Abstract
Key message
Interactions among phytohormones are essential for providing tolerance of sorghum plants to aphids.
Abstract
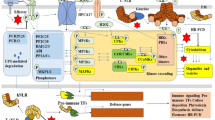
Plant’s encounter with insect herbivores trigger defense signaling networks that fine-tune plant resistance to insect pests. Although it is well established that phytohormones contribute to antixenotic- and antibiotic-mediated resistance to insect pests, their role in conditioning plant tolerance, the most durable and promising category of host plant resistance, is largely unknown. Here, we screened a panel of sorghum (Sorghum bicolor) inbred lines to identify and characterize sorghum tolerance to sugarcane aphids (SCA; Melanaphis sacchari Zehntner), a relatively new and devastating pest of sorghum in the United States. Our results suggest that the sorghum genotype SC35, the aphid-tolerant line identified among the sorghum genotypes, displayed minimal plant biomass loss and a robust photosynthetic machinery, despite supporting higher aphid population. Phytohormone analysis revealed significantly higher basal levels of 12-oxo-phytodienoic acid, a precursor in the jasmonic acid biosynthesis pathway, in the sorghum SCA-tolerant SC35 plants. Salicylic acid accumulation appeared as a generalized plant response to aphids in sorghum plants, however, SCA feeding-induced salicylic acid levels were unaltered in the sorghum tolerant genotype. Conversely, basal levels of abscisic acid and aphid feeding-induced cytokinins were accumulated in the SCA-tolerant sorghum genotype. Our findings imply that the aphid-tolerant sorghum genotype tightly controls the relationship among phytohormones, as well as provide significant insights into the underlying mechanisms that contribute to plant tolerance to sap-sucking aphids.





Similar content being viewed by others
References
Andreas P, Kisiala A, Emery RJN, De Clerck-Floate R, Tooker JF, Price PW, Miller DG, Chen M-S, Connor EF (2020) Cytokinins are abundant and widespread among insect species. Plants 9:208. https://doi.org/10.3390/plants9020208
Armstrong JS, Rooney WL, Peterson GC, Villenueva RT, Brewer MJ, Sekula-Ortiz D (2015) Sugarcane aphid (Hemiptera: Aphididae): host range and sorghum resistance including cross-resistance from greenbug sources. J Econ Entomol 108:576–582. https://doi.org/10.1093/jee/tou065
Armstrong JS, Paudyal S, Limaje A, Elliott N, Hoback W (2018) Plant resistance in sorghums to the sugarcane aphid (Hemiptera: Aphididae). J Entomol Sci 53:478–485. https://doi.org/10.18474/JES17-106.1
Barbosa dos Santos I, Park S-W (2019) Versatility of cyclophilins in plant growth and survival: a case study in Arabidopsis. Biomolecules. https://doi.org/10.3390/biom9010020
Beck SD (1965) Resistance of plants to insects. Annu Rev Entomol 10:207–232. https://doi.org/10.1146/annurev.en.10.010165.001231
Bouchet S, Olatoye MO, Marla SR, Perumal R, Tesso T, Yu J, Tuinstra M, Morris GP (2017) Increased power to dissect adaptive traits in global sorghum diversity using a nested association mapping population. Genetics 206:573–585. https://doi.org/10.1534/genetics.116.198499
Bowling RD, Brewer MJ, Kerns DL, Gordy J, Seiter N, Elliott NE, Buntin GD, Way MO, Royer TA, Biles S, Maxson E (2016) Sugarcane aphid (Hemiptera: Aphididae): a new pest on sorghum in North America. J Integr Pest Manag. https://doi.org/10.1093/jipm/pmw011
Chapman KM, Marchi-Werle L, Hunt TE, Heng-Moss TM, Louis J (2018) Abscisic and jasmonic acids contribute to soybean tolerance to the soybean aphid (Aphis glycines Matsumura). Sci Rep 8:1–12. https://doi.org/10.1038/s41598-018-33477-w
