10 cm (R2 = 0.407, P < 0.001) (Fig. 4). Biochar application rate also affects the correlation between soil moisture content and soil δ15N. It could be seen from Fig. 5 that when the biochar application rate was 10 t/ha, the non-linear relationship between soil moisture content and soil δ15N (R2 = 0.750, P < 0.001) was stronger than those of the biochar application rates of 0 and 5 t/ha (R2 = 0.197, P = 0.08; R2 = 0.377, P = 0.046).
Discussions
The effect of N deposition and biochar application on plants
The negative δ15N values in the surface soil and foliage observed in this study are unusual for native forests (Tables 1, 3 and 4), as the isotope discrimination usually demonstrates a level of 15N enrichment in soil and plant (Robinson 2001; Blumfield et al. 2006; Burton et al. 2007; Murray 2013; Reis et al. 2017). In this study, the negative soil δ15N may result from background soil in the Toohey forest near a busy highway, being affected by atmospheric N deposition. Because the δ15N in NOx emitted from vehicles ranges from −13‰ to −2‰ (Heaton 1990; Zong et al. 2020), this resulted in the negative soil δ15N we observed in this experiment. Meanwhile, high N availability would inhibit the BNF of forest systems (Marcarelli and Wurtsbaugh 2007; Barron et al. 2009; Avila Clasen et al. 2023). It could be seen from the results of Table 1 that for the two selected legumes, Acacia leiocalyx and Acacia disparrima, BNF of Acacia disparrima was significantly higher than that of Acacia leiocalyx. It can also be seen in Fig. 1 that the linear relationship between foliar δ15N and BNF was also closer in Acacia disparrima. In the previous studies, the higher BNF in Acacia leiocalyx may be due to its location in less fertile soil, which provided more N for photosynthesis (Bai et al. 2012; Taresh et al. 2021). In this study, in the N deposition soil, as can be seen from Table 1, the total C value of Acacia leiocalyx was significantly lower than that of Acacia disparrima, indicating that Acacia leiocalyx grew faster than Acacia disparrima, and the rhizome system was also more developed than that of Acacia disparrima. At the same time, plants are more inclined to preferentially absorb N resources that are more readily available (from N deposition) and do not need to consume much energy (Gurmesa et al. 2016; Xie et al. 2021). From what has been observed in other ecosystems, increased N availability (such as N deposition) may inhibit BNF in forest systems (Compton et al. 2004; Saiz et al. 2021). Therefore, the BNF of Acacia leiocalyx was relatively lower in this study, which is consistent with the results obtained in other studies. Meanwhile, under high N deposition, biochar applications in this study significantly increased the total N of Acacia foliage (Table 1). Several studies have shown that biochar improves N dynamics in soil, thereby improving plant growth and yield and having a positive impact on plant nutrient content, which may increase plant N uptake and utilization (Rondon et al. 2007; Uzoma et al. 2011; Qian et al. 2019; Ali et al. 2020).
Meanwhile, in the previous studies, Acacia leiocalyx generally showed a higher δ13C value than that of Acacia disparrima, suggesting higher water use efficiency. Additionally, within plant species, there is a genetic component to the distinction in the δ13C value, which may be as great as 3‰ (Tieszen 1991; Audiard et al. 2018; Hussain et al. 2022), and the higher δ13C value of Acacia leiocalyx indicated that it has a better water use strategy under drought stress (Raddad and Luukkanen 2006; Taresh et al. 2021). In contrast, in this study, Acacia leiocalyx showed a significantly lower δ13C value, so it can be inferred that the forest soil is not restricted by moisture, and the water can be used freely by plants. In contrast, foliar δ15N of Acacia leiocalyx with much lower BNF in this study suggests that the 15N signature of deposition N can alter plant δ15N by direct uptake in the canopy and by altering the signature of available N in the soil. In contrast, the total inorganic N in deposition was 15N-depleted (−10‰) (Craine et al. 2015). The addition of N due to N depositional contamination resulted in the δ15N of plants towards the 15N signature of the added N, indicating the incorporation of added N into plants. Thus, foliar δ15N values were lower for Acacia leiocalyx, which is more likely to retain and take N from N deposition, than for Acacia disparrima. This again highlights the importance of the 15N signature of input N in controlling ecosystem δ15N.
The effect of N deposition on soil C and N isotope compositions
The δ13C and δ15N in the soil can be used as an overall indicator of soil environmental processes in forest ecosystems, thereby providing comprehensive insights into the long-term changes of soil C and N cycles (Amelung et al. 2008; Brunn et al. 2014; Guillaume et al. 2015; Wang et al. 2018). In the entire ecosystem, soil δ15N has been used as a potential indicator of soil N status (Schulze et al. 1998; Craine et al. 2009; Ladd et al. 2010; Zhang et al. 2022). They usually accumulate as soil N availability increases, and human disturbance and climatic factors will change the N cycle process in forest soil (Pardo et al. 2002; Wang et al. 2014; Mgelwa et al. 2019).
In the previous studies, soil δ15N and δ13C values usually increase with increasing depth (Eshetu 2004; Schneider et al. 2021). In this study, soil δ13C was significantly enriched with the increase of soil depth, which may be because deeper soil layers have greater decomposition and humification of organic matter or mainly increase the residence time of organic C in the soil (Bird et al. 2002; Lorenz et al. 2020), which is consistent with the previous research results (Brunn et al. 2014). The value of surface soil δ13C was close to the δ13C of the applied biochar in this study, which may be because of surface-applied biochar on soil C pool (Table 3). However, in this study, with the increase of soil depth, the changing trend of soil δ15N value is different from the results of the previous studies. The negative δ15N value of the surface soil may be the result of atmospheric N deposition because this forest is close to a busy highway. The δ15N of NOx emitted by vehicles could lead to the negative soil 15N values observed in this study at soil depths of 0–5 and 10–20 cm (Savard et al. 2009; Su et al. 2020). However, soil δ15N value showed an abnormally positive value (1.044‰) at 5–10 cm soil depth, which was significantly higher than those of the other two depths. In these N-rich ecosystems, the fate of the deposited N may be different, and most of the deposited N may be directly lost in humid climates (Amundson et al. 2003; Templer et al. 2012; Rivero-Villar et al. 2021). The forest system in this study has a large amount of N losses via denitrification and N leaching, so there is a large amount of N loss at a soil depth of 5-10 cm, which is consistent with the results of previous studies (Fang et al. 2008; Liu et al. 2023). In addition, we found that soil C and N content decreased with increasing soil depth as expected.
