Abstract
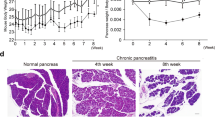
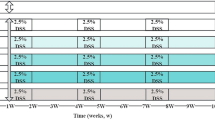
Clonorchis sinensis (C. sinensis) infection is a risk factor for cholangiocarcinoma. Whether it also contributes to the development of hepatocellular carcinoma (HCC) is still unclear. This study explored the potential relationship between C. sinensis infection and HCC. A total of 110 Sprague–Dawley rats were divided into four treatment groups, the negative control group (NC) received intragastric (i.g.) administration of saline, while the clonorchiasis group (CS) received i.g. administration of 150 C. sinensis metacercariae. The diethylnitrosamine-induced group (DEN) received intraperitoneal (i.p.) administration of DEN. The clonorchiasis DEN-induced group (CSDEN) received i.g. administration of 150 C. sinensis metacercariae followed by i.p. administration of DEN. Hematoxylin and eosin staining, immunohistochemistry, and Masson’s trichrome staining were performed for histopathological analysis of the isolated tissues. RNA-seq technology and RT-PCR were employed for gene expression. In the DEN group, 15 rats survived, of which 9 developed liver cirrhosis and 7 developed HCC. In the CSDEN group, all of the 17 surviving rats developed cirrhosis, and 15 showed development of HCC. The incidence of liver cirrhosis and HCC was significantly higher in the CSDEN group than in the DEN group. KEGG pathway analysis of the differentially expressed genes suggested significant upregulation in inflammation-associated pathways. Immunohistochemistry and RT-PCR results showed significant upregulation of hepatic progenitor cell markers (CK19, SOX9, EpCAM) in the CS group compared to the NC group, as well as in the CSDEN group compared to the DEN group. Our study suggests that C. sinensis infection increases risk of HCC in a rat model by stimulating proliferation of hepatic progenitor cells.











Similar content being viewed by others
References
Bahk YY, Pak JH (2016) Toll-like receptor-mediated free radical generation in Clonorchissinensis excretory-secretory product-treated cholangiocarcinoma cells. The Korean J Parasitol 54(5):679–684. https://doi.org/10.3347/kjp.2016.54.5.679
Bouvard V, Baan R, Straif K, Grosse Y, Secretan B, El Ghissassi F, Benbrahim-Tallaa L, Guha N, Freeman C, Galichet L, Cogliano V, WHO International Agency for Research on Cancer Monograph Working Group (2009) A review of human carcinogens–Part B: biological agents. The Lancet Oncol 10(4):321–322. https://doi.org/10.1016/s1470-2045(09)70096-8
Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A (2018) Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA: A Cancer J Clin 68(6):394–424. https://doi.org/10.3322/caac.21492
Di Giosia P, Stamerra CA, Giorgini P, Jamialahamdi T, Butler AE, Sahebkar A (2022) The role of nutrition in inflammaging. Ageing Res Rev 77:101596. https://doi.org/10.1016/j.arr.2022.101596
Diakos CI, Charles KA, McMillan DC, Clarke SJ (2014) Cancer-related inflammation and treatment effectiveness. The Lancet Oncol 15(11):e493–e503. https://doi.org/10.1016/S1470-2045(14)70263-3
Komuta M, Spee B, Vander Borght S, De Vos R, Verslype C, Aerts R, Yano H, Suzuki T, Matsuda M, Fujii H, Desmet VJ, Kojiro M, Roskams T (2008) Clinicopathological study on cholangiolocellular carcinoma suggesting hepatic progenitor cell origin. Hepatol (Baltimore Md) 47(5):1544–1556. https://doi.org/10.1002/hep.22238
Li XF, Chen C, Xiang DM, Qu L, Sun W, Lu XY, Zhou TF, Chen SZ, Ning BF, Cheng Z, Xia MY, Shen WF, Yang W, Wen W, Lee T, Cong WM, Wang HY, Ding J (2017) Chronic inflammation-elicited liver progenitor cell conversion to liver cancer stem cell with clinical significance. Hepatol (Baltimore Md) 66(6):1934–1951. https://doi.org/10.1002/hep.29372
Llovet JM, Castet F, Heikenwalder M, Maini MK, Mazzaferro V, Pinato DJ, Pikarsky E, Zhu AX, Finn RS (2022) Immunotherapies for hepatocellular carcinoma. Nat Rev Clin Oncol 19(3):151–172. https://doi.org/10.1038/s41571-021-00573-2
Moreira AJ, Rodrigues GR, Bona S, Fratta LX, Weber GR, Picada JN, Dos Santos JL, Cerski CT, Marroni CA, Marroni NP (2017) Ductular reaction, cytokeratin 7 positivity, and gamma-glutamyl transferase in multistage hepatocarcinogenesis in rats. Protoplasma 254(2):911–920. https://doi.org/10.1007/s00709-016-1000-0
Na BK, Pak JH, Hong SJ (2020) Clonorchis sinensis and clonorchiasis. Acta Trop 203:105309. https://doi.org/10.1016/j.actatropica.2019.105309
Prueksapanich P, Piyachaturawat P, Aumpansub P, Ridtitid W, Chaiteerakij R, Rerknimitr R (2018) Liver fluke-associated biliary tract cancer. Gut and Liver 12(3):236–245. https://doi.org/10.5009/gnl17102
Qian MB, Utzinger J, Keiser J, Zhou XN (2016) Clonorchiasis. Lancet (london, England) 387(10020):800–810. https://doi.org/10.1016/S0140-6736(15)60313-0