de Morais CL, Pinheiro SS, Martino HSD, Pinheiro-Sant’Ana HM (2017) Sorghum (Sorghum bicolor L.): nutrients, bioactive compounds, and potential impact on human health. Crit Rev Food Sci Nutr 57:372–390. https://doi.org/10.1080/10408398.2014.887057
Erb M, Meldau S, Howe GA (2012) Role of phytohormones in insect-specific plant reactions. Trends Plant Sci 17:250–259. https://doi.org/10.1016/j.tplants.2012.01.003
Foyer CH, Noctor G (2005) Redox homeostasis and antioxidant signaling: a metabolic interface between stress perception and physiological responses. Plant Cell 17:1866–1875. https://doi.org/10.1105/tpc.105.033589
Gallei M, Luschnig C, Friml J (2020) Auxin signalling in growth: Schrödinger’s cat out of the bag. Curr Opin Plant Biol 53:43–49. https://doi.org/10.1016/j.pbi.2019.10.003
González-Lamothe R, El Oirdi M, Brisson N, Bouarab K (2012) The conjugated auxin indole-3-acetic acid–aspartic acid promotes plant disease development. Plant Cell 24:762–777. https://doi.org/10.1105/tpc.111.095190
Großkinsky DK, Edelsbrunner K, Pfeifhofer H, van der Graaff E, Roitsch T (2013) Cis- and trans-zeatin differentially modulate plant immunity. Plant Signal Behav 8:e24798. https://doi.org/10.4161/psb.24798
Grover S, Varsani S, Kolomiets MV, Louis J (2020) Maize defense elicitor, 12-oxo-phytodienoic acid, prolongs aphid salivation. Commun Integr Biol 13:63–66. https://doi.org/10.1080/19420889.2020.1763562
Gururani MA, Mohanta TK, Bae H (2015) Current understanding of the interplay between phytohormones and photosynthesis under environmental stress. Int J Mol Sci 16:19055–19085. https://doi.org/10.3390/ijms160819055
Hillwig MS, Chiozza M, Casteel CL, Lau ST, Hohenstein J, Hernández E, Jander G, MacIntosh GC (2016) Abscisic acid deficiency increases defence responses against Myzus persicae in Arabidopsis. Mol Plant Pathol 17:225–235. https://doi.org/10.1111/mpp.12274
Howe GA, Jander G (2008) Plant immunity to insect herbivores. Annu Rev Plant Biol 59:41–66. https://doi.org/10.1146/annurev.arplant.59.032607.092825
Hui D, Iqbal J, Lehmann K, Gase K, Saluz HP, Baldwin IT (2003) Molecular interactions between the specialist herbivore Manduca sexta (Lepidoptera, Sphingidae) and its natural host Nicotiana attenuata: microarray analysis and further characterization of large-scale changes in herbivore-induced mRNAs. Plant Physiol 131:1877–1893. https://doi.org/10.1104/pp.102.018176
Kerchev PI, Fenton B, Foyer CH, Hancock RD (2012) Plant responses to insect herbivory: interactions between photosynthesis, reactive oxygen species and hormonal signalling pathways. Plant Cell Environ 35:441–453. https://doi.org/10.1111/j.1365-3040.2011.02399.x
Kiani M, Szczepaniec A (2018) Effects of sugarcane aphid herbivory on transcriptional responses of resistant and susceptible sorghum. BMC Genomics 19:774. https://doi.org/10.1186/s12864-018-5095-x
Koch KG, Chapman K, Louis J, Heng-Moss T, Sarath G (2016) Plant tolerance: a unique approach to control hemipteran pests. Front Plant Sci. https://doi.org/10.3389/fpls.2016.01363
Koo YM, Heo AY, Choi HW (2020) Salicylic acid as a safe plant protector and growth regulator. Plant Pathol J 36:1–10. https://doi.org/10.5423/ppj.rw.12.2019.0295
Kopriva S (2013) 12-Oxo-phytodienoic acid interaction with cyclophilin CYP20-3 is a benchmark for understanding retrograde signaling in plants. Proc Natl Acad Sci USA 110:9197–9198. https://doi.org/10.1073/pnas.1307482110
Kotchoni SO, Gachomo EW (2006) The reactive oxygen species network pathways: an essential prerequisite for perception of pathogen attack and the acquired disease resistance in plants. J Biosci 31:389–404. https://doi.org/10.1007/BF02704112