Responses of the relationship between soil moisture content and soil δ15N to biochar application
The critical role of soil moisture content in N leaching and denitrification is demonstrated from the non-linear relationship between soil δ15N and SMC in this study (Fig. 4), where higher SMC drives more soil N loss initially due to soil nitrification since N transformations such as nitrification, denitrification and N mineralization produce mobile, 15N-deleted N, it is easy to enrich soil δ 15N by leaching or gaseous emission of soil N (Bai et al. 2013). However, the soil N loss rate slows as the soil moisture content continues to increase. The nitrification rates decline under these conditions since oxygen is essential for nitrification (Zhu et al. 2011; Ouyang et al. 2017). Meanwhile, the heavy rainfall event before sampling significantly increased leachate volume in the soil in this study (Yang et al. 2015). With increasing biochar application rate (10 t/ha), a stronger non-linear relationship between soil δ15N enrichment and soil moisture content could be found in our study (Fig. 5). The reason for this is that applying biochar with a porous structure and higher surface area to the soil in this study, the ability of the soil to absorb and retain more water can be improved, thus providing more moisture to the soil-plant system (Zhang et al. 2008; Li et al. 2018). Moreover, it can also be seen in Table 2 that with the biochar application, the soil total N content was higher. The regression relationship between soil total N and δ15N was also closer, and the enrichment of soil δ15N slows down (Fig. 3). This may be because biochar addition can reduce nitrate leaching by retain more soil water and total N loss from forest soils by modifying the soil characteristics (Kanthle et al. 2016; Xu et al. 2016; Sun et al. 2018).
In Table 4, soil moisture content from the 0–5 and 5–10 cm soil layers increased with the increasing biochar application rates. This can be attributed to the fact that the soil was added with biochar, which has a highly porous structure to retain water physically (Atkinson et al. 2010; Kang et al. 2022). Meanwhile, water movement down into the deep soil is called infiltration. The application of biochar leads to the creation of more pores in the soil matrix and the formation of tortuous interstitial spaces between the soil and the biochar particles, thereby increasing the moisture penetration rate, which is consistent with previous research results (Ajayi et al. 2016; Liu et al. 2016).
Responses of soil δ13C and δ15N to biochar application
Previous studies showed that biochar application increased soil cation exchange capacity and absorbed N compounds, thereby reducing NH4+ leaching (Glaser et al. 2002; Liang et al. 2006; Sun et al. 2017). The influence of biochar applications on soil N cycling is especially complex and may have a significant impact on soil N transformation processes by altering soil microbial activities and community structures (Streubel et al. 2011; Song et al. 2014). It can be seen from Fig. 3 that at soil depths of 0–5 cm and 5–10 cm, the addition of biochar was an important adjustment factor for soil N loss on the corresponding variability of biochar. Significant differences were observed in this study. With the increase in the level of biochar addition, the influence of biochar on soil δ15N increased significantly (Fig. 3). The reason may be that the biochar addition can promote soil N transformation by increasing the abundance of soil ammonia-oxidizing bacteria and the activity of nitrifying bacteria, while retaining NH4+ through its acidic functional groups, thus offsetting the rapid soil N loss from biochar amended with soil N transformations (Zheng et al. 2013; Zhang et al. 2020). At the same time, as can be seen from Fig. 3b with the gradual increase in the biochar application rate, the trend of soil N loss gradually flattened, and the apex of the parabola appears later with the biochar addition of 5 t/ha. The results of the study are consistent (Cao et al. 2014). When the biochar application rate reached 10 t/ha, the observed positive correlation between the soil δ13C and δ15N was tighter (Fig. 2), which also illustrated that biochar application could improve the activity and structure of soil microorganisms, thereby providing new information on soil C and N turnover controlled by biochar application rates (Dijkstra et al. 2006; Werth and Kuzyakov 2010; Gerschlauer et al. 2019). These results highlight the importance of appropriate biochar application rates in soil N improvement.
Compared with the treatment without biochar application, a significant increase in the soil total C and total N content was observed under biochar application, especially for the surface soil (Tables 3 and 4), which is consistent with the results of previous studies (Qiao-Hong et al. 2014). It was shown that the biochar application improved soil characteristics. In addition to soil samples, the total N content of foliage also increased significantly under biochar application (Table 1). These results are consistent with previous studies (Ibrahim et al. 2020). Among them, the biochar application has been proven to increase N uptake in plant and soil N content (Fiorentino et al. 2019; Wang et al. 2020b). Previous studies have proposed that biochar application generates a large amount of ion exchange capacity and improves the adsorption and immobilization of N forms required for plant uptake. When these N forms are adsorbed by biochar, they can be released again and become available for plant absorption and utilization (Xue et al. 2017), thus improving N use efficiency. Applying biochar to soil is a potential way to improve the bioavailability of nutrients and reduce soil nutrient loss (Lehmann et al. 2003; Shen et al. 2016).
Conclusions
The N deposition existing in urban forest soils had an impact on the internal N cycle of the soil-plant system, and the resulting high N availability significantly inhibited the BNF capacity of the forest system. The biochar application significantly reduced N losses due to leaching and denitrification, and significantly increased the potential of soil to retain N. This also improved N uptake by plants and N utilization efficiency. In this study, when the biochar application rate was applied at 10 t/ha, it had the best effect on improving soil N transformations while reducing soil N loss as well as improved plant growth.
