Abstract
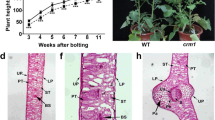
Leaf color mutants are valuable resources for studying regulatory mechanisms of photosynthetic pigment metabolism. In this study, a chlorophyll-deficient golden leaf mutant lcm1 of Chinese cabbage was identified from its wild-type “FT” by ethyl methanesulfonate (EMS) mutagenesis. The phenotype of the golden leaf mutant lcm1 was that the leaves remained golden throughout development. Pigment measurements showed that the chlorophyll content of the mutant lcm1 was less than that of the wild-type “FT”. There were no obvious grana lamellae observed in the chloroplast microstructure of the mutant lcm1. Genetic analysis revealed that the mutant lcm1 phenotype was controlled by a single recessive nuclear gene, lcm1 (BrChlH). The MutMap method and Kompetitive Allele Specific PCR genotyping were used to predict that Bra006208 encoding a Mg-chelatase H subunit, the candidate gene for the mutant lcm1. The 4249-bp lcm1 gene had five exons and a single nucleotide substitution (G to A) in the third exon that resulted in an amino acid substitution (A to V). This SNP2523351 occurred in the highly conserved CobN-Mg_chel domain of the LCM1 (BrCHLH) protein. This study suggests that lcm1 gene plays an important role in the function of CHLH and provides a solid foundation for further studies on the development of plant leaf color.




Similar content being viewed by others
References
Abdi H (2007) Bonferroni and Šidák corrections for multiple comparisons. In: Salkind N (ed) Encyclopedia of Measurement and Statistics. Thousand Oak, pp 103–107
Abe A, Kosugi S, Yoshida K, Natsume S, Takagi H, Kanzaki H, Matsumura H, Yoshida K, Mistsuoka C, Muluneh T, Innan H, Cano L, Kamoun S, Teraushi R (2012) Genome sequencing reveals agronomically important loci in rice using MutMap. Nat Biotechnol 30:174–178
Arnon DI (1949) Copper enzymes in isolated chloroplasts: polyphenoloxidase in Beta vulgaris. Plant Physiol 24:1–15
Ashburner M, Ball CA, Blake JA, Botstein D, Butler H, Cherry JM, Davis AP, Dolinski K, Dwight SS, Eppig JT, Harris MA, Hill DP, Issel-Tarver L, Kasarskis A, Lewis S, Matese JC, Richardson JE, Ringwald M, Rubin GM, Sherlock G (2000) Gene ontology: tool for the unification of biology. Nat Genet 25:25–29
Beale SI (2005) Green genes gleaned. Trends Plant Sci 10:309–312
Benjamini Y, Hochberg Y (1995) Controlling the false discovery rate: a practical and powerful approach to multiple testing. J R Stat Soc Ser B Methodol 57:289–300
Braumann I, Stein N, Hansson M (2014) Reduced chlorophyll biosynthesis in heterozygous barley magnesium chelatase mutants. Plant Physiol Biochem 78:10–14
Chen H, Cheng ZJ, Ma XD, Wu H, Liu YL, Zhou KN, Chen YL, Ma WW, Bi JC, Zhang X, Guo XP, Wang JL, Lei CL, Wu FQ, Lin QB, Liu YQ, Liu LL, Jiang L (2013) A knockdown mutation of YELLOW-GREEN LEAF2 blocks chlorophyll biosynthesis in rice. Plant Cell Rep 32:1855–1867
Cingolani P, Platts A, Wang le L, Coon M, Nguyen T, Wang L, Land SJ, Lu X, Ruden DM (2012) A program for annotating and predicting the effects of single nucleotide polymorphisms, SnpEff: SNPs in the genome of Drosophila melanogaster strain w 1118 ; iso-2; iso-3. Fly (Austin) 6:80–92