Rizvi S, Fischbach SR, Bronk SF, Hirsova P, Krishnan A, Dhanasekaran R, Smadbeck JB, Smoot RL, Vasmatzis G, Gores GJ (2017) YAP-associated chromosomal instability and cholangiocarcinoma in mice. Oncotarget 9(5):5892–5905. https://doi.org/10.18632/oncotarget.23638
Sia D, Villanueva A, Friedman SL, Llovet JM (2017) Liver cancer cell of origin, molecular class, and effects on patient prognosis. Gastroenterology 152(4):745–761. https://doi.org/10.1053/j.gastro.2016.11.048
Songserm N, Prasongwattana J, Sithithaworn P, Sripa B, Pipitkool V (2009) Cholangiocarcinoma in experimental hamsters with long-standing Opisthorchis viverrini infection. Asian Pacific J Cancer Prev : APJCP 10(2):299–302
TanTan SK, Qiu XQ, Yu HP, Zeng XY, Zhao YN, Hu L (2008) Evaluation of the risk of clonorchiasis inducing primary hepatocellular carcinoma. Zhonghua Gan Zang Bing Za Zhi 16(2):114–116 (Chinese)
Thamavit W, Ngamying M, Boonpucknavig V, Boonpucknavig S, Moore MA (1987) Enhancement of DEN-induced hepatocellular nodule development by Opisthorchis viverrini infection in Syrian golden hamsters. Carcinogenesis 8(9):1351–1353. https://doi.org/10.1093/carcin/8.9.1351
Thamavit W, Moore MA, Hiasa Y, Ito N (1988) Enhancement of DHPN induced hepatocellular, cholangiocellular and pancreatic carcinogenesis by Opisthorchis viverrini infestation in Syrian golden hamsters. Carcinogenesis 9(6):1095–1098. https://doi.org/10.1093/carcin/9.6.1095
Villanueva A (2019) Hepatocellular carcinoma. N Engl J Med 380(15):1450–1462. https://doi.org/10.1056/NEJMra1713263
Williams MJ, Clouston AD, Forbes SJ (2014) Links between hepatic fibrosis, ductular reaction, and progenitor cell expansion. Gastroenterology 146(2):349–356. https://doi.org/10.1053/j.gastro.2013.11.034
Xia J, Jiang SC, Peng HJ (2015) Association between liver fluke infection and hepatobiliary pathological changes: a systematic review and meta-analysis. PLoS ONE 10(7):e0132673. https://doi.org/10.1371/journal.pone.0132673
Xie X, Wang CY, Cao YX, Wang W, Zhuang R, Chen LH, Dang NN, Fang L, Jin BQ (2005) Expression pattern of epithelial cell adhesion molecule on normal and malignant colon tissues. World J Gastroenterol 11(3):344–347. https://doi.org/10.3748/wjg.v11.i3.34
Yan C, Li B, Fan F, Du Y, Ma R, Cheng XD, Li XY, Zhang B, Yu Q, Wang YG, Tang RX, Zheng KY (2017) The roles of Toll-like receptor 4 in the pathogenesis of pathogen-associated biliary fibrosis caused by Clonorchis sinensis. Sci Rep 7(1):3909. https://doi.org/10.1038/s41598-017-04018-8
Yip TC, Wong VW, Chan HL, Tse YK, Lui GC, Wong GL (2020) Tenofovir is associated with lower risk of hepatocellular carcinoma than entecavir in patients with chronic HBV INFection in China. Gastroenterology 158(1):215-225.e6. https://doi.org/10.1053/j.gastro.2019.09.025
Zhao Q, Yu WL, Lu XY, Dong H, Gu YJ, Sheng X, Cong WM, Wu MC (2016) Combined hepatocellular and cholangiocarcinoma originating from the same clone: a pathomolecular evidence-based study. Chin J Cancer 35(1):82. https://doi.org/10.1186/s40880-016-0146-7
Zhao H, Wu L, Yan G, Chen Y, Zhou M, Wu Y, Li Y (2021) Inflammation and tumor progression: signaling pathways and targeted intervention. Signal Transduct Target Ther 6(1):263. https://doi.org/10.1038/s41392-021-00658-5
Zheng S, Zhu Y, Zhao Z, Wu Z, Okanurak K, Lv Z (2017) Liver fluke infection and cholangiocarcinoma: a review. Parasitol Res 116(1):11–19. https://doi.org/10.1007/s00436-016-5276-y
Funding
This work was supported by grants from the National Natural Science Foundation of China (81960450), National Major Special Science and Technology Project (2017ZX10203207), High-level Innovation Team and Outstanding Scholar Program of Guangxi Colleges and Universities,“139” Projects for Training of High-level Medical Science Talents from Guangxi, Key Research and Development Project of Guangxi (AA18221001, AB18050020, 2020AB34006), Key Laboratory of Early Prevention and Treatment for Regional High Frequency Tumors, Ministry of Education of Guangxi, Independent Research Projects (GKE2017-ZZ02, GKE2018-KF02, GKE2019-ZZ07), and Development and Application of Medical and Health-appropriate Technology in Guangxi (S2019039).
Author information
Authors and Affiliations
Contributions
Bangde Xiang: obtained funding; conceived and designed the study, Yapeng Qi: analyzed the data and drafted the first version of the manuscript. Junwen Hu, Jiahao Liang Xiaoyin Hu: acquisition of data; analysis and interpretation of data. Ning Ma: material support; critical revision of the manuscript for important intellectual content; study supervision. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Section Editor: David Bruce Conn
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Qi, Y., Hu, J., Liang, J. et al. Clonorchis sinensis infection contributes to hepatocellular carcinoma progression in rat. Parasitol Res 121, 3403–3415 (2022). https://doi.org/10.1007/s00436-022-07699-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00436-022-07699-x