Abstract
Purpose
Evaluation of tumor budding in colorectal cancer (CRC) may help to predict the tumors’ metastatic potential and patients with an aggressive tumor, although not yet metastasized at time of surgery might benefit from adjuvant therapy.
Methods
The degree of intratumoral tumor budding (ITB) was classified as low, intermediate, and high grade according to the recommendations of the International Tumor Budding Consensus Conference (ITBCC) 2016 on H&E and pankeratin-stained TMA sections from 1262 CRC, no special type (NST), including 655 stage II CRC and was correlated to clinicopathological data and overall survival.
Results
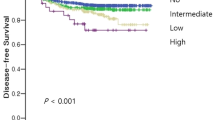
Results show that higher ITB rates are significantly linked to higher tumor grade and stage, positive nodal status, lymphovascular invasion (P < 0.0001 each), absence of peritumoral lymphocytes, infiltrating type invasive tumor margin, left-sided cancer localization, and mismatch-repair proficient cancers (P < 0.05 each). In a cohort of 655 stage II CRC, ITB was associated with lymphovascular invasion (P = 0.0459) and adverse clinical outcome (P < 0.0001). In a multivariate analysis including tumor stage, tumor grade, lymphovascular invasion, ITB, and tumor localization, only low tumor stage (P = 0.0022) and absence of lymphovascular invasion (P = 0.0043) showed independent prognostic significance.
Conclusion
In conclusion, our findings argue towards a clinical utility of ITB as a prognostic biomarker in stage II colorectal cancer to define patients who might benefit from adjuvant therapy. ITB might be used as additional or surrogate marker in CRC in which peritumoral tumor budding is difficult to assess.



Similar content being viewed by others
References
Parkin DM, Bray F, Ferlay J, Pisani P (2005) Global cancer statistics, 2002. CA Cancer J Clin 55(2):74–108
Bilchik AJ, Nora DT, Saha S, Turner R, Wiese D, Kuo C, Ye X, Morton DL, Hoon DS (2002) The use of molecular profiling of early colorectal cancer to predict micrometastases. Arch Surg 137(12):1377–1383
De Smedt L, Palmans S, Sagaert X (2016) Tumour budding in colorectal cancer: what do we know and what can we do? Virchows Arch 468(4):397–408. https://doi.org/10.1007/s00428-015-1886-5
Lugli A, Karamitopoulou E, Zlobec I (2012) Tumour budding: a promising parameter in colorectal cancer. Br J Cancer 106(11):1713–1717. https://doi.org/10.1038/bjc.2012.127
Mitrovic B, Schaeffer DF, Riddell RH, Kirsch R (2012) Tumor budding in colorectal carcinoma: time to take notice. Mod Pathol 25(10):1315–1325. https://doi.org/10.1038/modpathol.2012.94
Rogers AC, Winter DC, Heeney A, Gibbons D, Lugli A, Puppa G, Sheahan K (2016) Systematic review and meta-analysis of the impact of tumour budding in colorectal cancer. Br J Cancer 115(7):831–840. https://doi.org/10.1038/bjc.2016.274
van Wyk HC, Park J, Roxburgh C, Horgan P, Foulis A, McMillan DC (2015) The role of tumour budding in predicting survival in patients with primary operable colorectal cancer: a systematic review. Cancer Treat Rev 41(2):151–159. https://doi.org/10.1016/j.ctrv.2014.12.007
Ueno H, Murphy J, Jass JR, Mochizuki H, Talbot IC (2002) Tumour 'budding' as an index to estimate the potential of aggressiveness in rectal cancer. Histopathology 40(2):127–132
Lugli A, Vlajnic T, Giger O, Karamitopoulou E, Patsouris ES, Peros G, Terracciano LM, Zlobec I (2011) Intratumoral budding as a potential parameter of tumor progression in mismatch repair-proficient and mismatch repair-deficient colorectal cancer patients. Hum Pathol 42(12):1833–1840. https://doi.org/10.1016/j.humpath.2011.02.010
Jass JR, Barker M, Fraser L, Walsh MD, Whitehall VL, Gabrielli B, Young J, Leggett BA (2003) APC mutation and tumour budding in colorectal cancer. J Clin Pathol 56(1):69–73