Colombo N, Emanuel C, Lainez V, Maldonado S, Prina AR, Börner T (2008) The barley plastome mutant CL2 affects expression of nuclear and chloroplast housekeeping genes in a cell-age dependent manner. Mol Gen Genomics 279:403–414
Dai M, Thompson RC, Maher C, Contreras-Galindo R, Kaplan MH, Markovitz DM, Omenn G, Meng F (2010) NGSQC: cross-platform quality analysis pipeline for deep sequencing data. BMC Genomics 11:S7
Deng LC, Qin P, Liu Z, Wang GL, Chen WL, Tong JH, Xiao LT, Tu B, Sun YT, Yan W, He H, Tan J, Chen XW, Wang YP, Li SG, Ma BT (2017) Characterization and fine-mapping of a novel premature leaf senescence mutant yellow leaf and dwarf 1 in rice. Plant Physiol Biochem 111:50–58
Dong H, Fei GL, Wu CY, Wu FQ, Sun YY, Chen MJ, Ren YL, Zhou KN, Cheng ZJ, Wang JL, Jiang L, Zhang X, Guo XP, Lei CL, Su N, Wang H, Wan JM (2013) A rice virescent-yellow leaf mutant reveals new insights into the role and assembly of plastid caseinolytic protease in higher plants. Plant Physiol 162:1867–1880
Fekih R, Takagi H, Tamiru M, Abe A, Natsume S, Yaegashi H, Sharma S, Sharma S, Kanzaki H, Matsumura H, Saitoh H, Mitsuoka C, Utsushi H, Uemura A, Kanzaki E, Kosugi S, Yoshida K, Cano L, Kamoun S, Terauchi R (2013) MutMap+: genetic mapping and mutant identification without crossing in rice. PLoS One 8:e68529
Fodje MN, Hansson A, Hansson M, Olsen JG, Gough S, Willows RD, Al-Karadaghi S (2001) Interplay between an AAA module and an integrin I domain may regulate the function of magnesium chelatase. J Mol Biol 311:111–122
Gao ML, Hu LL, Li YH, Weng YQ (2016) The chlorophyll-deficient golden leaf mutation in cucumber is due to a single nucleotide substitution in CsChlI for magnesium chelatase I subunit. Theor Appl Genet 129:1961–1973
Hao N, Du Y, Li H, Wang C, Wang C, Gong S, Zhou S, Wu T (2018) CsMYB36 is involved in the formation of yellow green peel in cucumber (Cucumis sativus L.). Theor Appl Genet 131:1659–1669
Ihnatowicz A, Siwinska J, Meharg AA, Carey M, Koornneef M, Reymond M (2014) Conserved histidine of metal transporter AtNRAMP1 is crucial for optimal plant growth under manganese deficiency at chilling temperatures. New Phytol 202:1173–1183
Imamura T, Takagi H, Miyazato A, Ohki S, Mizukoshi H, Mori M (2018) Isolation and characterization of the betalain biosynthesis gene involved in hypocotyl pigmentation of the allotetraploid Chenopodium quinoa. Biochem Biophys Res Commun 496:280–286
Jensen PE, Gibson LC, Hunter CN (1999) ATPase activity associated with the magnesium- protoporphyrin IX chelatase enzyme of Synechocystis PCC6803: evidence for ATP hydrolysis during Mg2+ insertion, and the MgATP-dependent interaction of the ChlI and ChlD subunits. Biochem J 339:127–134
Jiao Y, Burow G, Gladman N, Acosta-Martinez V, Chen J, Burke J, Ware D, Xin Z (2018) Efficient identification of causal mutations through sequencing of bulked F2 from two allelic bloomless mutants of Sorghum bicolor. Front Plant Sci 8:2267
Kanehisa M, Araki M, Goto S, Hattori M, Hirakawa M, Itoh M, Katayama T, Kawashima S, Okuda S, Tokimatsu T, Yamanishi Y (2008) KEGG for linking genomes to life and the environment. Nucleic Acids Res 36:D480–D484
