Abstract
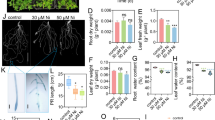
Nitric oxide (NO) is a signaling molecule controlling several steps of plant development and defense process under stress conditions. NO-induced alleviation of manganese (Mn) toxicity was investigated on bean plants submitted for 28 days to 500 µM MnCl2. Manganese excess decreased plant dry weight and elongation and increased levels of reactive oxygen species and lipid peroxidation leading to up-regulation of superoxide dismutase, catalase, and ascorbate peroxidase activities. The inhibitory effects of Mn on plant growth were associated to reduction of light-saturated carbon assimilation (Amax), stomatal conductance (gs), and transpiration (E). By contrast, Mn induced significant increase in the apparent quantum yield (ɸ) and light compensation point (LCP). Interestingly, intracellular CO2 (Ci) remains stable under Mn stress. Concomitantly, leaf membrane lipids have drastically reduced under high Mn concentration. After Mn exposition, leaf fatty acids exhibited a significant loss of linolenic acid, accompanied by an accumulation of palmitoleic, stearic, and linoleic acids leading to alteration of lipid desaturation. NO supply reversed Mn toxicity as evidenced by enhancement of growth biomass and recovery of Amax, E, ɸ, and LCP. Similarly, NO addition has positive effects on leaf lipid content and composition leading to restoration of lipid unsaturation. The modulation of fatty acid composition can be a way to reduce leaf membrane damages and maintain optimal photosynthesis and plant growth. Despite the absence of enough evidences in how NO is involved in lipid and photosynthesis recovery under Mn stress conditions, it is assumed that NO beneficial effects are attributable to NO/Mn cross-talk.








Similar content being viewed by others
Availability of data and material
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
Code availability
Not applicable.
References
Aebi H (1984) Catalase in vitro. Method Enzymol 105:121–126. https://doi.org/10.1016/S0076-6879(84)05016-3
Ahanger MA, Aziz U, Alsahli AA, Alyemeni MN, Ahmad P (2020) Influence of exogenous salicylic acid and nitric oxide on growth, photosynthesis, and ascorbate-glutathione cycle in salt stressed Vigna angularis. Biomolecules 10:42–57. https://doi.org/10.3390/biom10010042
Ahmad P, Ahanger MA, Alyemeni MN, Wijaya L, Alam P (2018) Exogenous application of nitric oxide modulates osmolyte metabolism, antioxidants, enzymes of ascorbate-glutathione cycle and promotes growth under cadmium stress in tomato. Protoplasma 255:79–93. https://doi.org/10.1007/s00709-017-1132-x
Anjum NA, Sofo A, Scopa A, Roychoudhury A, Gill SS, Iqbal M, Lukatkin AS, Pereira E, Duarte AC, Iqbal A (2015) Lipids and proteins major targets of oxidative modifications in abiotic stressed plants. Environ Sci Pollut Res 2:4099–4121. https://doi.org/10.1007/s11356-014-3917-1
Aranda-Caño L, Sánchez-Calvo B, Begara-Morales JC, Chaki M, Mata-Pérez C, Padilla V, Barroso RJ (2019) Post-translational modification of proteins mediated by nitro-fatty acids in plants: nitroalkylation. Plants 8:82–102. https://doi.org/10.3390/plants8040082
Arnon DI (1949) Copper enzymes in isolated chloroplasts: polyphenol oxidase in Beta vulgaris. Plant Physiol 24:1–16. https://doi.org/10.1104/pp.24.1.1
Arora D, Jain P, Singh N, Kaur H, Bhatla SC (2016) Mechanisms of nitric oxide crosstalk with reactive oxygen species scavenging enzymes during abiotic stress tolerance in plants. Free Radic Res 50:291–303. https://doi.org/10.3109/10715762.2015.1118473
