Abstract
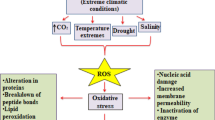
Temperatures above the optimum are sensed as heat stress (HS) by all living organisms and represent one of the major environmental challenges for plants. Plants can cope with HS by activating specific defense mechanisms to minimize damage and ensure cellular functionality. One of the most common effects of HS is the overproduction of reactive oxygen and nitrogen species (ROS and RNS). The role of ROS and RNS in the regulation of many plant physiological processes is well established. On the contrary, in plants very little is known about the physiological role of peroxynitrite (ONOO−), the RNS species generated by the interaction between NO and O2−. In this work, the role of ONOO− on some of the stress responses induced by HS in tobacco BY-2 cultured cells has been investigated by measuring these responses both in the presence and in the absence of 2,6,8-trihydroxypurine (urate), a specific scavenger of ONOO−. The obtained results suggest a potential role for ONOO− in some of the responses induced by HS in tobacco cultured cells. In particular, ONOO− seems implicated in a form of cell death showing apoptotic features and in the regulation of the levels of proteins involved in the response to stress.





Similar content being viewed by others
Abbreviations
- BiP:
-
Binding protein
- DAF-FM:
-
4-Amino-5-methylamino-2′,7′-difluorofluorescein
- HS:
-
Heat stress
- NO:
-
Nitric oxide
- ONOO:
-
Peroxynitrite
- PCD:
-
Programmed cell death
- ROS:
-
Reactive oxygen species
- RNS:
-
Reactive nitrogen species
- XTT:
-
Sodium,39-[1-[phenylamino-carbonyl]-3,4-tetrazolium]-bis(4-methoxy-6-nitro) benzene-sulfonic acid hydrate
References
Able AJ, Guest DI, Sutherland MW (1998) Use of a new tetrazolium-based assay to study the production of the superoxide radicals by tobacco cell cultures challenged with avirulent zoospores of Phytophthora parasitica var nicotianae. Plant Physiol 117(2):491–499. https://doi.org/10.1104/pp.117.2.491
Alamillo JM, García-Olmedo F (2001) Effects of urate, a natural scavenger of peroxynitrite-mediated toxicity, in the response of Arabidopsis thaliana to the bacterial pathogen Pseudomonas syringae. Plant J 25(5):529–540. https://doi.org/10.1046/j.1365-313x.2001.00984.x
Altieri DC, Stein GS, Lian JB, Languino LR (2012) TRAP-1 the mitochondrial Hsp90. Biochim Biophys Acta 1823(3):767–773. https://doi.org/10.1016/j.bbamcr.2011.08.007
Arasimowicz-Jelonek M, Floryszak-Wieczorek J (2011) Understanding the fate of peroxynitrite in plant cells—from physiology to pathophysiology. Phytochemistry 72(8):681–688. https://doi.org/10.1016/j.phytochem.2011.02.025
Arasimowicz-Jelonek M, Floryszak-Wieczorek J, Izbiańska K, Gzyl J, Jelonek T (2016) Implication of peroxynitrite in defence responses of potato to Phytophthora infestans. Plant Pathol 65(5):754–766. https://doi.org/10.1111/ppa.12471
Aslan M, Ryan TM, Townes TM, Coward L, Kirk MC, Barnes S, Alexander CB, Rosenfeld SS, Freeman BA (2003) Nitric oxide dependent generation of reactive species in sickle cell disease. Actin tyrosine nitration induces defective cytoskeletal polymerization. J Biol Chem 78:4194–4204
Baker CJ, Mock NM (1994) An improved method for monitoring cell death in cell suspension and leaf disk assays using Evan’s blue. Plant Cell Tissue Organ Cult 39(1):7–12. https://doi.org/10.1007/BF00037585