References
Ajayi AE, Holthusen D, Horn R (2016) Changes in microstructural behaviour and hydraulic functions of biochar amended soils. Soil Till Res 155:166–175. https://doi.org/10.1016/j.still.2015.08.007
Ali I, He L, Ullah S, Quan Z, Wei S, Iqbal A, Munsif F, Shah T, Xuan Y, Luo Y (2020) Biochar addition coupled with nitrogen fertilization impacts on soil quality, crop productivity, and nitrogen uptake under double-cropping system. Food Energy Secur 9:e208
Ameloot N, Sleutel S, Das K, Kanagaratnam J, De Neve S (2015) Biochar amendment to soils with contrasting organic matter level: effects on N mineralization and biological soil properties. Glob Change Biol Bioenergy 7:135–144
Amelung W, Brodowski S, Sandhage-Hofmann A, Bol R (2008) Combining biomarker with stable isotope analyses for assessing the transformation and turnover of soil organic matter. Adv Agron 100:155–250
Amundson R, Austin A, Schuur E, Yoo K, Matzek V, Kendall C, Uebersax A, Brenner DL, Baisden T (2003) Global patterns of the isotopic composition of soil and plant nitrogen. Global Biogeochem Cycl 17:31. https://doi.org/10.1029/2002GB001903
Aryal B, Gurung R, Camargo AF, Fongaro G, Treichel H, Mainali B, Angove MJ, Ngo HH, Guo W, Puadel SR (2022) Nitrous oxide emission in altered nitrogen cycle and implications for climate change. Environ Pollut 314:120272. https://doi.org/10.1016/j.envpol.2022.120272
Asadyar L, Xu C-Y, Wallace HM, Xu Z, Reverchon F, Bai SH (2021) Soil-plant nitrogen isotope composition and nitrogen cycling after biochar applications. Environ Sci Pollut Res 28:6684–6690. https://doi.org/10.1007/s11356-020-11016-3
Atkinson CJ, Fitzgerald JD, Hipps NA (2010) Potential mechanisms for achieving agricultural benefits from biochar application to temperate soils: a review. Plant Soil 337:1–18. https://doi.org/10.1007/s11104-010-0464-5
Audiard B, Blasco T, Brossier B, Fiorentino G, Battipaglia G, Théry-Parisot I (2018) δ13C referential in three Pinus species for a first archaeological application to Paleolithic contexts:“Between intra-and inter-individual variation and carbonization effect”. J Archaeol Sci Rep 20:775–783
Avila Clasen L, Permin A, Horwath AB, Metcalfe DB, Rousk K (2023) Do nitrogen and phosphorus additions affect nitrogen fixation associated with tropical mosses? Plants 12:1443
Bai SH, Sun F, Xu Z, Blumfield TJ, Chen C, Wild C (2012) Appraisal of 15N enrichment and 15N natural abundance methods for estimating N2 fixation by understorey Acacia leiocalyx and A. Disparimma in a native forest of subtropical Australia. J Soils Sediments 12:653–662
Bai E, Boutton TW, Liu F, Wu XB, Archer SR (2013) 15N isoscapes in a subtropical savanna parkland: spatial-temporal perspectives. Ecosphere 4:1–17
Bai SH, Blumfield T, Xu Z (2014) Survival, growth and physiological status of Acacia disparrima and Eucalyptus crebra seedlings with respect to site management practices in Central Queensland, Australia. Eur J For Res 133:165–175
Bai SH, Omidvar N, Gallart M, Kämper W, Tahmasbian I, Farrar MB, Singh K, Zhou G, Muqadass B, Xu CY, Koech R, Li Y, Nguyen TTN, Van Zwieten L (2022) Combined effects of biochar and fertilizer applications on yield: a review and meta-analysis. Sci Total Environ 808:152073. https://doi.org/10.1016/j.scitotenv.2021.152073
Barron AR, Wurzburger N, Bellenger JP, Wright SJ, Kraepiel AM, Hedin LO (2009) Molybdenum limitation of asymbiotic nitrogen fixation in tropical forest soils. Nat Geosci 2:42–45
Biederman LA, Harpole WS (2013) Biochar and its effects on plant productivity and nutrient cycling: a meta-analysis. GCB Bioenergy 5:202–214
Binkley D, Högberg P (1997) Does atmospheric deposition of nitrogen threaten Swedish forests? For Ecol Manag 92:119–152. https://doi.org/10.1016/S0378-1127(96)03920-5
Bird M, Santruckova H, Lloyd J, Lawson E (2002) The isotopic composition of soil organic carbon on a north–south transect in western Canada. Eur J Soil Sci 53:393–403
Blumfield TJ, Xu Z, Prasolova N, Mathers NJ (2006) Effect of overlying windrowed harvest residues on soil carbon and nitrogen in hoop pine plantations of subtropical Australia. J Soils Sediments 6:243–248
Bobbink R, Hicks K, Galloway J, Spranger T, Alkemade R, Ashmore M, Bustamante M, Cinderby S, Davidson E, Dentener F, Emmett B, Erisman JW, Fenn M, Gilliam F, Nordin A, Pardo L, De Vries W (2010) Global assessment of nitrogen deposition effects on terrestrial plant diversity: a synthesis. Ecol Appl 20:30–59. https://doi.org/10.1890/08-1140.1
Boddey RM, Peoples MB, Palmer B, Dart PJ (2000) Use of the 15N natural abundance technique to quantify biological nitrogen fixation by woody perennials. Nutr Cycl Agroecosyst 57:235–270. https://doi.org/10.1023/A:1009890514844
Bruckman VJ, Terada T, Uzun BB, Apaydın-Varol E, Liu J (2015) Biochar for climate change mitigation: tracing the in-situ priming effect on a forest site. Energy Procedia 76:381–387
Bruckman V, Klinglmüller M, Milenković M (2016) Biochar in the view of climate change mitigation: the FOREBIOM experience. In: Bruckman V (ed) Biochar: a regional supply chain approach in view of climate change mitigation. Cambridge University, pp 1–22
Brunn M, Spielvogel S, Sauer T, Oelmann Y (2014) Temperature and precipitation effects on δ13C depth profiles in SOM under temperate beech forests. Geoderma 235:146–153
Burrell LD, Zehetner F, Rampazzo N, Wimmer B, Soja G (2016) Long-term effects of biochar on soil physical properties. Geoderma 282:96–102
Burton J, Chen C, Xu Z, Ghadiri H (2007) Gross nitrogen transformations in adjacent native and plantation forests of subtropical Australia. Soil Biol Biochem 39:426–433
Cao CT, Farrell C, Kristiansen PE, Rayner JP (2014) Biochar makes green roof substrates lighter and improves water supply to plants. Ecol Eng 71:368–374
Case SD, Mcnamara NP, Reay DS, Stott AW, Grant HK, Whitaker J (2015) Biochar suppresses N2O emissions while maintaining N availability in a sandy loam soil. Soil Biol Biochem 81:178–185
Catterall CP, Piper SD, Bunn SE, Arthur JM (2001) Flora and fauna assemblages vary with local topography in a subtropical eucalypt forest. Austral Ecol 26:56–69
