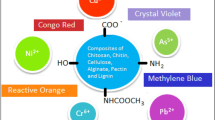
Abstract
Different composites consisting of Zeolite A, ZIF-8, and CuS NPs were prepared by a solvothermal method and their potential was studied for removal of methylene blue (MB) from aqueous solutions. All of the synthesized composites were characterized by Fourier-transform infrared spectroscopy, powder X-Ray diffraction, transmission electron microscopy, field emission scanning electron microscopy, energy dispersive X-Ray analysis, and diffuse reflectance spectroscopy. In order to optimize the experimental conditions, effect of many parameters including contact time, pH of solution, temperature, adsorbent dosage, and initial dye concentration was studied. It was found that CuS NPs/Zeolite A/ZIF-8 composite had a higher MB adsorption capacity than other adsorbents including Zeolite A/ZIF-8 and CuS NPs/ZIF-8. Under the optimized conditions, 75% dye removal efficiency was observed for the CuS NPs/Zeolite A/ZIF-8 composite. Kinetics and thermodynamics of the corresponding adsorption processes was also studied. The Langmuir isotherm model showed a better agreement with the obtained results and the adsorption process of MB fitted the pseudo-second-order kinetic model. Thermodynamic parameters, on the other hand, demonstrated that the adsorption process was exothermic. More interestingly, in presence of an affordable oxidant such as hydrogen peroxide, the MB removal efficiency was raised to 87%.
Graphical Abstract





















Similar content being viewed by others
Notes
Linde type A.
References
A. Chakraborty, D. A. Islam, and H. Acharya (2019). J. Solid State Chem. 269, 566–574. https://doi.org/10.1016/j.jssc.2018.10.036.
J. Fito, S. Abrham, and K. Angassa (2020). Int. J. Environ. Res. Public Health. 14, 501–511. https://doi.org/10.1007/s41742-020-00273-2.
A. Chakraborty and H. Acharya (2018). Colloid Interface Sci. Commun. 24, 35–39. https://doi.org/10.1016/j.colcom.2018.03.005.
S. K. Kansal and A. Kumari (2014). Chem. Rev. 114, 4993–5010. https://doi.org/10.1021/cr400093w.
T. Robinson, B. Chandran, and P. Nigam (2002). Water Res. 36, 2824–2830. https://doi.org/10.1016/S0043-1354(01)00521-8.
J. L. C. Rowsell and O. M. Yaghi (2004). Microporous Mesoporous Mater. 73, 3–14. https://doi.org/10.1016/j.micromeso.2004.03.034.
J. Wang, Y. Wang, Y. Liang, J. Zhou, L. Liu, S. Huang, and J. Cai (2021). Microporous Mesoporous Mater. 310. https://doi.org/10.1016/j.micromeso.2020.110662.
K. S. Park, Z. Ni, A. P. Coˆte´, J. Y. Choi, R. Huang, F. J. Uribe-Romo, H. K. Chae, M. O’Keeffe, and O. M. Yaghi (2006). PNAS Org. Chem. 103, 10186–10191. https://doi.org/10.1073/pnas.0602439103.
H. P. Jing, C. C. Wang, Y. W. Zhang, P. Wang, and R. Li (2014). RSC Adv. 4, 54454–54462. https://doi.org/10.1039/C4RA08820D.
U. P. N. Tran, K. K. A. Le, and N. T. S. Phan (2011). ACS. Catal. 1, 120–127. https://doi.org/10.1021/cs1000625.
V. A. Tran, K. B. Vu, T. T. T. Vo, V. T. Le, H. H. Do, L. G. Bach, and S. W. Lee (2021). Appl. Surf. Sci. 538. https://doi.org/10.1016/j.apsusc.2020.148065.
M. Gougazeh and J.-Ch. Buhl (2014). J. Assoc. Arab Univ. Basic Appl. Sci. 15, 35–42. https://doi.org/10.1016/j.jaubas.2013.03.007.
M. Foroughi, A. Salem, and S. Salem (2021). Mater. Chem. Phys. 258. https://doi.org/10.1016/j.matchemphys.2020.123892.
Y. Wang, F. Jiang, J. Chen, X. Sun, T. Xian, and H. Yang (2020). Nanomater. 10, 178. https://doi.org/10.3390/nano10010178.
L. Chen, W. Yu, and Y. Li (2009). Powder Technol. 191, 52–54. https://doi.org/10.1016/j.powtec.2008.09.007.
Y. Chen, C. Davoisne, J. M. Tarascon, and C. Gue´ry (2012). J. Mater. Chem. 22, 5295–5299. https://doi.org/10.1039/C2JM16692E.
S. Ramadan, L. Guo, Y. Li, B. Yan, and W. Lu (2012). Small. 8, 3143–3150. https://doi.org/10.1002/smll.201200783.
J. Kundu, D. Pradhan, and A. C. S. Appl (2014). Mater. Interfaces. 6, 1823–1834. https://doi.org/10.1021/am404829g.
S. Zhou, L. Gong, X. Zhao, Q. Liang, W. Zhang, L. Wang, K. Yu, and B. Zhou (2020). Chem. Phys. Lett. 759. https://doi.org/10.1016/j.cplett.2020.138034.