Asgher M, Per TS, Masood A, Fatma M, Freschi L, Corpas FJ, Khan NA (2017) Nitric oxide signaling and its crosstalk with other plant growth regulators in plant responses to abiotic stress. Environ Sci Pollut Res 24:2273–2285. https://doi.org/10.1007/s11356-016-7947-8
Astier J, Gross I, Durner J (2018) Nitric oxide production in plants: an update. J Exp Bot 69:3401–3411. https://doi.org/10.1093/jxb/erx420
Barbosa MR, Willadino L, Ulisses C, Camara TR (2014) Plant generation and enzymatic detoxification of reactive oxygen species. Cienc Rural 44:453–460. https://doi.org/10.1590/S0103-84782014000300011
Begara-Morales JC, Chaki M, Valderrama R, Sánchez-Calvo B, Mata-Pérez C, Padilla MN, Corpas FJ, Barroso JB (2018) Nitric oxide buffering and conditional nitric oxide release in stress response. J Exp Bot 69(14):3425–3438. https://doi.org/10.1093/jxb/ery072
Belkhadi A, Hediji H, Abbes Z, Nouairi I, Barhoumi Z, Zarrouk M, Chaïbi W, Djebali W (2010) Effects of exogenous salicylic acid pre-treatment on cadmium toxicity and leaf lipid content in Linum usitatissimum L. Ecotoxicol Environ Saf 73(5):1004–1011. https://doi.org/10.1016/j.ecoenv.2010.03.009
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254. https://doi.org/10.1016/0003-2697(76)90527-3
Chatterjee R, Lassalle L, Gul S, Fuller FD, Young ID, Ibrahim M, De Lichtenberg C, Mun Hon Cheah MH, Zouni A, Messinger J, Vittal K, Yachandra VK, Kern J, Yanoa J (2019) Structural isomers of the S2 state in photosystem II: do they exist at room temperature and are they important for function? Physiol Plant 166:60–72. https://doi.org/10.1111/ppl.12947
Chen F, Shen J, Min D, Ke L, Tian X, Korpelainen H, Li C (2018) Male Populus cathayana than female shows higher photosynthesis and less cellular injury through ABA-induced manganese transporting inhibition under high manganese condition. Trees 32:255–263. https://doi.org/10.1007/s00468-017-1628-1
Chernev P, Fischer S, Hoffmann J, Oliver N, Assunção R, Boram Yu, Burnap RL, Zaharieva I, Nürnberg DJ, Haumann M, Dau H (2020) Light-driven formation of manganese oxide by today’s photosystem II supports evolutionarily ancient manganese-oxidizing photosynthesis. Nat Commun 11:6110. https://doi.org/10.1038/s41467-020-19852-0
Corpas FJ, Del Río LA, Palma JM (2019) Plant peroxisomes at the crossroad of NO and H2O2 metabolism. J Integr Plant Biol 61:803–816. https://doi.org/10.1111/jipb.12772
Dimidchik V (2015) Mechanisms of oxidative stress in plants: from classical chemistry to cell biology. J Environ Exp Bot 109:212–228. https://doi.org/10.1016/j.envexpbot.2014.06.021
Djebali W, Zarrouk M, Brouquisse R, El Kahoui S, Limam F, Ghorbel MH, Chaïbi W (2005) Ultrastructure and lipid alterations induced by cadmium in tomato (Lycopersicon esculentum) chloroplast membranes. Plant Biol 7:258–268. https://doi.org/10.1055/s-2005-837696
El Maani AGD, Delrieu MJ, Roche O, Tremoliere A (1998) Mutant of Chlamydomonas reinhardtii affected in phophatidylglycerol metabolism and thylakoid biogenesis. Plant Physiol Biochem 36:609–619. https://doi.org/10.1016/S0981-9428(98)80009-0
Falco WF, Scherer MD, Oliveira SL, Wender H, Colbeck I, Lawson T, Caires ARL (2020) Phytotoxicity of silver nanoparticles on Vicia faba: evaluation of particle size effects on photosynthetic performance and leaf gas exchange. Sci Total Environ 701:134816–134871. https://doi.org/10.1016/j.scitotenv.2019.134816
Fancy NN, Bahlmann A, Loake GJ (2017) Nitric oxide function in plant abiotic stress. Plant Cell Environ 40:462–472. https://doi.org/10.1111/pce.12707
Fatma M, Masood A, Per TS, Khan NA (2016) Nitric oxide alleviates salt stress inhibited photosynthetic performance by interacting with sulfur assimilation in mustard. Front Plant Sci 7:521–536. https://doi.org/10.3389/fpls.2016.00521