Dawson H, Koelzer VH, Karamitopoulou E, Economou M, Hammer C, Muller DE, Lugli A, Zlobec I (2014) The apoptotic and proliferation rate of tumour budding cells in colorectal cancer outlines a heterogeneous population of cells with various impacts on clinical outcome. Histopathology 64(4):577–584. https://doi.org/10.1111/his.12294
Prall F, Ostwald C, Weirich V, Nizze H (2006) p16(INK4a) promoter methylation and 9p21 allelic loss in colorectal carcinomas: relation with immunohistochemical p16(INK4a) expression and with tumor budding. Hum Pathol 37(5):578–585. https://doi.org/10.1016/j.humpath.2006.01.005
Zlobec I, Lugli A (2010) Epithelial mesenchymal transition and tumor budding in aggressive colorectal cancer: tumor budding as oncotarget. Oncotarget 1(7):651–661. https://doi.org/10.18632/oncotarget.199
Hostettler L, Zlobec I, Terracciano L, Lugli A (2010) ABCG5-positivity in tumor buds is an indicator of poor prognosis in node-negative colorectal cancer patients. World J Gastroenterol 16(6):732–739
Koelzer VH, Zlobec I, Lugli A (2016) Tumor budding in colorectal cancer--ready for diagnostic practice? Hum Pathol 47(1):4–19. https://doi.org/10.1016/j.humpath.2015.08.007
Wang LM, Kevans D, Mulcahy H, O'Sullivan J, Fennelly D, Hyland J, O'Donoghue D, Sheahan K (2009) Tumor budding is a strong and reproducible prognostic marker in T3N0 colorectal cancer. Am J Surg Pathol 33(1):134–141. https://doi.org/10.1097/PAS.0b013e318184cd55
Hammond ME, Hayes DF, Dowsett M, Allred DC, Hagerty KL, Badve S, Fitzgibbons PL, Francis G, Goldstein NS, Hayes M, Hicks DG, Lester S, Love R, Mangu PB, McShane L, Miller K, Osborne CK, Paik S, Perlmutter J, Rhodes A, Sasano H, Schwartz JN, Sweep FC, Taube S, Torlakovic EE, Valenstein P, Viale G, Visscher D, Wheeler T, Williams RB, Wittliff JL, Wolff AC (2010) American Society of Clinical Oncology/college of American pathologists guideline recommendations for immunohistochemical testing of estrogen and progesterone receptors in breast cancer. J Clin Oncol : official journal of the American Society of Clinical Oncology 28(16):2784–2795
Humphrey PA (2004) Gleason grading and prognostic factors in carcinoma of the prostate. Mod Pathol 17(3):292–306. https://doi.org/10.1038/modpathol.3800054
Lugli A, Kirsch R, Ajioka Y, Bosman F, Cathomas G, Dawson H, El Zimaity H, Flejou JF, Hansen TP, Hartmann A, Kakar S, Langner C, Nagtegaal I, Puppa G, Riddell R, Ristimaki A, Sheahan K, Smyrk T, Sugihara K, Terris B, Ueno H, Vieth M, Zlobec I, Quirke P (2017) Recommendations for reporting tumor budding in colorectal cancer based on the international tumor budding consensus conference (ITBCC) 2016. Mod Pathol 30(9):1299–1311. https://doi.org/10.1038/modpathol.2017.46
Simon R, Mirlacher M, Sauter G (2004) Tissue microarrays. Biotechniques 36(1):98–105
Boxberg M, Kuhn PH, Reiser M, Erb A, Steiger K, Pickhard A, Strassen U, Koob I, Kolk A, Warth A, Jesinghaus M, Weichert W (2019) Tumor budding and cell Nest size are highly prognostic in laryngeal and Hypopharyngeal squamous cell carcinoma: further evidence for a unified Histopathologic grading system for squamous cell carcinomas of the upper Aerodigestive tract. Am J Surg Pathol 43(3):303–313. https://doi.org/10.1097/PAS.0000000000001178
Karamitopoulou E, Zlobec I, Born D, Kondi-Pafiti A, Lykoudis P, Mellou A, Gennatas K, Gloor B, Lugli A (2013) Tumour budding is a strong and independent prognostic factor in pancreatic cancer. Eur J Cancer 49(5):1032–1039. https://doi.org/10.1016/j.ejca.2012.10.022
Koelzer VH, Langer R, Zlobec I, Lugli A (2014) Tumor budding in upper gastrointestinal carcinomas. Front Oncol 4:216. https://doi.org/10.3389/fonc.2014.00216
