Abstract
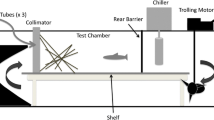
The functional response describes the relationship between feeding rate and prey density, and is important ecologically as it describes how foraging behaviour may change in response to food availability. The effects of habitat complexity and food item size were experimentally tested on the foraging parameters and functional responses of the freshwater fish roach Rutilus rutilus. Habitat complexity was varied through the manipulation of substrate and turbidity, and food item size was varied by using fishmeal pellets in two sizes. As water turbidity and substrate complexity increased, the reaction distance and consumption rate (per number) significantly decreased. Increased food item size significantly decreased consumption rates (per number) but had no influence on any other foraging parameter. Analysis of the interactions between substrate complexity, turbidity and food item size revealed food item size had the greatest influence on consumption rate (per number). Turbidity had the least effect on all the foraging parameters tested. Across all experiments, the functional responses were best described by the Type II response, a relatively consistent finding for R. rutilus. These outputs reveal that fish foraging behaviours and functional responses are highly context dependent, varying with environmental parameters and the availability of food resources of different sizes.



Similar content being viewed by others
References
Abrams, P. A., 1982. Functional responses of optimal foragers. The American Naturalist 120: 382–390.
Aksnes, D. L. & C. W. Utne, 1997. A revised model of visual range in fish. Sarsia 82: 137–147.
Alexander, M. E., J. T. A. Dick, N. E. O’Connor, N. R. Haddaway & K. Farnsworth, 2012. Functional responses of the intertidal amphipod Echinogammarus marinus: effects of prey supply, model selection and habitat complexity. Marine Ecology Progress Series 468: 191–202.
Alexander, M. E., J. T. A. Dick & N. E. O’Connor, 2013. Trait-mediated indirect interactions in a marine intertidal system as quantified by functional responses. Oikos 122: 1521–1531.
Allouche, S. & P. Gaudin, 2001. Effects of avian predation threat, water flow and cover on growth and habitat use by chub, Leuciscus cephalus, in an experimental stream. Oikos 94: 481–492.
Bean, C. W. & I. J. Winfield, 1983. Habitat use and activity patterns of roach (Rutilus rutilus (L.)), rudd (Scardinius erythrophthalmus (L.)), perch (Perca fluviatilis (L.)) and pike (Esox lucius (L.)) in the laboratory: the role of predation threat and structural complexity. Ecology of Freshwater Fish 4: 37–46.
Bogacka-kapusta, E. & A. Kapusta, 2007. The diet of roach, Rutilus rutilus (L.), and bleak Alburnus alburnus (L.) larvae and fry in the shallow littoral zone of a heated lake. Archives of Polish Fisheries 15: 401–413.
Bollache, L., J. T. A. Dick, K. D. Farnsworth & W. I. Montgomery, 2008. Comparison of the functional responses of invasive and native amphipods. Biology Letters 4: 166–169.
Britton, J. R., J. Pegg, D. Baker & C. Williams, 2012. Do lower feeding rates result in reduced growth of a cyprinid fish infected with the Asian tapeworm? Ecology of Freshwater Fish 21: 172–175.
Brabrand, A. & B. Faafeng, 1993. Habitat shift in roach (Rutilus rutilus) induced by pikeperch (Stizostedion lucioperca) introduction: predation risk versus pelagic behaviour. Oecologia 95: 38–46.
Buckel, J. A. & A. W. Stoner, 2000. Functional response and switching behavior of young-of-the-year piscivorous bluefish. Journal of Experimental Marine Biology and Ecology 245: 25–41.
Chick, J. H. & C. C. McIvor, 1997. Habitat selection by three littoral zone fishes: effects of predation pressure, plant density and macrophyte type. Ecology of Freshwater Fish 6: 27–35.
Copp, G. & S. Doherty, 1997. Microhabitat use of fish larvae and 0 + juveniles in a highly regulated section of the River Great Ouse. Regulated Rivers: Research & Management 13: 267–276.
Dick, J. T. A., K. Gallagher, S. Avlijas, H. C. Clarke, S. E. Lewis, S. Leung & A. Ricciardi, 2012. Ecological impacts of an invasive predator explained and predicted by comparative functional responses. Biological Invasions 15: 837–846.