Belenghi B, Romero-Puertas MC, Vercammen D, Brackenier A, Inzé D, Delledonne M, van Breusegem F (2007) Metacaspase activity of Arabidopsis thaliana is regulated by S-nitrosylation of a critical cysteine residue. J Biol Chem 282(2):1352–1358. https://doi.org/10.1074/jbc.M608931200
Challinor AJ, Ewert F, Arnold S, Simelton E, Fraser E (2009) Crops and climate change: progress, trends, and challenges in simulating impacts and informing adaptation. J Exp Bot 60(10):2775–2789. https://doi.org/10.1093/jxb/erp062
Dobrá J, Černý M, Štorchová H, Dobrev P, Skalák J, Jedelský PL, Lukšanová H, Gaudinová A, Pešek B, Malbeck J, Vanek T, Brzobohatý B, Vanková R (2015) The impact of heat stress targeting on the hormonal and transcriptomic response in Arabidopsis. Plant Sci 231:52–61. https://doi.org/10.1016/j.plantsci.2014.11.005
Farnese FS, Menezes-Silva PE, Gusman GS, Oliveira JA (2016) When bad guys become good ones: the key role of reactive oxygen species and nitric oxide in the plant responses to abiotic stress. Front Plant Sci 7:471. https://doi.org/10.3389/fpls.2016.00471
Gaupels F, Spiazzi-Vandelle E, Yang D, Delledonne M (2011) Detection of peroxynitrite accumulation in Arabidopsis thaliana during the hypersensitive defense response. Nitric Oxide 25(2):222–228. https://doi.org/10.1016/j.niox.2011.01.009
Gould KS, Lamotte O, Klinguer A, Pugin A, Wendehenne D (2003) Nitric oxide production in tobacco leaf cells: a generalized stress response? Plant Cell Environ 26(11):1851–1862. https://doi.org/10.1046/j.1365-3040.2003.01101.x
Gzyl J, Izbiańska K, Floryszak-Wieczorek J, Jelonek T, Arasimowicz-Jelonek M (2016) Cadmium affects peroxynitrite generation and tyrosine nitration in seedling roots of soybean (Glycine max L.) Environ Exp Bot 131:155–163. https://doi.org/10.1016/j.envexpbot.2016.07.009
Halliwell B (2006) Reactive species and antioxidants. Redox biology is a fundamental theme of aerobic life. Plant Physiol 141(2):312–322. https://doi.org/10.1104/pp.106.077073
Hancock JT (2017) Harnessing evolutionary toxins for signaling: reactive oxygen species, nitric oxide and hydrogen sulfide in plant cell regulation. Front Plant Sci 8:189. https://doi.org/10.3389/fpls.2017.00189
Hasanuzzaman M, Nahar K, Mahabub Alam MD, Roychowdhury R, Fujita M (2013) Physiological, biochemical, and molecular mechanisms of heat stress tolerance in plants. Int J Mol Sci 14(5):9643–9684. https://doi.org/10.3390/ijms14059643
Kadota Y, Shirazu K (2012) The HSP90 complex of plants. Biochim Biophys Acta 1823(3):689–697. https://doi.org/10.1016/j.bbamcr.2011.09.016
Kampinga HH, Brunsting JF, Stege GJ, Burgman PW, Konings AW (1995) Thermal protein denaturation and protein aggregation in cells made thermotolerant by various chemicals: role of heat shock proteins. Exp Cell Res 219(2):536–546. https://doi.org/10.1006/excr.1995.1262
Kang BH, Plescia J, Dohi T, Rosa J, Doxsey SJ, Altieri DC (2007) Regulation of tumor cell mitochondrial homeostasis by an organelle-specific Hsp90 chaperone network. Cell 131(2):257–270. https://doi.org/10.1016/j.cell.2007.08.028
Kotak S, Larkindale J, Lee U, von Koskull-Döring P, Vierling E, Scharf K-D (2007) Complexity of the heat stress response in plants. Curr Opin Plant Biol 10(3):310–316. https://doi.org/10.1016/j.pbi.2007.04.011