Fernando DR, Mizuno T, Woodrow IE, Baker AJM, Collins RN (2010) Characterization of foliar manganese (Mn) in Mn (hyper) accumulators using X-ray absorption spectroscopy. New Phytol 188:1014–1027. https://doi.org/10.1111/j.1469-8137.2010.03431.x
Fernando DR, Marshall ALT, Forster PI, Hoebee SE, Siegele RA (2013) Multiple metal accumulation within, a manganese-specific genus. Am J Bot 100:690–700. https://doi.org/10.3732/ajb.1200545
Flexas J, Bota J, Loreto F, Cornic G, Sharkey TD (2004) Diffusive and metabolic limitations to photosynthesis under drought and salinity in C3 plants. Plant Biol 6:269–279. https://doi.org/10.1055/s-2004-820867
Gondor OK, Szalai G, Kovács V, Janda T, Pál M (2014) Impact of UV-B on drought- or cadmium-induced changes in the fatty acid composition of membrane lipid fractions in wheat. Ecotoxicol Environ Saf 108:129–134. https://doi.org/10.1016/j.ecoenv.2014.07.002
Hasanuzzaman M, Nahar K, Rahman A, Inafuku M, Oku H, Fujita M (2018) Exogenous nitric oxide donor and arginine provide protection against short-term drought stress in wheat seedlings. Physiol Mol Biol Plant 24:993–1004. https://doi.org/10.1007/s12298-018-0531-6
Kaur S, Singh HP, Batish DR, Negi A, Mahajan P, Rana S, Kohli RK (2012) As inhibits radicals emergence and elongation in Phaseolus aureus by altering starch-metabolizing enzymes vis-a-vis disruption of oxidative metabolism. Biol Trace Elem Res 146:360–368. https://doi.org/10.1007/s12011-011-9258-8
Küpper H, Andresen E (2016) Mechanisms of metal toxicity in plants. Metallomics 8:269–285. https://doi.org/10.1039/c5mt00244c
Liu Y, Wu R, Wan Q, Xie G, Bi Y (2007) Glucose-6-phosphate dehydrogenase plays a pivotal role in nitric oxide-involved defense against oxidative stress under salt stress in red kidney bean roots. Plant Cell Physiol 48:511–522. https://doi.org/10.1093/pcp/pcm020
Liu B, Lianga J, Tang G, Wang X, Liua F, Zhaoa D (2019) Drought stress effects on growth, water use efficiency, gas exchange and chlorophyll fluorescence of Juglans root stocks. Sci Hort 250:230–235. https://doi.org/10.1016/j.scienta.2019.02.056
Mahjoubi Y, Rzigui T, Ben Massoud M, Kharbech O, Loussaief N, Chaoui A, Djebali W (2020) Leaf gas exchange of bean (Phaseolus vulgaris L.) seedlings subjected to manganese stress. Russ J Plant Physiol 67:168–174. https://doi.org/10.1134/S1021443720010100
Maksymiec W (2007) Signaling responses in plants to heavy metal stress. Acta Physiol Plant 29:177–187. https://doi.org/10.1007/s11738-007-0036-3
Manai J, Gouia H, Corpas FJ (2014) Redox and nitric oxide homeostasis are affected in tomato (Solanum lycopersicum) roots under salinity-induced oxidative stress. J Plant Physiol 171(12):1028–1035. https://doi.org/10.1016/j.jplph.2014.03.012
Marques DM, Júnior VV, Da Silva AB, Mantovani JR, Magalhães PC, De Souza TC (2018) Copper toxicity on photosynthetic responses and root morphology of Hymenaea courbaril L. (Caesalpinioideae). Water Air Soil Pollut 229:138–151. https://doi.org/10.1007/s11270-018-3769-2
Mata-Pérez C, Sánchez-Calvo B, Padilla MN, Begara-Morales JC, Valderrama R, Corpas FJ, Barroso JB (2017) Nitro-fatty acids in plant signaling: new key mediators of nitric oxide metabolism. Redox Biol 11:554–561. https://doi.org/10.1016/j.redox.2017.01.002
Meng F, Cao R, Yang D, Niklas KJ, Sun S (2014) Trade-offs between light interception and leaf water shedding: a comparison of shade- and sun-adapted species in a subtropical rain forest. Oecologia 174:13–22. https://doi.org/10.1007/s00442-013-2746-0