Bosch SL, Teerenstra S, de Wilt JH, Cunningham C, Nagtegaal ID (2013) Predicting lymph node metastasis in pT1 colorectal cancer: a systematic review of risk factors providing rationale for therapy decisions. Endoscopy 45(10):827–834. https://doi.org/10.1055/s-0033-1344238
Ueno H, Mochizuki H, Hashiguchi Y, Shimazaki H, Aida S, Hase K, Matsukuma S, Kanai T, Kurihara H, Ozawa K, Yoshimura K, Bekku S (2004) Risk factors for an adverse outcome in early invasive colorectal carcinoma. Gastroenterology 127(2):385–394. https://doi.org/10.1053/j.gastro.2004.04.022
Beaton C, Twine CP, Williams GL, Radcliffe AG (2013) Systematic review and meta-analysis of histopathological factors influencing the risk of lymph node metastasis in early colorectal cancer. Color Dis 15(7):788–797. https://doi.org/10.1111/codi.12129
Kawachi H, Eishi Y, Ueno H, Nemoto T, Fujimori T, Iwashita A, Ajioka Y, Ochiai A, Ishiguro S, Shimoda T, Mochizuki H, Kato Y, Watanabe H, Koike M, Sugihara K (2015) A three-tier classification system based on the depth of submucosal invasion and budding/sprouting can improve the treatment strategy for T1 colorectal cancer: a retrospective multicenter study. Mod Pathol 28(6):872–879. https://doi.org/10.1038/modpathol.2015.36
Kajiwara Y, Ueno H, Hashiguchi Y, Mochizuki H, Hase K (2010) Risk factors of nodal involvement in T2 colorectal cancer. Dis Colon Rectum 53(10):1393–1399. https://doi.org/10.1007/DCR.0b013e3181ec5f66
Kitajima K, Fujimori T, Fujii S, Takeda J, Ohkura Y, Kawamata H, Kumamoto T, Ishiguro S, Kato Y, Shimoda T, Iwashita A, Ajioka Y, Watanabe H, Watanabe T, Muto T, Nagasako K (2004) Correlations between lymph node metastasis and depth of submucosal invasion in submucosal invasive colorectal carcinoma: a Japanese collaborative study. J Gastroenterol 39(6):534–543. https://doi.org/10.1007/s00535-004-1339-4
Kaneko I, Tanaka S, Oka S, Yoshida S, Hiyama T, Arihiro K, Shimamoto F, Chayama K (2007) Immunohistochemical molecular markers as predictors of curability of endoscopically resected submucosal colorectal cancer. World J Gastroenterol 13(28):3829–3835. https://doi.org/10.3748/wjg.v13.i28.3829
Tateishi Y, Nakanishi Y, Taniguchi H, Shimoda T, Umemura S (2010) Pathological prognostic factors predicting lymph node metastasis in submucosal invasive (T1) colorectal carcinoma. Mod Pathol 23(8):1068–1072. https://doi.org/10.1038/modpathol.2010.88
Ueno H, Hashiguchi Y, Kajiwara Y, Shinto E, Shimazaki H, Kurihara H, Mochizuki H, Hase K (2010) Proposed objective criteria for "grade 3" in early invasive colorectal cancer. Am J Clin Pathol 134(2):312–322. https://doi.org/10.1309/AJCPMQ7I5ZTTZSOM
Keum MA, Lim SB, Kim SA, Yoon YS, Kim CW, Yu CS, Kim JC (2012) Clinicopathologic factors affecting recurrence after curative surgery for stage I colorectal cancer. J Korean Soc Coloproctol 28(1):49–55. https://doi.org/10.3393/jksc.2012.28.1.49
Nakadoi K, Tanaka S, Kanao H, Terasaki M, Takata S, Oka S, Yoshida S, Arihiro K, Chayama K (2012) Management of T1 colorectal carcinoma with special reference to criteria for curative endoscopic resection. J Gastroenterol Hepatol 27(6):1057–1062. https://doi.org/10.1111/j.1440-1746.2011.07041.x
Suh JH, Han KS, Kim BC, Hong CW, Sohn DK, Chang HJ, Kim MJ, Park SC, Park JW, Choi HS, Oh JH (2012) Predictors for lymph node metastasis in T1 colorectal cancer. Endoscopy 44(6):590–595. https://doi.org/10.1055/s-0031-1291665
Wada H, Shiozawa M, Katayama K, Okamoto N, Miyagi Y, Rino Y, Masuda M, Akaike M (2015) Systematic review and meta-analysis of histopathological predictive factors for lymph node metastasis in T1 colorectal cancer. J Gastroenterol 50(7):727–734. https://doi.org/10.1007/s00535-015-1057-0