Cecchini G, Andreetta A, Marchetto A, Carnicelli S (2021) Soil solution fluxes and composition trends reveal risks of nitrate leaching from forest soils of Italy. CATENA 200:105175. https://doi.org/10.1016/j.catena.2021.105175
Cheng Y, Wang J, Wang J, Wang S, Chang SX, Cai Z, Zhang J, Niu S, Hu S (2020) Nitrogen deposition differentially affects soil gross nitrogen transformations in organic and mineral horizons. Earth-Sci Rev 201:103033. https://doi.org/10.1016/j.earscirev.2019.103033
Compton JE, Watrud LS, Arlene Porteous L, Degrood S (2004) Response of soil microbial biomass and community composition to chronic nitrogen additions at Harvard forest. For Ecol Manag 196:143–158. https://doi.org/10.1016/j.foreco.2004.03.017
Conley DJ, Paerl HW, Howarth RW, Boesch DF, Seitzinger SP, Havens KE, Lancelot C, Likens GE (2009) Controlling eutrophication: nitrogen and phosphorus. Science 323:1014–1015. https://doi.org/10.1126/science.1167755
Craine JM, Elmore AJ, Aidar MP, Bustamante M, Dawson TE, Hobbie EA, Kahmen A, Mack MC, Mclauchlan KK, Michelsen A (2009) Global patterns of foliar nitrogen isotopes and their relationships with climate, mycorrhizal fungi, foliar nutrient concentrations, and nitrogen availability. New Phytol 183:980–992
Craine JM, Brookshire E, Cramer MD, Hasselquist NJ, Koba K, Marin-Spiotta E, Wang L (2015) Ecological interpretations of nitrogen isotope ratios of terrestrial plants and soils. Plant Soil 396:1–26
Cross A, Sohi SP (2011) The priming potential of biochar products in relation to labile carbon contents and soil organic matter status. Soil Biol Biochem 43:2127–2134
Das SK, Ghosh GK, Avasthe R (2021) Applications of biomass derived biochar in modern science and technology. Environ Technol Innov 21:101306
Das SK, Ghosh GK, Avasthe R (2023) Biochar application for environmental management and toxic pollutant remediation. Biomass Convers Biorefinery 13:555–566. https://doi.org/10.1007/s13399-020-01078-1
Deluca TH, Gundale MJ, Mackenzie MD, Jones DL (2015) Biochar effects on soil nutrient transformations. Biochar Environ Manag: Sci Technol Implement 2:421–454
Dijkstra P, Ishizu A, Doucett R, Hart SC, Schwartz E, Menyailo OV, Hungate BA (2006) 13C and 15N natural abundance of the soil microbial biomass. Soil Biol Biochem 38:3257–3266
Ding Z, Hu X, Wan Y, Wang S, Gao B (2016) Removal of lead, copper, cadmium, zinc, and nickel from aqueous solutions by alkali-modified biochar: batch and column tests. J Ind Eng Chem 33:239–245
Eshetu Z (2004) Natural 15N abundance in soils under young-growth forests in Ethiopia. For Ecol Manag 187:139–147. https://doi.org/10.1016/S0378-1127(03)00315-3
Fang YT, Gundersen P, Mo JM, Zhu WX (2008) Input and output of dissolved organic and inorganic nitrogen in subtropical forests of South China under high air pollution. Biogeosci 5:339–352. https://doi.org/10.5194/bg-5-339-2008
Farrar MB, Wallace HM, Xu CY, Joseph S, Nguyen TTN, Dunn PK, Bai SH (2022) Biochar compound fertilisers increase plant potassium uptake 2 years after application without additional organic fertiliser. Environ Sci Pollut Res 29:7170–7184. https://doi.org/10.1007/s11356-021-16236-9
Fiorentino N, Sánchez-Monedero M, Lehmann J, Enders A, Fagnano M, Cayuela M (2019) Interactive priming of soil N transformations from combining biochar and urea inputs: a 15N isotope tracer study. Soil Biol Biochem 131:166–175
Fowler D, Coyle M, Skiba U, Sutton MA, Cape JN, Reis S, Sheppard LJ, Jenkins A, Grizzetti B, Galloway JN (2013) The global nitrogen cycle in the twenty-first century. Philos Trans R Soc B Biol Sci 368:20130164
Francos M, Úbeda X (2021) Prescribed fire management. Curr Opin Environ Sci Health 21:100250. https://doi.org/10.1016/j.coesh.2021.100250
Galloway JN, Townsend AR, Erisman JW, Bekunda M, Cai Z, Freney JR, Martinelli LA, Seitzinger SP, Sutton MA (2008) Transformation of the nitrogen cycle: recent trends, questions, and potential solutions. Science 320:889–892
Gerschlauer F, Saiz G, Schellenberger Costa D, Kleyer M, Dannenmann M, Kiese R (2019) Stable carbon and nitrogen isotopic composition of leaves, litter, and soils of various ecosystems along an elevational and land-use gradient at Mount Kilimanjaro, Tanzania. Biogeosci 16:409–424
Glaser B, Lehmann J, Zech W (2002) Ameliorating physical and chemical properties of highly weathered soils in the tropics with charcoal–a review. Biol Fertil Soils 35:219–230
Guillaume T, Damris M, Kuzyakov Y (2015) Losses of soil carbon by converting tropical forest to plantations: erosion and decomposition estimated by δ13C. Glob Change Biol 21:3548–3560
Guinto DF, Xu Z, House AP, Saffigna PG (2000) Assessment of N2 fixation by understorey acacias in recurrently burnt eucalypt forests of subtropical Australia using 15N isotope dilution techniques. Can J For Res 30:112–121
Gundale MJ, Nilsson M-C, Pluchon N, Wardle DA (2016) The effect of biochar management on soil and plant community properties in a boreal forest. GCB Bioenergy 8:777–789. https://doi.org/10.1111/gcbb.12274
Gurmesa GA, Lu X, Gundersen P, Mao Q, Zhou K, Fang Y, Mo J (2016) High retention of 15N-labeled nitrogen deposition in a nitrogen saturated old-growth tropical forest. Glob Change Biol 22:3608–3620
Gurwick NP, Moore LA, Kelly C, Elias P (2013) A systematic review of biochar research, with a focus on its stability in situ and its promise as a climate mitigation strategy. PLoS One 8:e75932
Haldar D, Purkait MK (2021) A review on the environment-friendly emerging techniques for pretreatment of lignocellulosic biomass: mechanistic insight and advancements. Chemosphere 264:128523
Heaton T (1990) 15N/14N ratios of NOx from vehicle engines and coal-fired power stations. Tellus Ser B Chem Phys Meteorol 42:304–307
Hietz P, Turner Benjamin L, Wanek W, Richter A, Nock Charles A, Wright SJ (2011) Long-term change in the nitrogen cycle of tropical forests. Science 334:664–666. https://doi.org/10.1126/science.1211979
Huang Y, Kang R, Mulder J, Zhang T, Duan L (2015) Nitrogen saturation, soil acidification, and ecological effects in a subtropical pine forest on acid soil in Southwest China. J Geophys Res Biogeosci 120:2457–2472. https://doi.org/10.1002/2015JG003048