Li S, Zhao J, Zhai Y, Yuan Q, Zhang H, Wu X, Lu Y, Peng J, Sun Z, Lin L, Zheng H, Chen J, Yan F (2019) The hypersensitive induced reaction 3 (HIR3) gene contributes to plant basal resistance via an EDS1 and salicylic acid-dependent pathway. Plant J 98:783–797. https://doi.org/10.1111/tpj.14271
Liu X, Williams CE, Nemacheck JA, Wang H, Subramanyam S, Zheng C, Chen M-S (2010) Reactive oxygen species are involved in plant defense against a gall midge. Plant Physiol 152:985–999. https://doi.org/10.1104/pp.109.150656
Louis J, Shah J (2013) Arabidopsis thaliana—Myzus persicae interaction: shaping the understanding of plant defense against phloem-feeding aphids. Front Plant Sci. https://doi.org/10.3389/fpls.2013.00213
Louis J, Basu S, Varsani S, Castano-Duque L, Jiang V, Williams WP, Felton GW, Luthe DS (2015) Ethylene contributes to maize insect resistance 1-mediated maize defense against the phloem sap-sucking corn leaf aphid. Plant Physiol 169:313–324. https://doi.org/10.1104/pp.15.00958
Machado RAR, Arce CCM, Ferrieri AP, Baldwin IT, Erb M (2015) Jasmonate-dependent depletion of soluble sugars compromises plant resistance to Manduca sexta. New Phytol 207:91–105. https://doi.org/10.1111/nph.13337
Mapes CC, Davies PJ (2001) Cytokinins in the ball gall of Solidago altissima and in the gall forming larvae of Eurosta solidaginis. New Phytol 151:203–212. https://doi.org/10.1046/j.1469-8137.2001.00158.x
Moran PJ, Cheng Y, Cassell JL, Thompson GA (2002) Gene expression profiling of Arabidopsis thaliana in compatible plant-aphid interactions. Arch Insect Biochem Physiol 51:182–203. https://doi.org/10.1002/arch.10064
Nalam V, Louis J, Shah J (2019) Plant defense against aphids, the pest extraordinaire. Plant Sci 279:96–107. https://doi.org/10.1016/j.plantsci.2018.04.027
Negin B, Yaaran A, Kelly G, Zait Y, Moshelion M (2019) Mesophyll abscisic acid restrains early growth and flowering but does not directly suppress photosynthesis. Plant Physiol 180:910–925. https://doi.org/10.1104/pp.18.01334
Nibouche S, Costet L, Holt JR, Jacobson A, Pekarcik A, Sadeyen J, Armstrong JS, Peterson GC, McLaren N, Medina RF (2018) Invasion of sorghum in the Americas by a new sugarcane aphid (Melanaphis sacchari) superclone. PLoS ONE. https://doi.org/10.1371/journal.pone.0196124
Osugi A, Kojima M, Takebayashi Y, Ueda N, Kiba T, Sakakibara H (2017) Systemic transport of trans-zeatin and its precursor have differing roles in Arabidopsis shoots. Nat Plants 3:1–6. https://doi.org/10.1038/nplants.2017.112
Painter RH (1951) Insect resistance in crop plants. Macmillan, New York
Panda N, Khush GA (1995) Host plant resistance to insects. CAB International, Wallingford
Peterson RKD, Varella AC, Higley LG (2017) Tolerance: the forgotten child of plant resistance. PeerJ 5:e3934. https://doi.org/10.7717/peerj.3934
Prerostova S, Dobrev PI, Gaudinova A, Knirsch V, Körber N, Pieruschka R, Fiorani F, Brzobohatý B, Černý M, Spichal L, Humplik J, Vanek T, Schurr U, Vankova R (2018) Cytokinins: their impact on molecular and growth responses to drought stress and recovery in Arabidopsis. Front Plant Sci. https://doi.org/10.3389/fpls.2018.00655
Singh V, Louis J, Ayre BG, Reese JC, Shah J (2011) TREHALOSE PHOSPHATE SYNTHASE11-dependent trehalose metabolism promotes Arabidopsis thaliana defense against the phloem-feeding insect Myzus persicae. Plant J 67:94–104. https://doi.org/10.1111/j.1365-313X.2011.04583.x
Smith CM (2005) Plant resistance to arthropods: molecular and conventional approaches. Springer, Dordrecht