Diehl, S., 1988. Foraging efficiency of three freshwater fishes: effects of structural complexity and light. Oikos 53: 207.
Elliott, J. M., 2003. Interspecific interference and the functional response of four species of carnivorous stoneflies. Freshwater Biology 48: 1527–1539.
Elvira, B. & A. Almodovar, 2001. Freshwater fish introductions in Spain: facts and figures at the beginning of the 21st century. Journal of Fish Biology 59: 323–331.
Event; Bournemouth University (Version 1.0 Software). Available from: http://individualecology.bournemouth.ac.uk/software.html. Last accessed 24 September 2013.
Galarowicz, T. L. & D. H. Wahl, 2005. Foraging by a young-of-the-year piscivore: the role of predator size, prey type, and density. Canadian Journal of Fisheries and Aquatic Sciences 62: 2330–2342.
Garner, P., 1996. Microhabitat use and diet of 0+ cyprinid fishes in a lentic, regulated reach of the River Great Ouse, England. Journal of Fish Biology 48: 367–382.
Gotceitas, V., 1990. Variation in plant stem density and its effects on foraging success of juvenile bluegill sunfish. Environmental Biology of Fishes 27: 63–70.
Gregg, W. & F. Rose, 1985. Influences of aquatic macrophytes on invertebrate community structure, guild structure, and microdistribution in streams. Hydrobiologia 128: 45–56.
Grenouillet, G. & D. Pont, 2001. Juvenile fishes in macrophyte beds: influence of food resources, habitat structure and body size. Journal of Fish Biology 59: 939–959.
Hassell, M. P., J. H. Lawton & J. R. Beddington, 1977. Sigmoid functional responses by invertebrate predators and parasitoids. Journal of Animal Ecology 46: 249–262.
Heck Jr, K. L. & L. B. Crowder, 1991. Habitat Structure and Predator—Prey Interactions in Vegetated Aquatic Systems. In Bell, S. S., E. D. McCoy & H. Muchinsky (eds), Habitat Structure: The Physical Arrangement of Objects in Space. Chapman and Hall, New York: 281–299.
Henson, S. M. & T. G. Hallam, 1995. Optimal feeding via constrained processes. Journal of Theoretical Biology 176: 170–177.
Hjelm, J., G. H. van de Weerd & F. A. Sibbing, 2003. Functional link between foraging performance, functional morphology, and diet shift in roach (Rutilus rutilus). Canadian Journal of Fisheries and Aquatic Sciences 60: 700–709.
Hölker, F. & B. Breckling, 2005. A spatiotemporal individual-based fish model to investigate emergent properties at the organismal and the population level. Ecological Modelling 186: 406–426.
Hölker, F., S. S. Haertel, S. Steiner & T. Mehner, 2002. Effects of piscivore-mediated habitat use on growth, diet and zooplankton consumption of roach: an individual-based modelling approach. Freshwater biology 47: 2345–2358.
Holling, C. S., 1959. Some characteristics of simple types of predation and parasitism. The Canadian Entomologist 91: 385–398.
Holling, C. S., 1966. The functional response of predators to prey density and its role in mimicry and population regulation. Memoirs of the Entomological Society of Canada 48: 5–60.
Hurlbert, S. H., 1984. Pseudoreplication and the design of ecological field experiments. Ecological Monographs 54: 187.
Ioannou, C. C., C. R. Tosh, L. Neville & J. Krause, 2007. The confusion effect from neural networks to reduced predation risk. Behavioral Ecology 19: 126–130.
Jeppesen, E., M. Meerhoff, K. Holmgren, I. González-Bergonzoni, F. Teixeira-de Mello, S. A. J. Declerck, L. Meester, M. Søndergaard, T. L. Lauridsen, R. Bjerring, J. M. Conde-Porcuna, N. Mazzeo, C. Iglesias, M. Reizenstein, H. J. Malmquist, Z. Liu, D. Balayla & X. Lazzaro, 2010. Impacts of climate warming on lake fish community structure and potential effects on ecosystem function. Hydrobiologia 646: 73–90.
Jeschke, J. M. & K. Hohberg, 2008. Predicting and testing functional responses: an example from a tardigrade–nematode system. Basic and Applied Ecology 9: 145–151.