Miah MR, Ijomone OI, Okoh COA, Ijomone KO, Akingbade GT, Ke T, Krum B, Martins AC Jr, Akinyemi A, Aranoff N, Soares FAA, Bowman AB, Aschner M (2020) The effects of manganese overexposure on brain health. Neurochem Int 135:104688. https://doi.org/10.1016/j.neuint.2020.104688
Millaleo R, Reyes-Díaz M, Ivanov AG, Mora ML, Alberdi M (2010) Manganese as essential and toxic element for plants: transport, accumulation and resistance mechanisms. J Soil Sci Plant Nutr 10:476–494. https://doi.org/10.4067/S0718-95162010000200008
Misra HP, Fridovich I (1972) The role of superoxide anion in the autoxidation of epinephrine and a simple assay for superoxide dismutase. J Biol Chem 247:3170–3175. https://doi.org/10.1016/S0021-9258(19)45228-9
Mostofa MG, Seraj ZI, Fujita M (2014) Exogenous sodium nitroprusside and glutathione alleviate copper toxicity by reducing copper uptake and oxidative damage in rice (Oryza sativa L.) seedlings. Protoplasma 251:1373–1386. https://doi.org/10.1007/s00709-014-0639-7
Mostofa MG, Fujita M, Tran LSP (2015) Nitric oxide mediates hydrogen peroxide-and salicylic acid-induced salt tolerance in rice (Oryza sativa L.) seedlings. Plant Growth Regul 77:265–277. https://doi.org/10.1007/s10725-015-0061-y
Nakano Y, Asada K (1981) Hydrogen peroxide is scavenged by ascorbate-specific peroxidase in spinach chloroplasts. Plant Cell Physiol 22:867–880. https://doi.org/10.1093/oxfordjournals.pcp.a076232
Nawaz F, Shabbir RN, Shahbaz M, Majeed S, Raheel M, Hassan W, Sohail MA (2017) Cross talk between nitric oxide and phytohormones regulate plant development during abiotic stresses. In: M El-Esawi (ed) Phytohormones-signaling mechanisms and crosstalk in plant development and stress responses. InTech Publisher, pp 117–141. https://doi.org/10.5772/intechopen.69812
Paul S, Roychoudhury A (2020) Regulation of physiological aspects in plants by hydrogen sulfide and nitric oxide under challenging environment. Physiol Plant 168(2):374–393. https://doi.org/10.1111/ppl.13021
Prakash V, Singh VP, Tripathi DK, Sharma S, Corpas FJ (2019) Cross-talk between nitric oxide (NO) and abscisic acid (ABA) signalling molecules in higher plants. Environ Exp Bot 161:41–49. https://doi.org/10.1016/j.envexpbot.2018.10.033
Rastogi A, Zivcak M, Tripathi BK, Yadav S, Kalaji HM, Brestic M (2019) Phytotoxic effect of silver nanoparticles in Triticum aestivum. improper regulation of photosystem I activity as the reason for oxidative damage in the chloroplast. Photosynthetica 57:209–216. https://doi.org/10.32615/ps.2019.019
Rizwana BSN, Rupesh T, Hussainb A, Kulkarnic KP, Imrana QM, Muna BG, Byung-Wook Yuna BW (2019) Nitric oxide regulates plant responses to drought, salinity, and heavy metal stress. Environ Exp Bot 161:157–165. https://doi.org/10.1016/j.envexpbot.2019.02.003
Saidi I, Nawel N, Djebali W (2014) Role of selenium in preventing manganese toxicity in sunflower (Helianthus annuus) seedling. S Afr J Bot 94:88–94. https://doi.org/10.1016/j.sajb.2014.06.005
Salin ML, Lyon DS (1983) Iron-containing superoxide dismutase in eukaryotes: localization in chloroplasts in water lily, Nuphar luteum. Oxy radicals and their scavenger systems. Elsevier Science Publishing Co., New York, pp 344–347
Sami F, Siddiqui H, Hayat S (2020) Nitric oxide-mediated enhancement in photosynthetic efficiency, ion uptake and carbohydrate metabolism that boosts overall photosynthetic machinery in mustard plants. J Plant Growth Regul. https://doi.org/10.1007/s00344-020-10166-5
Sergiev I, Alexieva V, Karanov E (1997) Effect of spermine, atrazine and combination between them on some endogenous protective systems and stress markers in plants. C R Acad Bulg Sci 51:121–124