Kusaba M, Ito H, Morita R, Iida S, Sato Y, Fujimoto M, Kawasaki S, Tanaka R, Hirochika H, Nishimura M, Tanaka A (2007) Rice NON-YELLOW COLORING1 is involved in light-harvesting complex II and grana degradation during leaf senescence. Plant Cell 19:1362–1375
Kusumi K, Sakata C, Nakamura T, Kawasaki S, Yoshimura A, Iba K (2011) A plastid protein NUS1 is essential for build-up of the genetic system for early chloroplast development under cold stress conditions. Plant J 68:1039–1050
Lee S, Lee SC, Byun DH, Lee DY, Park JY, Lee JH, Lee HO, Sung SH, Yang TJ (2014) Association of molecular markers derived from the BrCRTISO1 gene with prolycopene-enriched orange-colored leaves in Brassica rapa. Theor Appl Genet 127:179–191
Li H, Durbin R (2009) Fast and accurate short read alignment with burrows-wheeler transform. Bioinformatics 25:1754–1760
Li H, Handsaker B, Wysoker A, Fennell T, Ruan J, Homer N, Marth G, Abecasis G, Durbin R, 1000 Genome Project Data Processing Subgroup (2009a) The sequence alignment/map (SAM) format and SAMtools. Bioinformatics 25:2078–2079
Li RQ, Yu C, Li YR, Lam TW, Yiu SM, Kristiansen K, Wang J (2009b) SOAP2: an improved ultrafast tool for short read alignment. Bioinformatics 25:966–1967
Mamidi S, Lee RK, Goos JR, McClean PE (2014) Genome-wide association studies identifies seven major regions responsible for iron deficiency chlorosis in soybean (Glycine max). PLoS One 9:e107469
Mochizuki N, Tanaka R, GrimmB MT, Moulin M, Smith AG, Tanaka A, Terry MJ (2010) The cell biology of tetrapyrroles: a life and death struggle. Trends Plant Sci 15:488–498
Mortazavi A, Williams BA, McCue K, Schaeffer L, Wold B (2008) Mapping and quantifying mammalian transcriptomes by RNASeq. Nat Methods 5:621–628
Murray MG, Thompson WF (1980) Rapid isolation of high molecular weight plant DNA. Nucleic Acids Res 8:4321–4325
Pogson BJ, Albrecht V (2011) Genetic dissection of chloroplast biogenesis and development: an overview. Plant Physiol 155:1545–1551
Reid JD, Hunter CN (2002) Current understanding of the function of magnesium chelatase. Biochem Soc Trans 30:643–645
Shi DY, Zheng X, Li L, Lin WH, Xie WJ, Yang JP, Chen SJ, Jin WW (2013) Chlorophyll deficiency in the maize elongated mesocotyl2 mutant is caused by a defective heme oxygenase and delaying grana stacking. PLoS One 8:e80107
Sozer O, Komenda J, Ughy B, Domonkos I, Laczkó-Dobos H, Malec P, Gombos Z, Kis M (2010) Involvement of carotenoids in the synthesis and assembly of protein subunits of photosynthetic reaction centers of synechocystis sp. PCC 6803. Plant Cell Physiol 51:823–835
Stenbaek A, Jensen PE (2010) Redox regulation of chlorophyll biosynthesis. Phytochemistry 71(8–9):853–859
Takagi H, Uemura A, Yaegashi H, Tamiru M, Abe A, Mitsuoka C, Utsushi H, Natsume S, Kanzaki H, Matsumura H, Saitoh H, Yoshida K, Cano LM, Kamoun S, Terauchi R (2013) MutMap-Gap: whole-genome resequencing of mutant F2 progeny bulk combined with de novo assembly of gap regions identifies the rice blast resistance gene Pii. New Phytol 200:276–283
Takagi H, Tamiru M, Abe A, Yoshida K, Uemura A, Yaegashi H, Obara T, Oikawa K, Utsushi H, Kanzaki E, Mitsuoka C, Natsume S, Kosugi S, Kanzaki H, Matsumura H, Urasaki N, Kamoun S, Terauchi R (2015) MutMap accelerates breeding of a salt-tolerant rice cultivar. Nat Biotechnol 33:445–449
