Abstract
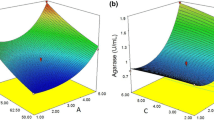
Agarase is a promising biocatalyst for several industrial applications. Agarase production was evaluated by the marine fungus Dendryphiella arenaria utilizing Palisada perforata as a basal substrate in semi-solid state fermentation. Seaweed biomass, glucose, and sucrose were the most significant parameters affecting agarase production, and their levels were further optimized using Box-Behnken design. The maximum agarase activity was 7.69 U/mL. Agarase showed a degree of thermostability with half-life of 99 min at 40 °C, and declining to 44.72 min at 80 °C. Thermodynamics suggested an important process of protein aggregation during thermal inactivation. Additionally, the enzymatic saccharification of the seaweed biomass using crude agarase was optimized with respect to biomass particle size, solid/liquid ratio, and enzyme loadings. The amount of biosugars obtained after optimization was 26.15 ± 1.43 mg/g. To the best of our knowledge, this is the first report on optimization of agarase in D. arenaria.



Similar content being viewed by others
References
Abdel-Gawad KM, Hifney AF, Issa AA, Gomaa M (2014) Spatio-temporal, environmental factors, and host identity shape culturable-epibiotic fungi of seaweeds in the Red Sea, Egypt. Hydrobiologia 740:37–49
Abdel-Gawad KM, Hifney AF, Fawzy MA, Gomaa M (2017) Technology optimization of chitosan production from Aspergillus niger biomass and its functional activities. Food Hydrocoll 63:593–601
Choi HJ, Hong JB, Park JJ, Chi W-J, Kim M-C, Chang Y-K, Hong S-K (2011) Production of agarase from a novel Micrococcus sp. GNUM-08124 strain isolated from the East Sea of Korea. Biotechnol Bioprocess Eng 16:81–88
Cui F, Dong S, Shi X, Zhao X, Zhang X-H (2014) Overexpression and characterization of a novel thermostable β-agarase YM01-3, from marine bacterium Catenovulum agarivorans YM01T. Mar Drugs 12:2731–2747
de Castro RJS, Ohara A, Nishide TG, Albernaz JRM, Soares MH, Sato HH (2015) A new approach for proteases production by Aspergillus niger based on the kinetic and thermodynamic parameters of the enzymes obtained. Biocatal Agric Biotechnol 4:199–207
Fawzy MA, Gomaa M, Hifney AF, Abdel-Gawad KM (2017) Optimization of alginate alkaline extraction technology from Sargassum latifolium and its potential antioxidant and emulsifying properties. Carbohydr Polym 157:1903–1912
Fernández LE, Valiente OG, Mainardi V, Bello JL, Vélez H, Rosado A (1989) Isolation and characterization of an antitumor active agar-type polysaccharide of Gracilaria dominguensis. Carbohydr Res 190:77–83
Fu XT, Kim SM (2010) Agarase: review of major sources, categories, purification method, enzyme characteristics and applications. Mar Drugs 8:200–218
Gomaa M, Hifney AF, Fawzy MA, Issa AA, Abdel-Gawad KM (2015) Biodegradation of Palisada perforata (Rhodophyceae) and Sargassum sp. (Phaeophyceae) biomass by crude enzyme preparations from algicolous fungi. J Appl Phycol 27:2395–2404
Gupta V, Trivedi N, Kumar M, Reddy C, Jha B (2013) Purification and characterization of exo-β-agarase from an endophytic marine bacterium and its catalytic potential in bioconversion of red algal cell wall polysaccharides into galactans. Biomass Bioenergy 49:290–298
Han W, Lam WC, Melikoglu M, Wong MT, Leung HT, Ng CL, Yan P, Yeung SY, Lin CSK (2015) Kinetic analysis of a crude enzyme extract produced via solid state fermentation of bakery waste. ACS Sustain Chem Eng 3:2043–2048
Hifney AF, Fawzy MA, Abdel-Gawad KM, Issa AA, Gomaa M (2017) In vitro comparative evaluation of antioxidant activity of hydrophobic and hydrophilic extracts from Algicolous fungi. J Aquat Food Prod Technol 26:124–131
Hifney AF, Fawzy MA, Abdel-Gawad KM, Gomaa M (2016) Industrial optimization of fucoidan extraction from Sargassum sp. and its potential antioxidant and emulsifying activities. Food Hydrocoll 54:77–88
