Abstract
Objectives
Resting cells of Pseudonocardia sp. KCTC 1029BP were used for the bioconversion of vitamin D3 to calcifediol which is widely used to treat osteomalacia and is industrially produced by chemical synthesis.
Results
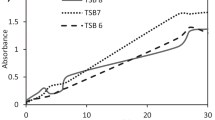
To obtain the maximum bioconversion yield of calcifediol by the microbial conversion of vitamin D3, a two-step optimization process was used, including the Plackett–Burman and the central composite designs. Six variables, namely agitation speed, aeration rate, resting cell concentration, vitamin D3 concentration, temperature, and pH, were monitored. Of these, aeration rate, resting cell concentration, and temperature were selected as key variables for calcifediol production and were optimized using the central composite design. Optimal bioconversion conditions obtained were as follows: aeration rate of 0.2 vvm, resting cell concentration of 4.7 % w/v, and temperature of 33 °C.
Conclusion
Using the optimal conditions, 356 mg calcifediol l−1 was obtained with a bioconversion yield of 59.4 % in a 75 l fermentor. These are the highest values reported to date.



Similar content being viewed by others
References
Anderson MJ, Whitcomb PJ (2005) RSM simplified: optimizing processes using response surface methods for design of experiments. Productivity Press, Boca Raton
Andrews DR, Barton DH, Hesse RH, Pechet MM (1986) Synthesis of 25-hydroxy-and 1 alpha., 25-dihydroxy vitamin D3 from vitamin D2 (calciferol). J Org Chem 51:4819–4828
Bar A, Rosenberg J, Perlman R, Hurwitz S (1987) Field rickets in turkeys: relationship to vitamin D. Poult Sci 66:68–72
Brooks MH, Bell NH, Love L, Stern PH, Orfei E, Queener SF, Hamstra AJ, DeLuca HF (1978) Vitamin-D-dependent rickets type II: resistance of target organs to 1, 25-dihydroxyvitamin D. New Engl J Med 298:996–999
Clements M, Johnson L, Fraser D (1987) A new mechanism for induced vitamin D deficiency in calcium deprivation. Nature 32:62–66
de Carvalho CC (2011) Enzymatic and whole cell catalysis: finding new strategies for old processes. Biotechnol Adv 29:75–83
DeLuca HF, Schnoes HK (1983) Vitamin D: recent advances. Annu Rev Biochem 52:411–439
Eisman J, Shepard R, DeLuca H (1977) Determination of 25-hydroxyvitamin D2 and 25-hydroxyvitamin D3 in human plasma using high-pressure liquid chromatography. Anal Biochem 80:298–305
Flores ER, Pérez F, de la Torre M (1997) Scale-up of Bacillus thuringiensis fermentation based on oxygen transfer. J Ferment Bioeng 83:561–564
Gangadharan D, Sivaramakrishnan S, Nampoothiri KM, Sukumaran RK, Pandey A (2008) Response surface methodology for the optimization of α amylase production by Bacillus amyloliquefaciens. Biores Technol 99:4597–4602
Hatakeyama S, Sugawara K, Numata H, Takano S (1991) A novel convergent synthesis of (+)-1α, 25-dihydroxyvitamin D3 using a chromium (II)-mediated coupling reaction. J Org Chem 56:461–463
Imoto N, Nishioka T, Tamura T (2011) Permeabilization induced by lipid II-targeting lantibiotic nisin and its effect on the bioconversion of vitamin D3 to 25-hydroxyvitamin D3 by Rhodococcus erythropolis. Biochem Biophys Res Commun 405:393–398
Kametani T, Furuyama H (1987) Synthesis of vitamin D3 and related compounds. Med Res Rev 7:147–171
Kang D, Lee H, Park J, Bang J, Hong S, Kim T (2006) Optimization of culture conditions for the bioconversion of vitamin D3 to 1α, 25-dihydroxyvitamin D3 using Pseudonocardia autotrophica ID 9302. Biotechnol Bioprocess Eng 11:408–413
Kang D, Im J, Kang J, Kim K (2015) Whole cell bioconversion of vitamin D3 to calcitriol using Pseudonocardia sp. KCTC 1029BP. Bioproc Biosyst Eng. doi:10.1007/s00449-015-1368-9
Kulprecha S, Ueda T, Nihira T, Yoshida T, Taguchi H (1985) Optimum conditions for ursodeoxycholic acid production from lithocholic acid by Fusarium equiseti M41. Appl Environ Microbiol 49:338–344