Ueno H, Hase K, Hashiguchi Y, Shimazaki H, Yoshii S, Kudo SE, Tanaka M, Akagi Y, Suto T, Nagata S, Matsuda K, Komori K, Yoshimatsu K, Tomita Y, Yokoyama S, Shinto E, Nakamura T, Sugihara K (2014) Novel risk factors for lymph node metastasis in early invasive colorectal cancer: a multi-institution pathology review. J Gastroenterol 49(9):1314–1323. https://doi.org/10.1007/s00535-013-0881-3
Betge J, Kornprat P, Pollheimer MJ, Lindtner RA, Schlemmer A, Rehak P, Vieth M, Langner C (2012) Tumor budding is an independent predictor of outcome in AJCC/UICC stage II colorectal cancer. Ann Surg Oncol 19(12):3706–3712. https://doi.org/10.1245/s10434-012-2426-z
Petrelli F, Pezzica E, Cabiddu M, Coinu A, Borgonovo K, Ghilardi M, Lonati V, Corti D, Barni S (2015) Tumour budding and survival in stage II colorectal Cancer: a systematic review and pooled analysis. J Gastrointest Cancer 46(3):212–218. https://doi.org/10.1007/s12029-015-9716-1
Lai YH, Wu LC, Li PS, Wu WH, Yang SB, Xia P, He XX, Xiao LB (2014) Tumour budding is a reproducible index for risk stratification of patients with stage II colon cancer. Color Dis 16(4):259–264. https://doi.org/10.1111/codi.12454
Nakamura T, Mitomi H, Kanazawa H, Ohkura Y, Watanabe M (2008) Tumor budding as an index to identify high-risk patients with stage II colon cancer. Dis Colon Rectum 51(5):568–572. https://doi.org/10.1007/s10350-008-9192-9
Hayes BD, Maguire A, Conlon N, Gibbons D, Wang LM, Sheahan K (2010) Reproducibility of the rapid bud count method for assessment of tumor budding in stage II colorectal cancer. Am J Surg Pathol 34(5):746–748. https://doi.org/10.1097/PAS.0b013e3181da76b6
Graham RP, Vierkant RA, Tillmans LS, Wang AH, Laird PW, Weisenberger DJ, Lynch CF, French AJ, Slager SL, Raissian Y, Garcia JJ, Kerr SE, Lee HE, Thibodeau SN, Cerhan JR, Limburg PJ, Smyrk TC (2015) Tumor budding in colorectal carcinoma: confirmation of prognostic significance and histologic cutoff in a population-based cohort. Am J Surg Pathol 39(10):1340–1346. https://doi.org/10.1097/PAS.0000000000000504
Horcic M, Koelzer VH, Karamitopoulou E, Terracciano L, Puppa G, Zlobec I, Lugli A (2013) Tumor budding score based on 10 high-power fields is a promising basis for a standardized prognostic scoring system in stage II colorectal cancer. Hum Pathol 44(5):697–705. https://doi.org/10.1016/j.humpath.2012.07.026
Kinoshita H, Watanabe T, Yanagisawa A, Nagawa H, Kato Y, Muto T (2004) Pathological changes of advanced lower-rectal cancer by preoperative radiotherapy. Hepatogastroenterology 51(59):1362–1366
Huebner M, Wolff BG, Smyrk TC, Aakre J, Larson DW (2012) Partial pathologic response and nodal status as most significant prognostic factors for advanced rectal cancer treated with preoperative chemoradiotherapy. World J Surg 36(3):675–683. https://doi.org/10.1007/s00268-011-1409-8
Du C, Xue W, Li J, Cai Y, Gu J (2012) Morphology and prognostic value of tumor budding in rectal cancer after neoadjuvant radiotherapy. Hum Pathol 43(7):1061–1067. https://doi.org/10.1016/j.humpath.2011.07.026
Sannier A, Lefevre JH, Panis Y, Cazals-Hatem D, Bedossa P, Guedj N (2014) Pathological prognostic factors in locally advanced rectal carcinoma after neoadjuvant radiochemotherapy: analysis of 113 cases. Histopathology 65(5):623–630. https://doi.org/10.1111/his.12432
Mou S, Soetikno R, Shimoda T, Rouse R, Kaltenbach T (2013) Pathologic predictive factors for lymph node metastasis in submucosal invasive (T1) colorectal cancer: a systematic review and meta-analysis. Surg Endosc 27(8):2692–2703. https://doi.org/10.1007/s00464-013-2835-5
Giger OT, Comtesse SC, Lugli A, Zlobec I, Kurrer MO (2012) Intra-tumoral budding in preoperative biopsy specimens predicts lymph node and distant metastasis in patients with colorectal cancer. Mod Pathol 25(7):1048–1053. https://doi.org/10.1038/modpathol.2012.56