Jeschke, J. M., M. Kopp & R. Tollrian, 2002. Predator functional Responses: discriminating between handling and digesting prey. Ecological Monographs 72: 95–112.
Jeschke, J. M., M. Kopp & R. Tollrian, 2004. Consumer-food systems: why Type I functional responses are exclusive to filter feeders. Biological Reviews 79: 337–349.
Johansson, L. & L. Persson, 1986. The Fish Community of Temperate Eutrophic Lakes. In Riemann, B. & M. Sondergaard (eds), Carbon Dynamics in Eutrophic, Temper-ate Lakes. Elsevier, Amsterdam: 237–266.
Koski, M. L. & B. M. Johnson, 2002. Functional response of kokanee salmon (Oncorhynchus nerka) to Daphnia at different light levels. Canadian Journal of Fisheries and Aquatic Sciences 716: 707–716.
Lappalainen, J., A. S. Tarkan & C. Harrod, 2008. A meta-analysis of latitudinal variations in life-history traits of roach, Rutilus rutilus, over its geographical range: linear or non- linear relationships? Freshwater Biology 53: 1491–1501.
Lipcius, R. N. & A. H. Hines, 1986. Variable functional responses of a marine predator in dissimilar homogeneous microhabitats. Ecology 67(1361–1371): 1.
Milinski, M. & R. Heller, 1978. Influence of a predator on the optimal foraging behaviour of sticklebacks (Gasterosteus aculeatus L.). Nature 275: 642–644.
Mills, N. J., 1982. Satiation and the functional response: a test of a new model. Ecological Entomology 7: 305–315.
Morgan, R. A. & J. S. Brown, 1996. Using giving-up densities to detect search images. American Naturalist 148: 1059–1074.
Murray, G. P. D., R. A. Stillman, R. E. Gozlan & J. R. Britton, 2013. Experimental predictions of the functional response of a freshwater fish. Ethology 119: 751–761.
Nurminen, L., Z. Pekcan-Hekim & J. Horppila, 2010. Feeding efficiency of planktivorous perch Perca fluviatilis and roach Rutilus rutilus in varying turbidity: an individual-based approach. Journal of Fish Biology 76: 1848–1855.
Oksanen, L. & P. Lundberg, 1995. Optimization of reproductive effort and foraging time in mammals: the influence or resource level and predation risk. Evolutionary Ecology 9: 45–46.
Oyugi, D. O., J. Cucherousset, D. J. Baker & J. R. Britton, 2012a. Effects of temperature on the foraging and growth rate of juvenile common carp, Cyprinus carpio. Journal of Thermal Biology 37: 89–94.
Oyugi, D. O., J. Cucherousset & J. R. Britton, 2012b. Temperature-dependent feeding interactions between two invasive fishes competing through interference and exploitation. Reviews in Fish Biology & Fisheries 22: 499–508.
Persson, L., 1983. Food consumption and the significance of detritus and algae to intraspecific competition in roach Rutilus rutilus in a shallow eutrophic lake. Oikos 41: 118–125.
Persson, L., 1987. Effects of habitat and season on competitive interactions between roach (Rutilus rutlius) and perch (Perca fluviatilis). Oecologia 73: 170–177.
Podolszky, D., F. Uiblein & H. Winkler, 1995. Visual habitat choice in cyprinid fishes: an experimental approach. Ecology of Freshwater Fish 4: 160–167.
Priyadarshana, T., T. Asaeda & J. Manatunge, 2006. Hunger-induced foraging behavior of two cyprinid fish: Pseudorasbora parva and Rasbora daniconius. Hydrobiologia 568: 341–352.
R Development Core Team 2012. R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. ISBN 3-900051-07-0, URL http://www.R-project.org/.
Real, L. A., 1977. The kinetics of functional response. The American Naturalist 11: 289–300.
Railsback, S. F. & B. C. Harvey, 2002. Analysis of habitat-selection rules using an individual-based model. Ecology 83: 1817–1830.
Savino, J. F. & R. A. Stein, 1989. Behavior of fish predators and their prey: habitat choice between open water and dense vegetation. Environmental Biology of Fishes 24: 287–293.