Lau J, Tran C, Licari P, Galazzo J (2004) Development of a high cell-density fed-batch bioprocess for the heterologous production of 6-deoxyerythronolide B in Escherichia coli. J Biotechnol 110:95–103
Lund J, DeLuca HF (1966) Biologically active metabolite of vitamin D3 from bone, liver, and blood serum. J Lipid Res 7:739–744
Lythgoe B (1980) Simonsen Lecture. Synthetic approaches to vitamin D and its relatives. Chem Soc Rev 9:449–475
Madhok T, DeLuca H (1979) Characteristics of the rat liver microsomal enzyme system converting cholecalciferol into 25-hydroxycholecalciferol. Evidence for the participation of cytochrome p-450. Biochem J 184:491–499
Mandenius C, Brundin A (2008) Bioprocess optimization using design of experiments methodology. Biotechnol Prog 24:1191–1203
Plackett R, Burman J (1946) The design of optimum multifactorial experiments. Biometrika 33:305–325
Raissi S, Farsani R (2009) Statistical process optimization through multi-response surface methodology. World Acad Sci Eng Technol 51:267–271
Sasaki J, Mikami A, Mizoue K, Omura S (1991) Transformation of 25- and 1α-hydroxyvitamin D3 to 1α, 25-dihydroxyvitamin D3 by using Streptomyces sp. strains. Appl Environ Microbiol 57:2841–2846
Sasaki J, Miyazaki A, Saito M, Adachi T, Mizoue K, Hanada K, Omura S (1992) Transformation of vitamin D3 to 1α, 25-dihydroxyvitamin D3 via 25-hydroxyvitamin D3 using Amycolata sp. strains. Appl Microbiol Biotechnol 38:152–157
Seino Y, Tanaka H, Yamaoka K, Yabuuchi H (1987) Circulating 1α, 25-dihydroxyvitamin D levels after a single dose of 1α, 25-dihydroxyvitamin D3 or 1α-hydroxyvitamin D3 in normal men. Bone Miner 2:479–485
Soares J, Kerr J, Gray R (1995) 25-hydroxycholecalciferol in poultry nutrition. Poult Sci 74:1919–1934
Sreekumar G, Krishnan S (2010) Enhanced biomass production study on probiotic Bacillus subtilis SK09 by medium optimization using response surface methodology. Afr J Biotechnol 9:8078–8084
Takeda K, Asou T, Matsuda A, Kimura K, Okamura K, Okamoto R, Sasaki J, Adachi T, Omura S (1994) Application of cyclodextrin to microbial transformation of vitamin D3 to 25-hydroxyvitamin D3 and 1α, 25-dihydroxyvitamin D3. J Ferment Bioeng 78:380–382
Uzura A, Katsuragi T, Tani Y (2001) Conversion of various aromatic compounds by resting cells of Fusarium moniliforme strain MS31. J Biosci Bioeng 92:381–384
Vanmaele L, De Clercq P, Vandewalle M (1985) An efficient synthesis of 1α, 25-dihydroxy vitamin D3. Tetrahedron 41:141–144
Vieth R (1999) Vitamin D supplementation, 25-hydroxyvitamin D concentrations, and safety. Am J Clin Nutr 69:842–856
Wang Z, Zhuge J, Fang H, Prior B (2001) Glycerol production by microbial fermentation: a review. Biotechnol Adv 19:201–223
Xiong Z, Tu X, Tu G (2008) Optimization of medium composition for actinomycin X2 production by Streptomyces spp. JAU4234 using response surface methodology. J Ind Microbiol Biotechnol 35:729–734
Yasutake Y, Nishioka T, Imoto N, Tamura T (2013) A single mutation at the ferredoxin binding site of p450 Vdh enables efficient biocatalytic production of 25 hydroxyvitamin D3. ChemBioChem 14:2284–2291
Yee L, Blanch H (1993) Defined media optimization for growth of recombinant Escherichia coli X90. Biotechnol Bioeng 41:221–230
Acknowledgments
KHK acknowledges the Grant support by the Ministry of Trade, Industry & Energy (10048684) and the facility support at the Korea University Food Safety Hall for the Institute of Biomedical Science and Food Safety.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kang, DJ., Im, JH., Kang, JH. et al. Bioconversion of vitamin D3 to calcifediol by using resting cells of Pseudonocardia sp.. Biotechnol Lett 37, 1895–1904 (2015). https://doi.org/10.1007/s10529-015-1862-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10529-015-1862-9