Shahzad B, Tanveer M, Rehman A, Cheema SA, Fahad S, Rehman S, Sharma A (2018) Nickel; whether toxic or essential for plants and environment. Plant Physiol Biochem 132:641–651. https://doi.org/10.1016/j.plaphy.2018.10.014
Shrestha A, Dziwornu AK, Ueda Y, Wu LB, Mathew B, Frei M (2018) Genome-wide association study to identify candidate loci and genes for Mn toxicity tolerance in rice. PLoS One 13(2):0192116. https://doi.org/10.1371/journal.pone.0192116
Surrey K (1964) Spectrophotometric method for determination of lipoxidase activity. Plant Physiol 39:65–70. https://doi.org/10.1104/pp.39.1.65
Tremolieres A, Drapier D, DubacqMazliak JP (1980) Oleyl-coenzyme a metabolization by sub-cellular fractions from growing pea leaves. Plant Sci Lett 18:257–269. https://doi.org/10.1016/0304-4211(80)90116-9
Tremolieres A, Siegenthaler PA (1998) Reconstitution of Photosynthetic Structures and Activities with Lipids. In : Siegenthaler PA, Murata N (eds) Lipids in Photosynthesis: Structure, Function and Genetics. Kluwer Academic Publishers, Dordrecht, pp 175–189
Tukaj Z, Baścik-Remisiewicz A, Skowroński T, Tukaj C (2007) Cadmium effect on the growth, photosynthesis, ultrastructure and phytochelatin content of green microalga Scenedesmus armatus: a study at low and elevated CO2 concentration. Environ Exp Bot 60:291–299. https://doi.org/10.1016/j.envexpbot.2006.12.002
Turkan I (2018) ROS and RNS: key signalling molecules in plants. J Exp Bot 69:3313–3315. https://doi.org/10.1093/jxb/ery198
Wallis JG, Browse J (2002) Mutant of Arabidopsis reveals many roles for membrane lipids. Prog Lipid Res 41:254–278. https://doi.org/10.1016/S0163-7827(01)00027-3
Wei L, Zhang M, Wei S, Zhang J, Wang C, Liao W (2020) Roles of nitric oxide in heavy metal stress in plants: cross-talk with phytohormones and protein S-nitrosylation. Environ Pollut 259:113943. https://doi.org/10.1016/j.envpol.2020.113943
Wu XX, Ding HD, Chen JL, Zhang HJ, Zhu WM (2010) Attenuation of salt-induced changes in photosynthesis by exogenous nitric oxide in tomato (Lycopersicon esculentum Mill. L.) seedlings. Afr J Biotechnol 9:7837–7846. https://doi.org/10.5897/AJB10.1187
Xiong J, Fu G, Tao L, Zhu C (2010) Roles of nitric oxide in alleviating heavy metal toxicity in plants. Nitric oxide synthesis and signaling in plants. Arch Biochem Biophys 497:13–20. https://doi.org/10.1016/j.abb.2010.02.014
Acknowledgements
We gratefully acknowledge Abbes Wacharine, Imen Oueslati, and Zouhaier Nasr for their technical help.
Funding
This work was supported by the Tunisian Ministry of Higher Education and Scientific Research (LR18ES38).
Author information
Authors and Affiliations
Contributions
YM performed all laboratory experiments, and wrote the manuscript; TR helped in manuscript drafting and performed photosynthesis measurements; OK statistically analyzed the data; SNM performed CPG analysis; LA analyzed CPG data; AC helped in manuscript revision; IN helped in lipid data revision and WD supervised the experiments and helped in the physiological analyses and revision step. All authors have read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval
Not applicable.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Conflict of interest
The authors declare no competing interests.
Additional information
Handling Editor: Néstor Carrillo
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Mahjoubi, Y., Rzigui, T., Kharbech, O. et al. Exogenous nitric oxide alleviates manganese toxicity in bean plants by modulating photosynthesis in relation to leaf lipid composition. Protoplasma 259, 949–964 (2022). https://doi.org/10.1007/s00709-021-01713-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00709-021-01713-2