Laemmli UK (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227(5259):680–685. https://doi.org/10.1038/227680a0
Lam E (2004) Controlled cell death, plant survival and development. Nat Rev Mol Cell Biol 5(4):305–315. https://doi.org/10.1038/nrm1358
Leshem YY, Wilisr BH, Ku VV (1998) Evidence for the function of the free radical gas nitric oxide as an endogenous maturation and senescence regulating factor in higher plant. Plant Physiol Biochem 36(11):825–833. https://doi.org/10.1016/S0981-9428(99)80020-5
Locato V, Gadaleta C, De Gara L, De Pinto MC (2008) Production of reactive species and modulation of antioxidant network in response to heat shock: a critical balance for cell fate. Plant Cell Environ 31(11):1606–1619. https://doi.org/10.1111/j.1365-3040.2008.01867.x
Lozano-Durán R, Robatzek S (2015) 14-3-3 proteins in plant-pathogen interactions. Mol Plant-Microbe Interact 28(5):511–518. https://doi.org/10.1094/MPMI-10-14-0322-CR
Malerba M, Cerana R (2015) Reactive oxygen and nitrogen species in defense/stress responses activated by chitosan in sycamore cultured cells. Int J Mol Sci 16(2):3019–3034. https://doi.org/10.3390/ijms16023019
Malerba M, Contran N, Tonelli M, Crosti P, Cerana R (2008) Role of nitric oxide in actin depolymerization and programmed cell death induced by fusicoccin in sycamore (Acer pseudoplatanus L.) cultured cells. Physiol Plant 133(2):449–457. https://doi.org/10.1111/j.1399-3054.2008.01085.x
Malerba M, Crosti P, Cerana R (2010) Effect of heat stress on actin cytoskeleton and endoplasmic reticulum of tobacco BY-2 cultured cells and its inhibition by Co2+. Protoplasma 239(1-4):23–30. https://doi.org/10.1007/s00709-009-0078-z
Malerba M, Crosti P, Cerana R (2012) Defense/stress responses activated by chitosan in sycamore cultured cells. Protoplasma 249(1):89–98. https://doi.org/10.1007/s00709-011-0264-7
Marsoni M, Cantara C, De Pinto MC, Gadatela C, De Gara L, Bracale M, Vannini C (2010) Exploring the soluble proteome of tobacco bright Yellow-2 cells at the switch towards different cell fates in response to heat shocks. Plant Cell Environ 33(7):1161–1175. https://doi.org/10.1111/j.1365-3040.2010.02137.x
Mc Cabe PF, Leaver CJ (2000) Programmed cell death in cell cultures. Plant Mol Biol 44(3):359–368. https://doi.org/10.1023/A:1026500810877
Nagata T, Nemoto Y, Hasezawa S (1992) Tobacco BY-2 cell line as the “HeLa” cell in the cell biology of higher plants. Int Rev Cytol 132:1–30. https://doi.org/10.1016/S0074-7696(08)62452-3
Niles JC, Wishnok JS, Tannenbau SR (2006) Peroxynitrite-induced oxidation and nitration products of guanine 8-oxoguanine: structures and mechanisms of product formation. Nitric Oxide 14(2):109–121. https://doi.org/10.1016/j.niox.2005.11.001
Ohama N, Sato H, Shinozaki K, Yamaguchi-Shinozaki K (2017) Transcriptional regulatory network of plant heat stress response. Trends Plant Sci 22(1):53–65. https://doi.org/10.1016/j.tplants.2016.08.015
Parankusam S, Adimulam SS, Bhatnagar-Mathur P, Sharma KK (2017) Nitric oxide (NO) in plant heat stress tolerance: current knowledge and perspectives. Front Plant Sci 8:1582. https://doi.org/10.3389/fpls.2017.01582