Simpson, A. L., N. B. Metcalfe, F. A. Huntingford & J. E. Thorpe, 1996. Pronounced seasonal differences in appetite of atlantic salmon parr, Salmo salar: effects of nutritional state and life-history strategy. Functional Ecology 10: 760–767.
Solomon, M. E., 1949. The natural control of animal populations. Journal of Animal Ecology 18: 1–35.
Stillman, R. A., 2008. MORPH—an individual-based model to predict the effect of environmental change on foraging animal populations. Ecological Modelling 216: 265–276.
Sweka, J. A. & K. J. Hartman, 2003. Reduction of reactive distance and foraging success in smallmouth bass, Micropterus dolomieu, exposed to elevated turbidity levels. Environmental Biology of Fishes 67: 341–347.
Tosh, C. R., J. Krause & G. D. Ruxton, 2009. Basic features, conjunctive searches, and the confusion effect in predator–prey interactions. Behavioral Ecology and Sociobiology 63: 473–475.
Utne-Palm, A. C., 1999. The effect of prey mobility, prey contrast, turbidity and spectral composition on the reaction distance of Gobiusculus flavescens to its planktonic prey. Journal of Fish Biology 54: 1244–1258.
Vahl, W. K., F. J. Van Der Meer, D. V. Weissing, D. V. Van Dullenmen & T. Piersma, 2005. The mechanisms of interference competition: two experiments on foraging waders. Behavioral Ecology 16: 845–855.
Van Den Berg, C., J. G. M. Van Den Boogaart, F. A. Sibbing & J. W. M. Osse, 1994. Zooplankton feeding in common Bream (Abramis brama), White Bream (Blicca Bjoerkna) and Roach (Rutilus rutilus): experiments, models and energy intake. Netherlands Journal of Zoology 44: 15–42.
Vollset, K. W. & K. M. Bailey, 2011. Interplay of individual interactions and turbidity affects the functional response of three-spined sticklebacks Gasterosteus aculeatus. Journal of Fish Biology 78: 1954–1964.
Wankowski, J. W. J. & J. E. Thorpe, 1979. The role of food particle size in the growth of juvenile Atlantic salmon (Salmo salar L.). Journal of Fish Biology 14: 351–370.
Wanzenböck, J., 1995. Changing handling times during feeding and consequences for prey size selection of 0 + zooplanktivorous fish. Oecologia 104: 372–378.
Wanzenböck, J., M. Zaunreiter, C. M. Wahl & D. L. G. Noakes, 1996. Comparison of behavioural and morphological measures of visual resolution during ontogeny of roach (Rutilus rutilus) and yellow perch (Perca flavescens). Canadian journal of Fisheries and Aquatic Sciences 53: 1506–1512.
Warfe, D. M. & L. A. Barmuta, 2004. Habitat structural complexity mediates the foraging success of multiple predator species. Oecologia 141: 171–178.
Werner, E. E., J. F. Gilliam, D. J. Hall & G. G. Mittelbach, 1983. An experimental test of the effects of predation risk on habitat use in fish. Ecology 64: 1540–1548.
Winfield, I. J., J. M. Fletcher & J. B. James, 2011. Invasive fish species in the largest lakes of Scotland, Northern Ireland, Wales and England: the collective UK experience. Hydrobiologia 660: 93–103.
Winkler, H. & C. P. Orellana, 1992. Functional responses of five cyprinid species to planktonic prey. Environmental Biology of Fishes 33: 53–62.
Zamor, R. M. & G. D. Grossman, 2007. Turbidity affects foraging success of drift-feeding Rosyside Dace. Transactions of the American Fisheries Society 136: 167–176.
Acknowledgments
We thank Calverton fish farm for supplying the fish used in the trials. We would also like to acknowledge the contributions of two anonymous referees for improving the manuscript. This study was funded by the Environment Agency and Bournemouth University.
Author information
Authors and Affiliations
Corresponding author
Additional information
Handling editor: Odd Terje Sandlund
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Murray, G.P.D., Stillman, R.A. & Britton, J.R. Habitat complexity and food item size modify the foraging behaviour of a freshwater fish. Hydrobiologia 766, 321–332 (2016). https://doi.org/10.1007/s10750-015-2465-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10750-015-2465-0