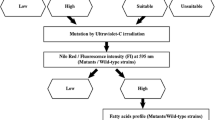
Abstract
Optimization of mass microalgae cultures is required for the efficient production of biodiesel feedstock in terms of total fatty acid (TFA) content and a conducive fatty acid profile. A mutant strain of Nannochloropsis sp. (MT-I5), as modified via random mutagenesis and flow cytometric cell sorting, was investigated in both a single- and two-stage cultivation using 250 L laboratory raceway ponds. Culture was based on photoautotrophic biomass production (stage 1) followed by a switch to photomixotrophic growth induced by adding sodium acetate (2 mM) (stage 2). The biomass yield of the mutant in two-stage cultivation was maintained at a level similar to that of the one-stage photoautotrophic culture, but TFA content was increased by 2.3-fold. The fatty acid profile of MT-I5 also had an increased level of desirable saturated fatty acids (SFA) for use as a biodiesel feedstock, i.e. from 43 to 48 % of TFA, as well as a decreased level of less desirable polyunsaturated fatty acids (PUFA), i.e. from 22 to 7 % of TFA. The two-stage cultivation process is of interest for the mass culture of microalgae for biofuel feedstocks, as biomass productivity can be maximized during the first stage of culture until N-starvation is achieved, followed by the enhanced synthesis of SFA in the second stage of culture by adding sodium acetate as a fixed-carbon source.


Similar content being viewed by others
References
Bhatnagar A, Chinnasamy S, Singh M, Das KC (2011) Renewable biomass production by mixotrophic algae in the presence of various carbon sources and wastewaters. Appl Energy 88:3425–3431
Borowitzka MA, Moheimani NR (2013) Sustainable biofuels from algae. Mitig Adapt Strateg Glob Chang 18:13–25
Chi Z, Liu Y, Frear C, Chen S (2009) Study of a two-stage growth of DHA-producing marine algae Schizochytrium limacinum SR21 with shifting dissolved oxygen level. Appl Microbiol Cell Physiol 81:1141–1148
Chiaramonti D, Prussi M, Casini D, Tredici MR, Rodolfi L, Bassi N, Zittelli GC, Bondioli P (2013) Review of energy balance in raceway ponds for microalgae cultivation: re-thinking a traditional system is possible. Appl Energy 102:101–111
Collos Y, Mornet F, Sciandra A, Waser N, Larson A, Harrison PJ (1999) An optical method for the rapid measurement of micromolar concentrations of nitrate in marine phytoplankton cultures. J Appl Phycol 11:179–184
Converti A, Casazza AA, Ortiz EY, Perego P, Del Borghi M (2009) Effect of temperature and nitrogen concentration on the growth and lipid content of Nannochloropsis oculata and Chlorella vulgaris for biodiesel production. Chem Eng Process 48:1146–1151
Demirbas MF (2011) Biofuels from algae for sustainable development. Appl Energy 88:3473–3480
Doan TTY, Obbard JP (2012) Enhanced intracellular lipid in Nannochloropsis sp. via random mutagenesis and flow cytometric cell sorting. Algal Res 1:17–21
Feng FY, Yang W, Jiang GZ, Xu YN, Kuang TY (2005) Enhancement of fatty acid production of Chlorella sp (Chlorophyceae) by addition of glucose and sodium thiosulphate to culture medium. Process Biochem 40:1315–1318
Griffiths MJ, Harrison STL (2009) Lipid productivity as a key characteristic for choosing algal species for biodiesel production. J Appl Phycol 21:493–507
Guillard RRL (1975) Culture of phytoplankton for feeding marine invertebrates. In: Smith WL, Chaley MH (eds) Culture of Marine Invertebrate Animals. Plenum Press, New York, pp 26–60
Hu HH, Gao KS (2003) Optimization of growth and fatty acid composition of a unicellular marine picoplankton, Nannochloropsis sp., with enriched carbon sources. Biotech Lett 25:421–425
Huntley ME, Redalje DG (2007) CO2 mitigation and renewable oil from photosynthetic microbes: a new appraisal. Mitig Adapt Strateg Glob Chang 12:573–608
Knothe G (2013) Production and properties of biodiesel from algal oils. In: Borowitzka MA, Moheimani NR (eds) Algae for Biofuels and Energy. Springer, Dordrecht, pp 207–221