Piszczek E, Gutman W (2007) Caspase-like proteases and their role in programmed cell death in plants. Acta Physiol Plant 29(5):391–398. https://doi.org/10.1007/s11738-007-0086-6
Saito S, Yamamoto-Katou A, Yoshioka H, Doke N, Kawakita K (2006) Peroxynitrite generation and tyrosine nitration in defense responses in tobacco BY-2 cells. Plant Cell Physiol 47(6):689–697. https://doi.org/10.1093/pcp/pcj038
Sakamoto A, Sakurao S, Fukunaga K, Matsubara T, Ueda-Hashimoto M, Tsukamoto S, Takahashi M, Morikawa H (2004) Three distinct Arabidopsis hemoglobins exhibit peroxidase-like activity and differentially mediate nitrite-dependent protein nitration. FEBS Lett 572(1-3):27–32. https://doi.org/10.1016/j.febslet.2004.07.005
Sandoval M, Zhang XJ, Liu X, Mannick EE, Clark DA, Miller MJS (1997) Peroxynitrite-induced apoptosis in T84 and raw 264.7 cells: attenuation by L-ascorbic acid. Free Radic Biol Med 22(3):489–495. https://doi.org/10.1016/S0891-5849(96)00374-7
Serrano I, Romero-Puertas MC, Rodríguez Serrano M, Sandalio LM, Olmedilla A (2012) Role of peroxynitrite in programmed cell death induced in self-incompatible pollen. Plant Signal Behav 7(7):779–781. https://doi.org/10.4161/psb.20570
Sun ZN, Wang HL, Liu FQ, Chen Y, Tam PKH, Yang D (2009) BODIPY-based fluorescent probe for peroxynitrite detection and imaging in living cells. Org Lett 11:1187–1190
Suzuky N, Mittler R (2006) Reactive oxygen species and temperature stresses: a delicate balance between signaling and destruction. Physiol Plant 126(1):45–51. https://doi.org/10.1111/j.0031-9317.2005.00582.x
Urade R (2007) Cellular response to unfolded proteins in the endoplasmic reticulum of plants. FEBS J 274(5):1152–1171. https://doi.org/10.1111/j.1742-4658.2007.05664.x
Vacca RA, de Pinto MC, Valenti D, Passarella S, Marra E, De Gara L (2004) Production of reactive oxygen species, alteration of cytosolic ascorbate peroxidase, and impairment of mitochondrial metabolism are early events in heat shock-induced programmed cell death in tobacco bright-yellow 2 cells. Plant Physiol 134(3):1100–1112. https://doi.org/10.1104/pp.103.035956
Vandelle E, Delledonne M (2011) Peroxynitrite formation and function in plants. Plant Sci 181(5):534–539. https://doi.org/10.1016/j.plantsci.2011.05.002
Wang W, Vinocur B, Shoseyov O, Altman A (2004) Role of plant heat-shock proteins and molecular chaperones in the abiotic stress response. Trends Plant Sci 9(5):244–252. https://doi.org/10.1016/j.tplants.2004.03.006
Yang X, Srivastava R, Stephen H, Howell SH, Bassham DC (2016) Activation of autophagy by unfolded proteins during endoplasmic reticulum stress. Plant J 85(1):83–95. https://doi.org/10.1111/tpj.13091
Yu M, Lamattina L, Spoel SH, Loake GJ (2014) Nitric oxide function in plant biology: a redox cue in deconvolution. New Phytol 202(4):1142–1156. https://doi.org/10.1111/nph.12739
Acknowledgments
The authors acknowledge the University of Milano-Bicocca, Fondo d’Ateneo per la Ricerca, for the financial support.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Handling Editor: Peter Nick
Rights and permissions
About this article
Cite this article
Malerba, M., Cerana, R. Role of peroxynitrite in the responses induced by heat stress in tobacco BY-2 cultured cells. Protoplasma 255, 1079–1087 (2018). https://doi.org/10.1007/s00709-017-1200-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00709-017-1200-2