S. Iqbal, A. Bahadur, S. Anwer, M. Shoaib, G. Liu, H. Li, M. Raheel, M. Javed, and B. Khalid (2020). CrystEngComm 22, 4162–4173. https://doi.org/10.1039/D0CE00421A.
M. Kamranifar, A. Allahresani, and A. Naghizadeh (2019). J. Hazard. Mater. 366, 545–555. https://doi.org/10.1016/j.jhazmat.2018.12.046.
Y. Liu, M. Li, Q. Zhang, P. Qin, X. Wang, G. He, and L. Li (2020). J. Chem. Technol. Biotechnol. 95, 665–674. https://doi.org/10.1002/jctb.6247.
M. Saranya, C. Santhosh, R. Ramachandran, P. Kollu, P. Saravanan, M. Vinoba, S. K. Jeong, and A. N. Grace (2014). Powder Technol. 252, 25–32. https://doi.org/10.1016/j.powtec.2013.10.031.
X. Liu, J. Luo, Y. Zhu, Y. Yang, and S. Yang (2015). J. Alloy. Compd. 648, 986–993. https://doi.org/10.1016/j.jallcom.2015.07.065.
R. Dod, G. Banerjee, and S. Saini (2012). Biotechnol Bioprocess Eng. 17, 862–874. https://doi.org/10.1007/s12257-011-0614-5.
S. Sohrabnezhad and A. Pourahmad (2010). Desalination 256, 84–89. https://doi.org/10.1016/j.desal.2010.02.009.
L.-Ch. Juang, Ch.-C. Wang, and Ch.-K. Lee (2006). Chemosphere. 64, 1920–1928. https://doi.org/10.1016/j.chemosphere.2006.01.024.
M. H. Moghaddam, R. Nabizadeh, M. H. Dehghani, B. Akbarpour, A. Azari, and M. Yousefi (2019). Microchem. J. 150. https://doi.org/10.1016/j.microc.2019.104185.
I. A. W. Tan, B. H. Hameed, and A. L. Ahmad (2007). Chem. Eng. J. 127, 111–119. https://doi.org/10.1016/j.cej.2006.09.010.
S. Wang, H. Li, and L. Xu (2006). J. Colloid Interface Sci. 29, 71–78. https://doi.org/10.1016/j.jcis.2005.08.006.
Z. Heidarinejad, O. Rahmanian, M. Fazlzadeh, and M. Heidari (2018). J. Mol. Liq. 264, 591–599. https://doi.org/10.1016/j.molliq.2018.05.100.
G. McKay, M. El Guendi, and M. M. Nassar (1987). Wat. Res. 21 (12), 1513–1520. https://doi.org/10.1016/0043-1354(87)90135-7.
Y. Li, K. Zhou, M. He, and J. Yao (2016). Microporous Mesoporous Mater. 234, 287–292. https://doi.org/10.1016/j.micromeso.2016.07.039.
K. Y. Foo and B. H. Hameed (2010). Chem. Eng. J. 156, 2–10. https://doi.org/10.1016/j.cej.2009.09.013.
M. Khajaviana, E. Salehi, and V. Vatanpour (2020). Sep. Purif. Technol. 241. https://doi.org/10.1016/j.seppur.2020.116759.
A. Mittal, R. Ahmad, and I. Hasan (2016). Desalin. Water. Treat. 57, 17790–17807. https://doi.org/10.1080/19443994.2015.1086900.
M. Monier, D. M. Ayad, Y. Wei, and A. A. Sarhan (2010). J. Hazard. Mater. 177, 962–970. https://doi.org/10.1016/j.jhazmat.2010.01.012.
S. Mazloomi, M. Yousefi, H. Nourmoradi, and M. Shams (2019). Health Sci. Eng. 17, 209–218. https://doi.org/10.1007/s40201-019-00341-6.
I. Mantasha, H. A. M. Saleh, K. M. A. Qasem, M. Shahid, M. Mehtab, and M. Ahmad (2020). Inorg. Chim. Acta. 511. https://doi.org/10.1016/j.ica.2020.119787.
Y. Chang, J. Y. Lai, and D. J. Lee (2016). Bioresour. Technol. 222, 513–516. https://doi.org/10.1016/j.biortech.2016.09.125.
D. A. Fungaro, L. C. Grosche, A. S. Pinheiro, J. C. Izidoro, and S. I. Borrely (2010). Electron. J. Chem. 2 (3), 235–247.
K. Y. Hor, J. MCh. Chee, M. N. Chong, B. Jin, C. Saint, P. E. Poh, and R. Aryal (2016). J. Clean. Product. 118, 197–209. https://doi.org/10.1016/j.jclepro.2016.01.056.
R. Chandra and M. Nath (2017). Chem. Select 2, 7711–7722. https://doi.org/10.1002/slct.201701195.
F. T. Johra, W. G. Jung, and J. Serb (2018). Chem. Soc. 82 (4), 503–513. https://doi.org/10.2298/JSC170810117T.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
There is no conflict of interest to declare.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Shaabanzadeh, Z., Khorshidi, A., Moafi, H.F. et al. CuS NPs/Zeolite A/ZIF-8 Dual-Action Composite for Removal of Methylene Blue from Aqueous Solutions. J Clust Sci 34, 487–499 (2023). https://doi.org/10.1007/s10876-022-02241-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10876-022-02241-6