Li YQ, Horsman M, Wang B, Wu N, Lan CQ (2008) Effects of nitrogen sources on cell growth and lipid accumulation of green alga Neochloris oleoabundans. Appl Microbiol Biotechnol 81:629–636
Liang YN, Sarkany N, Cui Y (2009) Biomass and lipid productivities of Chlorella vulgaris under autotrophic, heterotrophic and mixotrophic growth conditions. Biotech Lett 31:1043–1049
Moazami N, Ashori A, Ranjbar R, Tangestani M, Eghtesadi R, Nejad AS (2012) Large-scale biodiesel production using microalgae biomass of Nannochloropsis. Biomass Bioenergy 39:449–453
Oh SH, Kwon MC, Choi WY, Seo YC, Kim GB, Kang DH, Lee SY, Lee HY (2010) Long-term outdoor cultivation by perfusing spent medium for biodiesel production from Chlorella minutissima. J Biosci Bioeng 110:194–200
Pruvost J, Van Vooren G, Cogne G, Legrand J (2009) Investigation of biomass and lipids production with Neochloris oleoabundans in photobioreactor. Bioresour Technol 100:5988–5995
Pyle D, Wen Z (2008). Production of omega-3 fatty acid-rich microalgae from biodiesel derived crude glycerol: effects of glycerol impurities on algal growth and DHA production. Paper presented at the American Society of Agricultural and Biological Engineers Annual International Meeting 2008
Qiao H, Wang G (2009) Effect of carbon source on growth and lipid accumulation in Chlorella sorokiniana GXNN01. Chin J Oceanol Limnol 27:762–768
Rai MP, Nigam S, Sharma R (2013) Response of growth and fatty acid compositions of Chlorella pyrenoidosa under mixotrophic cultivation with acetate and glycerol for bioenergy application. Biomass Bioenergy 58:251–257
Rattanapoltee P, Kaewkannetra P (2014) Utilization of agricultural residues of pineapple peels and sugarcane bagasse as cost-saving raw materials in Scenedesmus acutus for lipid accumulation and biodiesel production. Appl Biochem Biotechnol 173:1495–1510
Rodolfi L, Zittelli GC, Bassi N, Padovani G, Biondi N, Bonini G, Tredici MR (2009) Microalgae for oil: strain selection, induction of lipid synthesis and outdoor mass cultivation in a low-cost photobioreactor. Biotechnol Bioeng 102:100–112
Schenk P et al (2008) Second generation biofuels: high-efficiency microalgae for biodiesel production. BioEnergy Res 1:20–43
Sheehan J, Dunahay T, Benemann J, Roessler P (1998) Look back at the U.S. Department of Energy’s Aquatic Species Program: biodiesel from algae. NREL/TP-580-24190, National Renewable Energy Lab., Golden, CO. Department of Energy, Washington, USA
Solovchenko AE, Khozin-Goldberg I, Didi-Cohen S, Cohen Z, Merzlyak MN (2008) Effects of light intensity and nitrogen starvation on growth, total fatty acids and arachidonic acid in the green microalga Parietochloris incisa. J Appl Phycol 20:245–251
Tomabene TG, Holzer G, Lien S, Burris N (1983) Lipid composition of the nitrogen starved green alga Neochloris oleoabundans. Enzyme Microb Technol 5:435–440
Williams PJL, Laurens LML (2010) Microalgae as biodiesel & biomass feedstocks: review & analysis of the biochemistry, energetics & economics. Energy Environ Sci 3:554–590
Yang C, Hua Q, Shimizu K (2000) Energetics and carbon metabolism during growth of microalgal cells under photoautotrophic, mixotrophic and cyclic light-autotrophic/dark-heterotrophic conditions. Biochem Eng J 6:87–102
Acknowledgements
Funding of this research project was provided by the Agency for Science, Technology and Research of Singapore (A*STAR). The authors are also grateful to the additional instrumental support provided by the Tropical Marine Science Institute at St John’s Island (Singapore).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Doan, Y.T.T., Obbard, J.P. Two-stage cultivation of a Nannochloropsis mutant for biodiesel feedstock. J Appl Phycol 27, 2203–2208 (2015). https://doi.org/10.1007/s10811-014-0490-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10811-014-0490-4