Hussain MI, Khan ZI, Farooq TH, Al Farraj DA, Elshikh MS (2022) Comparative plasticity responses of stable isotopes of carbon (δ13C) and nitrogen (δ15N), ion homeostasis and yield attributes in barley exposed to saline environment. Plants 11:1516
Ibell PT, Xu Z, Blumfield TJ (2013) The influence of weed control on foliar δ15N, δ13C and tree growth in an 8 year-old exotic pine plantation of subtropical Australia. Plant Soil 369:199–217. https://doi.org/10.1007/s11104-012-1554-3
Ibrahim MM, Tong C, Hu K, Zhou B, Xing S, Mao Y (2020) Biochar-fertilizer interaction modifies N-sorption, enzyme activities and microbial functional abundance regulating nitrogen retention in rhizosphere soil. Sci Total Environ 739:140065
Kang MW, Yibeltal M, Kim YH, Oh SJ, Lee JC, Kwon EE, Lee SS (2022) Enhancement of soil physical properties and soil water retention with biochar-based soil amendments. Sci Total Environ 836:155746. https://doi.org/10.1016/j.scitotenv.2022.155746
Kanthle AK, Lenka NK, Lenka S, Tedia K (2016) Biochar impact on nitrate leaching as influenced by native soil organic carbon in an Inceptisol of Central India. Soil Till Res 157:65–72
Klaus VH, Hölzel N, Prati D, Schmitt B, Schöning I, Schrumpf M, Solly EF, Hänsel F, Fischer M, Kleinebecker T (2016) Plant diversity moderates drought stress in grasslands: implications from a large real-world study on 13C natural abundances. Sci Total Environ 566:215–222
Kuzyakov Y, Xu X (2013) Competition between roots and microorganisms for nitrogen: mechanisms and ecological relevance. New Phytol 198:656–669. https://doi.org/10.1111/nph.12235
Kuzyakov Y, Subbotina I, Chen H, Bogomolova I, Xu X (2009) Black carbon decomposition and incorporation into soil microbial biomass estimated by 14C labeling. Soil Biol Biochem 41:210–219. https://doi.org/10.1016/j.soilbio.2008.10.016
Ladd B, Pepper D, Bonser S (2010) Competition intensity at local versus regional spatial scales. Plant Biol 12:772–779
Lan Z, Chen C, Rezaei Rashti M, Yang H, Zhang D (2018) High pyrolysis temperature biochars reduce nitrogen availability and nitrous oxide emissions from an acid soil. GCB Bioenergy 10:930–945
Lehmann J (2007) Bio-energy in the black. Front Ecol Environ 5:381–387. https://doi.org/10.1890/1540-9295(2007)5[381:BITB]2.0.CO;2
Lehmann J, Pereira Da Silva J, Steiner C, Nehls T, Zech W, Glaser B (2003) Nutrient availability and leaching in an archaeological Anthrosol and a Ferralsol of the Central Amazon basin: fertilizer, manure and charcoal amendments. Plant Soil 249:343–357. https://doi.org/10.1023/A:1022833116184
Lehmann J, Rillig MC, Thies J, Masiello CA, Hockaday WC, Crowley D (2011) Biochar effects on soil biota–a review. Soil Biol Biochem 43:1812–1836
Li S, Zhang Y, Yan W, Shangguan Z (2018) Effect of biochar application method on nitrogen leaching and hydraulic conductivity in a silty clay soil. Soil Till Res 183:100–108
Liang B, Lehmann J, Solomon D, Kinyangi J, Grossman J, O'neill B, Skjemstad JO, Thies J, Luizão FJ, Petersen J (2006) Black carbon increases cation exchange capacity in soils. Soil Sci Soc Am J 70:1719–1730
Liang B, Lehmann J, Sohi SP, Thies JE, O’neill B, Trujillo L, Gaunt J, Solomon D, Grossman J, Neves EG, Luizão FJ (2010) Black carbon affects the cycling of non-black carbon in soil. Org Geochem 41:206–213. https://doi.org/10.1016/j.orggeochem.2009.09.007
Liu Z, Dugan B, Masiello CA, Barnes RT, Gallagher ME, Gonnermann H (2016) Impacts of biochar concentration and particle size on hydraulic conductivity and DOC leaching of biochar–sand mixtures. J Hydrol 533:461–472. https://doi.org/10.1016/j.jhydrol.2015.12.007
Liu X, Wang H, Liu C, Sun B, Zheng J, Bian R, Drosos M, Zhang X, Li L, Pan G (2021) Biochar increases maize yield by promoting root growth in the rainfed region. Arch Agron Soil Sci 67:1411–1424. https://doi.org/10.1080/03650340.2020.1796981
Liu X, Zhang Y, Xiao T, Li P, Zhang L, Liu Y, Deng W (2023) Runoff velocity controls soil nitrogen leaching in subtropical restored forest in southern China. For Ecol Manag 548:121412. https://doi.org/10.1016/j.foreco.2023.121412
Lorenz M, Derrien D, Zeller B, Udelhoven T, Werner W, Thiele-Bruhn S (2020) The linkage of 13C and 15N soil depth gradients with C: N and O: C stoichiometry reveals tree species effects on organic matter turnover in soil. Biogeochemistry 151:203–220. https://doi.org/10.1007/s10533-020-00721-3
Luo L, Wang J, Lv J, Liu Z, Sun T, Yang Y, Zhu Y-G (2023) Carbon sequestration strategies in soil using biochar: advances, challenges, and opportunities. Environ Sci Technol 57:11357–11372. https://doi.org/10.1021/acs.est.3c02620
Mao J, Mao Q, Gundersen P, Gurmesa GA, Zhang W, Huang J, Wang S, Li A, Wang Y, Guo Y, Liu R, Mo J, Zheng M (2022) Unexpected high retention of 15N-labeled nitrogen in a tropical legume forest under long-term nitrogen enrichment. Glob Change Biol 28:1529–1543. https://doi.org/10.1111/gcb.16005
Marcarelli AM, Wurtsbaugh WA (2007) Effects of upstream lakes and nutrient limitation on periphytic biomass and nitrogen fixation in oligotrophic, subalpine streams. Freshw Biol 52:2211–2225. https://doi.org/10.1111/j.1365-2427.2007.01851.x
May B, Attiwill P (2003) Nitrogen-fixation by Acacia dealbata and changes in soil properties 5 years after mechanical disturbance or slash-burning following timber harvest. For Ecol Manag 181:339–355
Mgelwa AS, Hu Y-L, Liu J-F, Qiu Q, Liu Z, Ngaba MJY (2019) Differential patterns of nitrogen and δ15N in soil and foliar along two urbanized rivers in a subtropical coastal city of southern China. Environ Pollut 244:907–914
Mitchell PJ, Simpson AJ, Soong R, Simpson MJ (2015) Shifts in microbial community and water-extractable organic matter composition with biochar amendment in a temperate forest soil. Soil Biol Biochem 81:244–254
Moldan F, Wright RF (2011) Nitrogen leaching and acidification during 19 years of NH4NO3 additions to a coniferous-forested catchment at Gårdsjön, Sweden (NITREX). Environ Pollut 159:431–440. https://doi.org/10.1016/j.envpol.2010.10.025
Muqaddas B, Chen C, Lewis T, Wild C (2016) Temporal dynamics of carbon and nitrogen in the surface soil and forest floor under different prescribed burning regimes. For Ecol Manag 382:110–119