Rogers AC, Gibbons D, Hanly AM, Hyland JM, O'Connell PR, Winter DC, Sheahan K (2014) Prognostic significance of tumor budding in rectal cancer biopsies before neoadjuvant therapy. Mod Pathol 27(1):156–162. https://doi.org/10.1038/modpathol.2013.124
Zlobec I, Hadrich M, Dawson H, Koelzer VH, Borner M, Mallaev M, Schnuriger B, Inderbitzin D, Lugli A (2014) Intratumoural budding (ITB) in preoperative biopsies predicts the presence of lymph node and distant metastases in colon and rectal cancer patients. Br J Cancer 110(4):1008–1013. https://doi.org/10.1038/bjc.2013.797
Marx A, Simon P, Simon R, Mirlacher M, Izbicki JR, Yekebas E, Kaifi JT, Terracciano L, Sauter G (2008) AMACR expression in colorectal cancer is associated with left-sided tumor localization. Virchows Archiv : an international journal of pathology 453(3):243–248. https://doi.org/10.1007/s00428-008-0646-1
Kai K, Aishima S, Aoki S, Takase Y, Uchihashi K, Masuda M, Nishijima-Matsunobu A, Yamamoto M, Ide K, Nakayama A, Yamasaki M, Toda S (2016) Cytokeratin immunohistochemistry improves interobserver variability between unskilled pathologists in the evaluation of tumor budding in T1 colorectal cancer. Pathol Int 66(2):75–82. https://doi.org/10.1111/pin.12374
Lee GH, Malietzis G, Askari A, Bernardo D, Al-Hassi HO, Clark SK (2015) Is right-sided colon cancer different to left-sided colorectal cancer? - a systematic review. Eur J Surg Oncol : the journal of the European Society of Surgical Oncology and the British Association of Surgical Oncology 41(3):300–308. https://doi.org/10.1016/j.ejso.2014.11.001
Steinestel K, Lennerz JK, Eder S, Kraft K, Arndt A (2014) Invasion pattern and histologic features of tumor aggressiveness correlate with MMR protein expression, but are independent of activating KRAS and BRAF mutations in CRC. Virchows Archiv : an international journal of pathology 465(2):155–163. https://doi.org/10.1007/s00428-014-1604-8
Greenson JK, Huang SC, Herron C, Moreno V, Bonner JD, Tomsho LP, Ben-Izhak O, Cohen HI, Trougouboff P, Bejhar J, Sova Y, Pinchev M, Rennert G, Gruber SB (2009) Pathologic predictors of microsatellite instability in colorectal cancer. Am J Surg Pathol 33(1):126–133. https://doi.org/10.1097/PAS.0b013e31817ec2b1
Nosho K, Baba Y, Tanaka N, Shima K, Hayashi M, Meyerhardt JA, Giovannucci E, Dranoff G, Fuchs CS, Ogino S (2010) Tumour-infiltrating T-cell subsets, molecular changes in colorectal cancer, and prognosis: cohort study and literature review. J Pathol 222(4):350–366. https://doi.org/10.1002/path.2774
Author information
Authors and Affiliations
Contributions
Andreas H. Marx: conceptualization, methodology, formal analysis, writing–original draft. Claudius Mickler: investigation. Guido Sauter: project administration, supervision. Ronald Simon: formal analysis. Luigi M Terracciano: development or design of methodology, creation of models resources, data curation. Jakob R Izbicki: Resources, data curation. Till S Clauditz: validation, visualization, investigation, writing–review and editing.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical responsibility of authors
All individuals listed as co-authors of the manuscript qualify for every one of the four criteria listed in the ICMJE recommendation for qualification of authorship.
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Marx, A.H., Mickler, C., Sauter, G. et al. High-grade intratumoral tumor budding is a predictor for lymphovascular invasion and adverse outcome in stage II colorectal cancer. Int J Colorectal Dis 35, 259–268 (2020). https://doi.org/10.1007/s00384-019-03478-w
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00384-019-03478-w