Tanaka R, Tanaka A (2007) Tetrapyrrole biosynthesis in higher plants. Plant Biol 58:321–346
Tanaka R, Kobayashi K, Masuda T (2011) Tetrapyrrole metabolism in Arabidopsis thaliana. Arabidopsis Book /Am Soc Plant Biol 9:e0145
Terry MJ, Kendrick RE (1999) Feedback inhibition of chlorophyll synthesis in the phytochrome chromophore-deficient aurea and yellow-green-2 mutants of tomato. Plant Physiol 119:143–152
Wachsman G, Modliszewski JL, Valdes M, Benfey PN (2017) A SIMPLE pipeline for mapping point mutations. Plant Physiol 174:1307–1313
Wang X, Wang H, Wang J, Sun R, Wu J, Liu S, Bai Y, Mun JH, Bancroft I, Cheng F (2011) The genome of the mesopolyploid crop species Brassica rapa. Nat Genet 43:1035–1039
Wang H, Zhang Y, Sun L, Xu P, Tu R, Meng S, Wu W, Anis GB, Hussain K, Riaz A, Chen D, Cao L, Cheng S, Shen X (2018) WB1, a regulator of endosperm development in Rice, is identified by a modified MutMap method. Int J Mol Sci 19:2159
Wu ZM, Zhang X, He B, Diao LP, Sheng SL, Wang JL, Guo XP, Su N, Wang LF, Jiang L, Wang CM, Zhai HQ, Wan JM (2007) A chlorophyll-deficient rice mutant with impaired chlorophyllide esterification in chlorophyll biosynthesis. Plant Physiol 145:29–40
Xu J, Yang J, Wu Z, Liu H, Huang F, Wu Y, Carrie C, Narsai R, Murcha M, Whelan J, Wu P (2013) Identification of a dual-targeted protein belonging to the mitochondrial carrier family that is required for early leaf development in rice. Plant Physiol 161:2036–2048
Xu L, Wang C, Cao W, Zhou S, Wu T (2018) CLAVATA1-type receptor-like kinase CsCLAVATA1 is a putative candidate gene for dwarf mutation in cucumber. Mol Gen Genomics 293:1393–1405
Zhang H, Liu LL, Cai MH, Zhu SS, Zhao JY, Zheng TH, Xu XY, Zeng ZQ, Niu J, Jiang L, Chen SH, Wan JM (2015) A point mutation of magnesium chelatase OsCHLI gene dampens the interaction between CHLI and CHLD subunits in rice. Plant Mol Biol Report 33:1975–1987
Zhou KN, Ren YL, Lv J, Wang YH, Liu F, Zhou F, Zhao SL, Chen SH, Peng C, Zhang X, Guo XP, Cheng ZJ, Wang JL, Wu FQ, Jiang L, Wan JM (2012) Young Leaf Chlorosis 1, a chloroplast-localized gene required for chlorophyll and lutein accumulation during early leaf development in rice. Planta 237:279–292
Zhou S, Hu ZL, Zhu MK, Zhang B, Deng L, Pan Y, Chen GP (2013) Biochemical and molecular analysis of a temperature-sensitive albino mutant in kale named “white dove”. Plant Growth Regul 71:281–294
Zou T, Xiao Q, Li WJ, Luo T, Yuan GQ, He ZY, Liu MX, Li Q, Xu PZ, Zhu J, Liang YY, Deng QM, Wang SQ, Zheng AP, Wang LX, Li P, Li SC (2017) OsLAP6/OsPKS1, an orthologue of Arabidopsis PKSA/LAP6, is critical for proper pollen exine formation. Rice 10:53
Acknowledgements
The authors are grateful to the editors and the anonymous reviewers for their valuable comments.
Funding
This work was supported by the National Key Research and Development Program of China (2016YFD0101701) and National Natural Science Foundation of China (31772298).
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Fu, W., Ye, X., Ren, J. et al. Fine mapping of lcm1, a gene conferring chlorophyll-deficient golden leaf in Chinese cabbage (Brassica rapa ssp. pekinensis). Mol Breeding 39, 52 (2019). https://doi.org/10.1007/s11032-019-0945-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11032-019-0945-z