Hu Z, Lin B-K, Xu Y, Zhong M, Liu G-M (2009) Production and purification of agarase from a marine agarolytic bacterium Agarivorans sp. HZ105. J Appl Microbiol 106:181–190
Ishola MM, Jahandideh A, Haidarian B, Brandberg T, Taherzadeh MJ (2013) Simultaneous saccharification, filtration and fermentation (SSFF): a novel method for bioethanol production from lignocellulosic biomass. Bioresour Technol 133:68–73
Issa AA, Hifney AF, Abdel-Gawad KM, Gomaa M (2014) Spatio temporal and environmental factors influencing macroalgal β diversity in the Red Sea, Egypt. Bot Mar 57:99–110
Kim HT, Lee S, Kim KH, Choi I-G (2012) The complete enzymatic saccharification of agarose and its application to simultaneous saccharification and fermentation of agarose for ethanol production. Bioresour Technol 107:301–306
Kim SW, Kim Y-W, Hong C-H, Lyo I-W, Lim H-D, Kim G-J, Shin H-J (2017) Recombinant agarase increases the production of reducing sugars from HCl-treated Gracilaria verrucosa, a red algae. Algal Res. https://doi.org/10.1016/j.algal.2017.01.008
Kobayashi R, Takisada M, Suzuki T, Kirimura K, Usami S (1997) Neoagarobiose as a novel moisturizer with whitening effect. Biosci Biotechnol Biochem 61:162–163
Lakshmikanth M, Manohar S, Patnakar J, Vaishampayan P, Shouche Y, Lalitha J (2006) Optimization of culture conditions for the production of extracellular agarases from newly isolated Pseudomonas aeruginosa AG LSL-11. World J Microbiol Biotechnol 22:531–537
Long M, Yu Z, Xu X (2010) A novel β-agarase with high pH stability from marine Agarivorans sp. LQ48. Mar Biotechnol 12:62–69
Marangoni AG (2003) Enzyme kinetics: a modern approach. Wiley-Interscience, Hoboken
Marín E, Sanchez L, Perez M, Puyol P, Calvo M (2003) Effect of heat treatment on bovine lactoperoxidase activity in skim milk: kinetic and thermodynamic analysis. J Food Sci 68:89–93
Miller GL (1959) Use of dinitrosalicylic acid reagent for determination of reducing sugar. Anal Chem 31:426–428
Oh C, Nikapitiya C, Lee Y, Whang I, Kang D-H, Heo S-J, Choi Y-U, Lee J (2010) Molecular cloning, characterization and enzymatic properties of a novel βeta-agarase from a marine isolate Psudoalteromonas sp. AG52. Braz J Microbiol 41:876–889
Pal A, Khanum F (2011) Covalent immobilization of xylanase on glutaraldehyde activated alginate beads using response surface methodology: characterization of immobilized enzyme. Process Biochem 46:1315–1322
Park J-H, Hong J-Y, Jang HC, Oh SG, Kim S-H, Yoon J-J, Kim YJ (2012) Use of Gelidium amansii as a promising resource for bioethanol: a practical approach for continuous dilute-acid hydrolysis and fermentation. Bioresour Technol 108:83–88
Usov AI, Elashvili MY (1991) Polysaccharides of algae. 44. Investigation of sulfated Galactan from Laurencia nipponica Yamada (Rhodophyta, Rhodomelaceae) using partial reductive hydrolysis. Bot Mar 34:553–560
Wang J, Jiang X, Mou H, Guan H (2004) Anti-oxidation of agar oligosaccharides produced by agarase from a marine bacterium. J Appl Phycol 16:333–340
Wang J, Mou H, Jiang X, Guan H (2006) Characterization of a novel β-agarase from marine Alteromonas sp. SY37–12 and its degrading products. Appl Microbiol Biotechnol 71:833–839
Yun EJ, Kim HT, Cho KM, Yu S, Kim S, Choi I-G, Kim KH (2016) Pretreatment and saccharification of red macroalgae to produce fermentable sugars. Bioresour Technol 199:311–318
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Electronic Supplementary Material
ESM 1
(PDF 365 kb)
Rights and permissions
About this article
Cite this article
Gomaa, M., Hifney, A.F., Fawzy, M.A. et al. Statistical Optimization of Culture Variables for Enhancing Agarase Production by Dendryphiella arenaria Utilizing Palisada perforata (Rhodophyta) and Enzymatic Saccharification of the Macroalgal Biomass. Mar Biotechnol 19, 592–600 (2017). https://doi.org/10.1007/s10126-017-9778-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10126-017-9778-0