Abstract
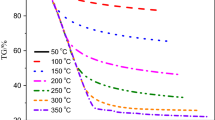
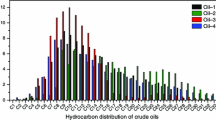
This research focused on the characterization and kinetics of Siberian and Tatarstan crude oils by gas chromatography, combustion calorimetry, and thermogravimetry (TG–DTG) techniques. Calorimetric experiments show that crude oil with higher saturate content and low resin fraction has higher heating value. TG–DTG curves indicates that the crude oils undergoes two major transitions when subjected to an oxidizing and constant rate environment known as low- and high-temperature oxidations at each heating rate studied. Kinetic analysis in the low- and high-temperature oxidation regions was performed using model-free methods knows as Ozawa–Flynn–Wall and Kissinger–Akahira–Sunose. Throughout the study, it was observed that the activation energy values of the crude oil samples are varied between 41–72 and 145–198 kJ mol−1 in low- and high-temperature oxidation regions, respectively.




Similar content being viewed by others
References
Kok MV. Characterization of medium and heavy crude oils using thermal analysis techniques. Fuel Process Technol. 2011;92:1026–2031.
Kok MV. Effect of pressure and particle size on the thermal cracking of light crude oils in sandstone matrix. J Therm Anal Calorim. 2009;97–2:403–7.
Kok MV. Effect of clay on crude oil combustion by thermal analysis techniques. J Therm Anal Calorim. 2006;96:123–7.
Kok MV, Iscan AG. Catalytic affects of metallic additives on the combustion properties of crude oils by thermal analysis techniques. J Therm Anal Calorim. 2011;64:1311–8.
Sonibare OO, Egashira R, Adedosu TA. Thermo-oxidative reactions of Nigerian bitumen. Thermochim Acta. 2003;405–2:195–205.
Castro LV, Vazquez F. Fraction and characterization of Mexican crude oils. Energy Fuel. 2009;23:1603–9.
Goncalves MLA, Teixeira MAG, Pereira RCL, Mercury RLP, Matos JR. Contribution of thermal analysis for characterization of asphaltenes from Brazilian crude oil. J Therm Anal Calorim. 2001;64–2:697–706.
Ni JH, Jia H, Pu WF, Jiang H, Jang JJ, Ren Q. Thermal kinetics study of light oil oxidation using TG/DTG techniques. J Therm Anal Calorim. 2014;17–3:1349–55.
Pereira AN, Trevisan OV. Thermoanalysis and reaction kinetics of heavy oil combustion. J Braz Soc Mech Eng. 2014;36–2:393–401.
Li YB, Zhao JZ, Pu WF, Jia H, Peng H, Zhong D, Wang SK. Catalytic effect analysis of metallic additives on light crude oil by TG and DSC tests. J Therm Anal Calorim. 2013;113–2:579–87.
ASTM D-2007, Standard test method for characteristic groups in rubber extender and processing oils and other petroleum-derived oils by the clay-gel absorption chromatographic method. Annual Book of ASTM Standards. 2002.
Kharrat A, Zacharia J, Cherian V, Anyatonwu A. Energy Fuels. 2007;2:3618–21.
Vyazovkin S. Model-free kinetics. J Therm Anal Calorim. 2006;83:45–51.
Vyazovkin S, Wight CA. Model-free and model-fitting approaches to kinetic analysis of isothermal and non-isothermal data. Thermochim Acta. 1999;53:340–1.
Ozawa T. A new method of analyzing thermogravimetric data. Bull Chem Soc Jpn. 1965;38:1881–6.
Flynn JH, Wall LA. General treatment of the thermogravimetry of polymers. J Res Natl Bur Stan. 1966;70A:487–523.
Flynn JH. The iso-conversional method for determination of energy of activation at constant heating rates: corrections for the Doyle approx. J Therm Anal Calorim. 1983;27:95–102.
Doyle CD. Estimating isothermal life from thermogravimetric data. J Appl Polym Sci. 1962;6:639–42.
Akahira T, Sunose T. Joint convention of four electrical institutes. Res Sci Technol. 1971;16:22–31.
Friedman HL. Kinetics of thermal degradation of char forming plastics from thermogravimetry. J Polym Sci. 1964;6:183–95.
Acknowledgements
The work has been performed according to the Russian Government Program of Competitive Growth of Kazan Federal University.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Varfolomeev, M.A., Nagrimanov, R.N., Galukhin, A.V. et al. Contribution of thermal analysis and kinetics of Siberian and Tatarstan regions crude oils for in situ combustion process. J Therm Anal Calorim 122, 1375–1384 (2015). https://doi.org/10.1007/s10973-015-4892-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10973-015-4892-6