Abstract
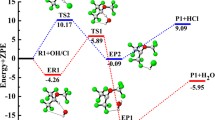
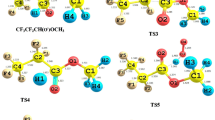
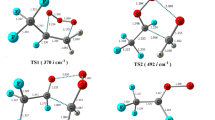
A theoretical study on the mechanism of the thermal decomposition of CF3C(O)OCH2O radical is presented for the first time. Geometry optimization and frequency calculations were performed at the MPWB1K/6–31 + G(d, p) level of theory and energetic information further refined by calculating the energy of the species using G2(MP2) theory. Three plausible decomposition pathways including α-ester rearrangement, reaction with O2 and thermal decomposition (C–O bond scission) were considered in detail. Our results reveal that reaction with O2 is the dominant path for the decomposition of CF3C(O)OCH2O radical in the atmosphere, involving the lowest energy barrier, which is in accord with experimental findings. Our theoretical results also suggest that α-ester rearrangement leading to the formation of trifluoroacetic acid TFA makes a negligible contribution to decomposition of the title alkoxy radical. The thermal rate constants for the above decomposition pathways were evaluated using canonical transition state theory (CTST) at 298 K.


Similar content being viewed by others
References
Molina MJ, Rowland FS (1974) Nature 249:810–814
Farman JD, Gardiner BG, Shanklin JD (1985) Nature 315:207–210
Powell RL (2002) J Fluor Chem 114:237–250
Sekiya A, Misaki S (2000) J Fluor Chem 101:215–221
Ravishankara RA, Turnipseed AA, Jensen NR, Barone S, Mills M, Howark CJ, Solomon S (1994) Science 263:71–75
Bravo I, Dıaz-de-Mera Y, Aranda A, Moreno E, Nutt DR, Marston G (2011) Phys Chem Chem Phys 13:17185–17193
Chen L, Kutsuna S, Tokuhashi K, Sekiya A, Tamai R, Hibino Y (2005) J Phys Chem A 109:4766–4771
Jordan A, Frank H (1999) Environ Sci Technol 33(4):522–527
Blanco MB, Teruel MA (2007) Atmos Environ 41(34):7330–7338
Blanco MB, Bejan I, Barnes I, Wiesen P, Teruel MA (2008) Chem Phys Lett 453:18–23
Blanco MB, Bejan I, Barnes I, Wiesen P, Teruel MA (2010) Environ Sci Technol 44:2354–2359
Stein TNN, Christensen LK, Platz J, Sehested J, Nielsen OJ, Wallington TJ (1999) J Phys Chem A 103:5705–5713
Blanco MB, Barnes I, Teruel MA (2010) J Phys Org Chem 23:950–954
Blanco MB, Rivela C, Teruel MA (2013) Chem Phys Lett 578:33–37
Mishra BK, Chakrabatty AK, Deka RC (2014) Struct Chem 25:463–470
Chakrabatty AK, Mishra BK, Bhattacharjee D, Deka RC (2013) Mol Phys 111:860–867
Mishra BK, Chakrabatty AK, Deka RC (2013) J Mol Model 19:2189–2195
Singh HJ, Tiwari L, Rao PK (2014) Mol Phys 112:1892–1898
Gour NK, Deka RC, Singh HJ, Mishra BK (2014) J Fluor Chem 160:64–71
Orlando JJ, Tyndall GS, Wallington TJ (2003) Chem Rev 103:4657–4689
Vereecken L, Francisco JS (2012) Chem Soc Rev 41:6259–6293
Mishra BK, Lily M, Chakrabartty AK, Bhattacharjee D, Deka RC, Chandra AK (2014) New J Chem 38:2813–2822
Lily M, Mishra BK, Chandra AK (2014) J Fluor Chem 161:51–59
Mishra BK, Lily M, Deka RC, Chandra AK (2014) J Mol Graph Model 50:90–99
Henon E, Bohr F, Gomex NS, Caralp F (2003) Phys Chem Chem Phys 5:5431–5437
Singh HJ, Mishra BK, Gour NK (2010) Theor Chem Acc 125:57–64
Singh HJ, Mishra BK, Rao PK (2012) Can J Chem 90:403–409
Singh HJ, Mishra BK (2011) J Chem Sci 123:733–741
Ferenac MA, Davis AJ, Holloway AS, Dibble TS (2003) J Phys Chem A 107:63–72
Rayez MT, Picquet-Varrault B, Caralp F, Rayez JC (2002) Phys Chem Chem Phys 4:5789–5794
Singh HJ, Mishra BK, Rao PK (2009) Bull Korean Chem Soc 30(12):2973–2978
Zhao Y, Truhlar DG (2004) J Phys Chem A 108:6908–6918
Devi KJ, Chandra AK (2011) Chem Phys Lett 502:23–28
Zeegers-Huyskens T, Lily M, Sutradhar D, Chandra AK (2013) J Phys Chem A 117:8010–8016
Chakrabartty AK, Mishra BK, Bhattacharjee D, Deka RC (2013) J Fluor Chem 154:60–66
Gonzalez C, Schlegel HB (1989) J Chem Phys 90:2154–2161
Curtiss LA, Raghavachari K, Pople JA (1993) J Chem Phys 98:1293–1298
Mishra BK, Lily M, Chakrabartty AK, Deka RC, Chandra AK (2014) J Fluor Chem 159:57–64
Deka RC, Mishra BK (2014) Chem Phys Lett 595:43–47
Lily M, Sutradhar D, Chandra AK (2013) Comp Theor Chem 1022:50–58
Chandra AK (2012) J Mol Model 18:4239–4247
Mishra BK (2014) RSC Adv 4:16759–16764
Frisch MJ et al (2009) GAUSSIAN 09 (Revision B.01). Gaussian Inc, Wallingford, CT
Laidler KJ (2004) Chemical kinetics, 3rd edn. Pearson Education, New Delhi
Chuang YY, Truhlar DG (2000) J Chem Phys 112:1221–1228
Brown RL (1981) J Res Natl Bur Stand 86:357–359
Xiao R, Noerpel M, Luk HL, Wei Z, Spinney R (2014) Int J Quant Chem 114:74–83
Acknowledgments
B.K.M. acknowledges financial support from University Grants Commission, New Delhi in form of a UGC-Dr. D.S. Kothari Fellowship.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Mishra, B.K. Theoretical investigation on the atmospheric fate of CF3C(O)OCH2O radical: alpha-ester rearrangement vs oxidation at 298 K. J Mol Model 20, 2444 (2014). https://doi.org/10.1007/s00894-014-2444-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00894-014-2444-9