Murray U (2013) Isotope discrimination provides new insight into biological nitrogen fixation. New Phytol 198:643–646
Nguyen T, Xu C, Tahmasbian I, Che R, Xu Z, Zhou X, Wallace H, Hosseini Bai S (2017) Effects of biochar on soil available inorganic nitrogen: a review and meta-analysis. Geoderma 288. https://doi.org/10.1016/j.geoderma.2016.11.004
Nogués I, Rumpel C, Sebilo M, Vaury V, Moral R, Bustamante MA (2023) Stable C and N isotope variation during anaerobic digestate composting and in the compost-amended soil-plant system. J Environ Manag 329:117063. https://doi.org/10.1016/j.jenvman.2022.117063
Ouyang W, Xu X, Hao Z, Gao X (2017) Effects of soil moisture content on upland nitrogen loss. J Hydrol 546:71–80
Page-Dumroese DS, Coleman MD, Thomas SC (2016) Opportunities and uses of biochar on forest sites in North America. Biochar: Reg Supply Chain Approach View Mitigating Clim Chang 15:315–336
Palansooriya KN, Ok YS, Awad YM, Lee SS, Sung J-K, Koutsospyros A, Moon DH (2019) Impacts of biochar application on upland agriculture: a review. J Environ Manag 234:52–64. https://doi.org/10.1016/j.jenvman.2018.12.085
Pardo L, Hemond H, Montoya J, Fahey T, Siccama T (2002) Response of the natural abundance of 15N in forest soils and foliage to high nitrate loss following clear-cutting. Can J For Res 32:1126–1136
Phoenix GK, Hicks WK, Cinderby S, Kuylenstierna JCI, Stock WD, Dentener FJ, Giller KE, Austin AT, Lefroy RDB, Gimeno BS, Ashmore MR, Ineson P (2006) Atmospheric nitrogen deposition in world biodiversity hotspots: the need for a greater global perspective in assessing N deposition impacts. Glob Change Biol 12:470–476. https://doi.org/10.1111/j.1365-2486.2006.01104.x
Qian Z, Kong L-j, Y-z S, X-d Y, H-j Z, Xie F-t, Xue A (2019) Effect of biochar on grain yield and leaf photosynthetic physiology of soybean cultivars with different phosphorus efficiencies. J Integr Agric 18:2242–2254
Qiao-Hong Z, Xin-Hua P, Huang T-Q, Zu-Bin X, Holden N (2014) Effect of biochar addition on maize growth and nitrogen use efficiency in acidic red soils. Pedosphere 24:699–708
Raddad EAY, Luukkanen O (2006) Adaptive genetic variation in water-use efficiency and gum yield in Acacia senegal provenances grown on clay soil in the Blue Nile region, Sudan. For Ecol Manag 226:219–229. https://doi.org/10.1016/j.foreco.2006.01.036
Rady MM, Semida WM, Hemida KA, Abdelhamid MT (2016) The effect of compost on growth and yield of Phaseolus vulgaris plants grown under saline soil. Int J Recycl Org Waste Agric 5:311–321
Rahim H, Mian IA, Arif M, Ahmad S, Khan Z (2020) Soil fertility status as influenced by the carryover effect of biochar and summer legumes. Asian J Agric Biol 8:11–16. https://doi.org/10.35495/ajab.2019.05.198
Rasul M, Cho J, Shin H-S, Hur J (2022) Biochar-induced priming effects in soil via modifying the status of soil organic matter and microflora: a review. Sci Total Environ 805:150304. https://doi.org/10.1016/j.scitotenv.2021.150304
Reis CRG, Nardoto GB, Rochelle ALC, Vieira SA, Oliveira RS (2017) Nitrogen dynamics in subtropical fringe and basin mangrove forests inferred from stable isotopes. Oecologia 183:841–848
Reverchon F, Xu Z, Blumfield T, Chen C, Abdullah K (2011) Impact of global climate change and prescribed burning on understorey legumes and associated belowground communities: implications for biogeochemical cycles in forest ecosystems. J Soils Sediments 12:150–160
Reverchon F, Abdullah KM, Bai SH, Villafán E, Blumfield TJ, Patel B, Xu Z (2020) Biological nitrogen fixation by two Acacia species and associated root-nodule bacteria in a suburban Australian forest subjected to prescribed burning. J Soils Sediments 20:122–132. https://doi.org/10.1007/s11368-019-02446-9
Rivero-Villar A, Ruiz-Suárez G, Templer PH, Souza V, Campo J (2021) Nitrogen cycling in tropical dry forests is sensitive to changes in rainfall regime and nitrogen deposition. Biogeochemistry 153:283–302. https://doi.org/10.1007/s10533-021-00788-6
Robertson GP, Vitousek PM (2009) Nitrogen in agriculture: balancing the cost of an essential resource. Annu Rev Environ Resour 34:97–125
Robertson SJ, Rutherford PM, Lopez-Gutierrez JC, Massicotte HB (2012) Biochar enhances seedling growth and alters root symbioses and properties of sub-boreal forest soils. Can J Soil Sci 92:329–340
Robinson D (2001) δ15N as an integrator of the nitrogen cycle. Trends Ecol Evol 16:153–162. https://doi.org/10.1016/S0169-5347(00)02098-X
Rondon MA, Lehmann J, Ramírez J, Hurtado M (2007) Biological nitrogen fixation by common beans (Phaseolus vulgaris L.) increases with bio-char additions. Biol Fertil Soils 43:699–708. https://doi.org/10.1007/s00374-006-0152-z
Saiz E, Sgouridis F, Drijfhout FP, Peichl M, Nilsson MB, Ullah S (2021) Chronic atmospheric reactive nitrogen deposition suppresses biological nitrogen fixation in peatlands. Environ Sci Technol 55:1310–1318. https://doi.org/10.1021/acs.est.0c04882
Sala OE, Chapin FS 3rd, Armesto JJ, Berlow E, Bloomfield J, Dirzo R, Huber-Sanwald E, Huenneke LF, Jackson RB, Kinzig A, Leemans R, Lodge DM, Mooney HA, Oesterheld M, Poff NL, Sykes MT, Walker BH, Walker M, Wall DH (2000) Global biodiversity scenarios for the year 2100. Science 287:1770–1774. https://doi.org/10.1126/science.287.5459.1770
Savard MM, Bégin C, Smirnoff A, Marion J, Rioux-Paquette E (2009) Tree-ring nitrogen isotopes reflect anthropogenic NOx emissions and climatic effects. Environ Sci Technol 43:604–609
Schlesinger WH (2009) On the fate of anthropogenic nitrogen. Proc Natl Acad Sci 106:203–208
Schneider F, Amelung W, Don A (2021) Origin of carbon in agricultural soil profiles deduced from depth gradients of C:N ratios, carbon fractions, δ13C and δ15N values. Plant Soil 460:123–148. https://doi.org/10.1007/s11104-020-04769-w
Schulze E, Williams R, Farquhar G, Schulze W, Langridge J, Miller J, Walker BH (1998) Carbon and nitrogen isotope discrimination and nitrogen nutrition of trees along a rainfall gradient in northern Australia. Funct Plant Biol 25:413–425
Shearer G, Kohl DH (1986) N2-fixation in field settings: estimations based on natural 15N abundance. Funct Plant Biol 13:699–756