Studham ME, MacIntosh GC (2012) Multiple phytohormone signals control the transcriptional response to soybean aphid infestation in susceptible and resistant soybean plants. Mol Plant Microbe Interact 26:116–129. https://doi.org/10.1094/MPMI-05-12-0124-FI
Tan S, Abas M, Verstraeten I, Glanc M, Molnár G, Hajný J, Lasák P, Petřík I, Russinova E, Petrášek J, Novák O, Pospíšil J, Friml J (2020) Salicylic acid targets protein phosphatase 2A to attenuate growth in plants. Curr Biol 30:381-395.e8. https://doi.org/10.1016/j.cub.2019.11.058
Tetreault HM, Grover S, Scully ED, Gries T, Palmer NA, Sarath G, Louis J, Sattler SE (2019) Global responses of resistant and susceptible sorghum (Sorghum bicolor) to sugarcane aphid (Melanaphis sacchari). Front Plant Sci 10:145. https://doi.org/10.3389/fpls.2019.00145
Tooker JF, De Moraes CM (2011) Feeding by a gall-inducing caterpillar species alters levels of indole-3-acetic and abscisic acid in Solidago altissima (Asteraceae) stems. Arthropod-Plant Interact 5:115–124. https://doi.org/10.1007/s11829-010-9120-5
van Butselaar T, Van den Ackerveken G (2020) Salicylic acid steers the growth–immunity tradeoff. Trends Plant Sci. https://doi.org/10.1016/j.tplants.2020.02.002
Vanderlip RL, Reeves HE (1972) Growth stages of sorghum [Sorghum bicolor, (L.) Moench.]. Agron J 64:13–16. https://doi.org/10.2134/agronj1972.00021962006400010005x
Varsani S, Grover S, Zhou S, Koch KG, Huang P-C, Kolomiets MV, Williams WP, Heng-Moss T, Sarath G, Luthe DS, Jander G, Louis J (2019) 12-Oxo-phytodienoic acid acts as a regulator of maize defense against corn leaf aphid. Plant Physiol. https://doi.org/10.1104/pp.18.01472
Verma V, Ravindran P, Kumar PP (2016) Plant hormone-mediated regulation of stress responses. BMC Plant Biol 16:86. https://doi.org/10.1186/s12870-016-0771-y
Voothuluru P, Meng J, Khajuria C, Louis J, Zhu L, Starkey S, Wilde GE, Baker CA, Smith CM (2006) Categories and inheritance of resistance to Russian wheat aphid (Homoptera: Aphididae) Biotype 2 in a selection from wheat cereal introduction 2401. J Econ Entomol 99:1854–1861. https://doi.org/10.1603/0022-0493-99.5.1854
Wang D, Pajerowska-Mukhtar K, Culler AH, Dong X (2007) Salicylic acid inhibits pathogen growth in plants through repression of the auxin signaling pathway. Curr Biol 17:1784–1790. https://doi.org/10.1016/j.cub.2007.09.025
Wasternack C, Strnad M (2018) Jasmonates: news on occurrence, biosynthesis, metabolism and action of an ancient group of signaling compounds. Int J Mol Sci. https://doi.org/10.3390/ijms19092539
Werner T, Motyka V, Strnad M, Schmülling T (2001) Regulation of plant growth by cytokinin. Proc Natl Acad Sci USA 98:10487–10492. https://doi.org/10.1073/pnas.171304098
White WH, Reagan TE, Hall DG (2001) Melanaphis sacchari (Homoptera: Aphididae), A sugarcane pest new to Louisiana. Fla Entomol 435
Xu Y, Huang B (2009) Effects of foliar-applied ethylene inhibitor and synthetic cytokinin on creeping bentgrass to enhance heat tolerance. Crop Sci 49:1876–1884. https://doi.org/10.2135/cropsci2008.07.0441
Ym K, Ay H, Hw C (2020) Salicylic acid as a safe plant protector and growth regulator. Plant Pathol J 36:1–10. https://doi.org/10.5423/ppj.rw.12.2019.0295
Yoshida T, Christmann A, Yamaguchi-Shinozaki K, Grill E, Fernie AR (2019) Revisiting the basal role of ABA—roles outside of stress. Trends Plant Sci 24:625–635. https://doi.org/10.1016/j.tplants.2019.04.008
Zhang N, Zhou S, Yang D, Fan Z (2020) Revealing shared and distinct genes responding to JA and SA signaling in Arabidopsis by meta-analysis. Front Plant Sci. https://doi.org/10.3389/fpls.2020.00908