Shen Q, Hedley M, Camps Arbestain M, Kirschbaum MUF (2016) Can biochar increase the bioavailability of phosphorus? J Soil Sci Plant Nutr 16:268–286
Song Y, Zhang X, Ma B, Chang SX, Gong J (2014) Biochar addition affected the dynamics of ammonia oxidizers and nitrification in microcosms of a coastal alkaline soil. Biol Fertil Soils 50:321–332
Song L, Drewer J, Zhu B, Zhou M, Cowan N, Levy P, Skiba U (2020) The impact of atmospheric N deposition and N fertilizer type on soil nitric oxide and nitrous oxide fluxes from agricultural and forest Eutric Regosols. Biol Fertil Soils 56:1077–1090. https://doi.org/10.1007/s00374-020-01485-6
Sponseller RA, Gundale MJ, Futter M, Ring E, Nordin A, Näsholm T, Laudon H (2016) Nitrogen dynamics in managed boreal forests: recent advances and future research directions. Ambio 45:175–187
Streubel J, Collins H, Garcia-Perez M, Tarara J, Granatstein D, Kruger C (2011) Influence of contrasting biochar types on five soils at increasing rates of application. Soil Sci Soc Am J 75:1402–1413
Su C, Kang R, Zhu W, Huang W, Song L, Wang A, Liu D, Quan Z, Zhu F, Fu P, Fang Y (2020) δ15N of nitric oxide produced under aerobic or anaerobic conditions from seven soils and their associated N isotope fractionations. J Geophys Res Biogeosci 125:e2020JG005705. https://doi.org/10.1029/2020JG005705
Succarie A, Xu Z, Wang W (2022) The variation and trends of nitrogen cycling and nitrogen isotope composition in tree rings: the potential for fingerprinting climate extremes and bushfires. J Soils Sediments 22:2343–2353
Sun H, Lu H, Chu L, Shao H, Shi W (2017) Biochar applied with appropriate rates can reduce N leaching, keep N retention and not increase NH(3) volatilization in a coastal saline soil. Sci Total Environ 575:820–825. https://doi.org/10.1016/j.scitotenv.2016.09.137
Sun M, Huo Z, Zheng Y, Dai X, Feng S, Mao X (2018) Quantifying long-term responses of crop yield and nitrate leaching in an intensive farmland using agro-eco-environmental model. Sci Total Environ 613:1003–1012
Taresh S, Bai SH, Abdullah KM, Zalucki J, Nessa A, Omidvar N, Wang D, Zhan J, Wang F, Yang J, Kichamu-Wachira E, Xu Z (2021) Long-term impact of prescribed burning on water use efficiency, biological nitrogen fixation, and tree growth of understory acacia species in a suburban forest ecosystem of subtropical Australia. J Soils Sediments 21:3620–3631. https://doi.org/10.1007/s11368-021-03030-w
Templer PH, Mack MC, Iii FSC, Christenson LM, Compton JE, Crook HD, Currie WS, Curtis CJ, Dail DB, D'antonio CM, Emmett BA, Epstein HE, Goodale CL, Gundersen P, Hobbie SE, Holland K, Hooper DU, Hungate BA, Lamontagne S et al (2012) Sinks for nitrogen inputs in terrestrial ecosystems: a meta-analysis of 15N tracer field studies. Ecology 93:1816–1829. https://doi.org/10.1890/11-1146.1
Thomas SC, Gale N (2015) Biochar and forest restoration: a review and meta-analysis of tree growth responses. New For 46:931–946
Thonicke K, Spessa A, Prentice I, Harrison SP, Dong L, Carmona-Moreno C (2010) The influence of vegetation, fire spread and fire behaviour on biomass burning and trace gas emissions: results from a process-based model. Biogeosci 7:1991–2011
Tieszen LL (1991) Natural variations in the carbon isotope values of plants: implications for archaeology, ecology, and paleoecology. J Archaeol Sci 18:227–248. https://doi.org/10.1016/0305-4403(91)90063-U
Tomczyk A, Sokołowska Z, Boguta P (2020) Biochar physicochemical properties: pyrolysis temperature and feedstock kind effects. Rev Environ Sci Biotechnol 19:191–215. https://doi.org/10.1007/s11157-020-09523-3
Uzoma KC, Inoue M, Andry H, Fujimaki H, Zahoor A, Nishihara E (2011) Effect of cow manure biochar on maize productivity under sandy soil condition. Soil Use Manag 27:205–212. https://doi.org/10.1111/j.1475-2743.2011.00340.x
Vitousek PM, Aber JD, Howarth RW, Likens GE, Matson PA, Schindler DW, Schlesinger WH, Tilman DG (1997) Human alteration of the global nitrogen cycle: sources and consequences. Ecol Appl 7:737–750
Voroney P (2019) Chapter 4 - soils for horse pasture management. In: Sharpe, ed. Horse Pasture Management. Academic Press, Cambridge 65–79
Wang Y, Xu Z, Zhou Q (2014) Impact of fire on soil gross nitrogen transformations in forest ecosystems. J Soils Sediments 14:1030–1040. https://doi.org/10.1007/s11368-014-0879-3
Wang J, Xiong Z, Kuzyakov Y (2016) Biochar stability in soil: meta-analysis of decomposition and priming effects. GCB Bioenergy 8:512–523. https://doi.org/10.1111/gcbb.12266
Wang C, Houlton BZ, Liu D, Hou J, Cheng W, Bai E (2018) Stable isotopic constraints on global soil organic carbon turnover. Biogeosci 15:987–995
Wang D, Abdullah KM, Xu Z, Wang W (2020a) Water extractable organic C and total N: the most sensitive indicator of soil labile C and N pools in response to the prescribed burning in a suburban natural forest of subtropical Australia. Geoderma 377:114586. https://doi.org/10.1016/j.geoderma.2020.114586
Wang Z, Wang Z, Luo Y, Zhan Y-N, Meng Y-L, Zhou Z-G (2020b) Biochar increases 15N fertilizer retention and indigenous soil N uptake in a cotton-barley rotation system. Geoderma 357:113944
Wang Y, Fu X, Wang T, Ma J, Gao H, Wang X, Pu W (2023) Large contribution of nitrous acid to soil-emitted reactive oxidized nitrogen and its effect on air quality. Environ Sci Technol 57:3516–3526. https://doi.org/10.1021/acs.est.2c07793
Werth M, Kuzyakov Y (2010) 13C fractionation at the root–microorganisms–soil interface: a review and outlook for partitioning studies. Soil Biol Biochem 42:1372–1384
Williams MM, Arnott JC (2010) A comparison of variable economic costs associated with two proposed biochar application methods. Ann Environ Sci 4:23–30 http://www.aes.neu.edu/table_contents/abstract21/
Witt GB, English NB, Balanzategui D, Hua Q, Gadd P, Heijnis H, Bird MI (2017) The climate reconstruction potential of Acacia cambagei (gidgee) for semi-arid regions of Australia using stable isotopes and elemental abundances. J Arid Environ 136:19–27
Wolfe AH, Patz JA (2002) Reactive nitrogen and human health: acute and long-term implications. Ambio 31:120–125. https://doi.org/10.1579/0044-7447-31.2.120