Zhu-Salzman K, Salzman RA, Ahn J-E, Koiwa H (2004) Transcriptional regulation of sorghum defense determinants against a phloem-feeding aphid. Plant Physiol 134:420–431. https://doi.org/10.1104/pp.103.028324
Zogli P, Pingault L, Grover S, Louis J (2020) Ento(o)mics: the intersection of “omic” approaches to decipher plant defense against sap-sucking insect pests. Curr Opin Plant Biol 56:153–161. https://doi.org/10.1016/j.pbi.2020.06.002
Züst T, Agrawal AA (2016) Mechanisms and evolution of plant resistance to aphids. Nat Plants 2:1–9. https://doi.org/10.1038/nplants.2015.206
Acknowledgements
We would like to acknowledge Manny Saluja and John Toy for help with LI-COR data measurements and seed production, respectively. We also thank Harkamal Walia and Tala Awada for providing access to LI-COR instrument, Emily Robinson with statistical analysis, and the Proteomic and Metabolomics Facility (Center for Biotechnology at the University of Nebraska-Lincoln) for the LC–MS assay and quantification of plant hormones.
Funding
Work in the Louis laboratory was supported by funds from US National Science Foundation CAREER grant IOS-1845588 and USDA-ARS (58-3042-6-070). This work was partially supported by USDA-ARS CRIS projects 3042-21000-034-00-D (GS) and 3042-21220-033-00-D (SES). E.A. was partly supported by Undergraduate Creative Activity and Research (UCARE) and Agricultural Research Division (ARD) Undergraduate Student Research funds from the University of Nebraska-Lincoln.
Author information
Authors and Affiliations
Contributions
SG and JL conceived and designed the research; SG and EA performed the research; GS and SES contributed reagents, methods development and provided guidance on experiments; SG and JL wrote the paper. All authors reviewed and edited the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflicts of interest to declare.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplementary file1 (PDF 398 kb)
Supp. Fig. S1 Total number of sugarcane aphids recovered seven days after aphid infestation of two-week-old sorghum RTx430, SC35, and SC1345 plants that were initially infested with 10 adult apterous aphids per plant.
Supp. Fig. S2 Percent plant biomass loss for each sorghum NAM founder line after 14 days of sugarcane aphid (SCA) infestation of two-week-old sorghum plants that were initially infested with 10 adult apterous aphids per plant.
Supp. Fig. S3 (A) Loading plot of principal components 1 and 2 from the principal component analysis (PCA) of data collected from tolerance experiment on sorghum NAM founder lines after 14 days of SCA infestation. The loading data depicts the unrotated loading matrix between the variables and the components. Uninfested plants of similar age were used to calculate changes in plant growth upon aphid infestation. (B) Loading data of the PCA which indicates the effects of components on different variables.
Table S1 Total number of sugarcane aphids recovered 14 days after aphid infestation of two-week-old sorghum plants that were initially infested with 10 adult apterous aphids per plant.
Table S2 Plant growth parameters of sorghum genotypes with and without sugarcane aphid infestation for 14 days.
Rights and permissions
About this article
Cite this article
Grover, S., Agpawa, E., Sarath, G. et al. Interplay of phytohormones facilitate sorghum tolerance to aphids. Plant Mol Biol 109, 639–650 (2022). https://doi.org/10.1007/s11103-020-01083-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11103-020-01083-y