Woolf D, Amonette JE, Street-Perrott FA, Lehmann J, Joseph S (2010) Sustainable biochar to mitigate global climate change. Nat Commun 1:56. https://doi.org/10.1038/ncomms1053
Wu H, Zeng G, Liang J, Chen J, Xu J, Dai J, Li X, Chen M, Xu P, Zhou Y (2016) Responses of bacterial community and functional marker genes of nitrogen cycling to biochar, compost and combined amendments in soil. Appl Microbiol Biotechnol 100:8583–8591
Wu H, Lai C, Zeng G, Liang J, Chen J, Xu J, Dai J, Li X, Liu J, Chen M (2017) The interactions of composting and biochar and their implications for soil amendment and pollution remediation: a review. Crit Rev Biotechnol 37:754–764
Xie D, Duan L, Si G, Liu W, Zhang T, Mulder J (2021) Long-term 15N balance after single-dose input of 15N-labeled NH4+ and NO3− in a subtropical forest under reducing N deposition. Glob Biogeochem Cycles 35:e2021GB006959
Xu Z, Saffigna P, Farquhar G, Simpson J, Haines R, Walker S, Osborne D, Guinto D (2000) Carbon isotope discrimination and oxygen isotope composition in clones of the F1 hybrid between slash pine and Caribbean pine in relation to tree growth, water-use efficiency and foliar nutrient concentration. Tree Physiol 20:1209–1217
Xu Z, Chen C, He J, Liu J (2009) Trends and challenges in soil research 2009: linking global climate change to local long-term forest productivity. J Soils Sediments 9:83–88
Xu N, Tan G, Wang H, Gai X (2016) Effect of biochar additions to soil on nitrogen leaching, microbial biomass and bacterial community structure. Eur J Soil Biol 74:1–8
Xue L, He S, Duan J, Zhang Z, Yang L (2017) Agricultural non-point source pollution nitrogen load reduction strategy and technology of nutrient reusing in agricultural fields to replace fertilizer. J Agro-Environ Sci 36:1226–1231
Yang X, Lu Y, Yin X (2015) A 5-year lysimeter monitoring of nitrate leaching from wheat–maize rotation system: comparison between optimum N fertilization and conventional farmer N fertilization. Agric Ecosyst Environ 199:34–42
Yu X, Tian X, Lu Y, Liu Z, Guo Y, Chen J, Li C, Zhang M, Wan Y (2018) Combined effects of straw-derived biochar and bio-based polymer-coated urea on nitrogen use efficiency and cotton yield. Chem Speciat Bioavailab 30:112–122. https://doi.org/10.1080/09542299.2018.1518730
Zhang X, Chen S, Sun H, Pei D, Wang Y (2008) Dry matter, harvest index, grain yield and water use efficiency as affected by water supply in winter wheat. Irrig Sci 27:1–10
Zhang H, Voroney R, Price G (2015) Effects of temperature and processing conditions on biochar chemical properties and their influence on soil C and N transformations. Soil Biol Biochem 83:19–28
Zhang M, Wang W, Bai SH, Zhou X, Teng Y, Xu Z (2018a) Antagonistic effects of nitrification inhibitor 3, 4-dimethylpyrazole phosphate and fungicide iprodione on net nitrification in an agricultural soil. Soil Biol Biochem 116:167–170
Zhang M, Wang W, Tang L, Heenan M, Xu Z (2018b) Effects of nitrification inhibitor and herbicides on nitrification, nitrite and nitrate consumptions and nitrous oxide emission in an Australian sugarcane soil. Biol Fertil Soils 54:697–706
Zhang M, Wang W, Wang D, Heenan M, Xu Z (2018c) Short-term responses of soil nitrogen mineralization, nitrification and denitrification to prescribed burning in a suburban forest ecosystem of subtropical Australia. Sci Total Environ 642:879–886
Zhang S, Du Q, Sun Y, Song J, Yang F, Tsang DC (2020) Fabrication of L-cysteine stabilized α-FeOOH nanocomposite on porous hydrophilic biochar as an effective adsorbent for Pb2+ removal. Sci Total Environ 720:137415
Zhang M, Hou R, Li T, Fu Q, Zhang S, Su A, Xue P, Yang X (2022) Study of soil nitrogen cycling processes based on the 15N isotope tracking technique in the black soil areas. J Clean Prod 375:134173. https://doi.org/10.1016/j.jclepro.2022.134173
Zheng H, Wang Z, Deng X, Herbert S, Xing B (2013) Impacts of adding biochar on nitrogen retention and bioavailability in agricultural soil. Geoderma 206:32–39
Zhou J, Zheng Y, Hou L, An Z, Chen F, Liu B, Wu L, Qi L, Dong H, Han P, Yin G, Liang X, Yang Y, Li X, Gao D, Li Y, Liu Z, Bellerby R, Liu M (2023) Effects of acidification on nitrification and associated nitrous oxide emission in estuarine and coastal waters. Nat Commun 14:1380. https://doi.org/10.1038/s41467-023-37104-9
Zhu Q, Schmidt JP, Buda AR, Bryant RB, Folmar GJ (2011) Nitrogen loss from a mixed land use watershed as influenced by hydrology and seasons. J Hydrol 405:307–315
Zhu X, Chen B, Zhu L, Xing B (2017) Effects and mechanisms of biochar-microbe interactions in soil improvement and pollution remediation: a review. Environ Pollut 227:98–115
Zimmerman AR, Gao B, Ahn M-Y (2011) Positive and negative carbon mineralization priming effects among a variety of biochar-amended soils. Soil Biol Biochem 43:1169–1179
Zong Z, Sun Z, Xiao L, Tian C, Liu J, Sha Q, Li J, Fang Y, Zheng J, Zhang G (2020) Insight into the variability of the nitrogen isotope composition of vehicular NOx in China. Environ Sci Technol 54:14246–14253. https://doi.org/10.1021/acs.est.0c04749
Zornoza R, Moreno-Barriga F, Acosta J, Muñoz M, Faz A (2016) Stability, nutrient availability and hydrophobicity of biochars derived from manure, crop residues, and municipal solid waste for their use as soil amendments. Chemosphere 144:122–130
Funding
Open Access funding enabled and organized by CAUL and its Member Institutions. This work was financially supported by the Griffith University.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
All authors certify that they have no relevant financial or non-financial interests to disclose.
Additional information
Responsible Editor: Xinhua He.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
ESM 1
(DOCX 30 kb)
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Sun, W., Li, Y., Xu, Z. et al. Biochar application for enhancing water and nitrogen use efficiency of understory acacia species in a suburban native forest subjected to nitrogen deposition in Southeast Queensland. Plant Soil (2024). https://doi.org/10.1007/s11104-024-06647-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11104-024-06647-1