Abstract
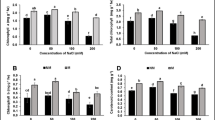
Salinization and sodication present significant threats to crop productivity in many parts of the world including Pakistan. Cultivating halophytes like quinoa presents a viable solution for the profitable use of salt-affected lands. This study specifically examines the performance and salt tolerance mechanism of four quinoa accessions under varying salinity and sodicity levels. In addition to a control group, different combinations of electrical conductivity and sodium adsorption ratio (ECe dS m−1 SAR levels) were established: 10:20, 10:40, 20:20, and 20:40, achieved by using a mixture of salts. Most of the quinoa cultivars exhibited robust growth, with the exception of GLN-22, which proved unable to withstand high levels of salinity and sodicity, resulting in a 78% reduction in yield. GLN-29, on the other hand, demonstrated superior performance across all levels of salinity and sodicity. UAF-Q7 excelled under conditions of high salinity and low sodicity compared to equivalent salinity levels but elevated sodicity. Meanwhile, GLN-33 exhibited enhanced growth under elevated sodicity levels but struggled in the face of high salinity stress. In terms of nutrient uptake, GLN-29 displayed a higher accumulation of Na+ (32%) in older leaves compared to younger ones, alongside elevated levels of antioxidant activity at all salinity and sodicity levels. Notably, GLN-29 exhibited excellent adaptation to both high salinity and sodicity levels, resulting in the highest grain yield (14.75 g/pot) and the salt tolerance mechanism was associated with highly efficient K+ retention and transport of Na+ to older leaves. This underscores the necessity for further comprehensive field studies to ascertain its suitability for the sustainable utilization of salt-affected soils.







Similar content being viewed by others
References
Abbas G, Amjad M, Saqib M, Murtaza B, Asif NM, Shabbir A (2021) Soil sodicity is more detrimental than salinity for quinoa (Chenopodium quinoa Willd.): A multivariate comparison of physiological, biochemical and nutritional quality attributes. J Agron Crop Sci 207:59–73
Abbas G, Saqib M, Akhtar J, Haq MAU (2015) Interactive effects of salinity and iron deficiency on different rice genotypes. J Plant Nutr Soil Sci 178:306–311. https://doi.org/10.1002/jpln.201400358
Abbas G, Saqib M, Akhtar J, Murtaza G (2017) Physiological and biochemical characterization of Acacia stenophylla and Acacia albida exposed to salinity under hydroponic conditions. Can J For Res 47:1293–1301. https://doi.org/10.1139/cjfr-2016-0499
Adolf VI, Jacobsen SE, Shabala S (2013) Salt tolerance mechanisms in quinoa (Chenopodium quinoa Willd.). Environ Exp Bot 92:43–54. https://doi.org/10.1016/j.envexpbot.2012.07.004
Adolf VI, Shabala S, Andersen MN, Razzaghi F, Jacobsen SE (2012) Varietal differences of quinoa’s tolerance to saline conditions. Plant Soil 357:117–129
Afzal I, Haq MZU, Ahmed S, Hirich A, Bazile D (2023) Challenges and perspectives for integrating quinoa into the agri-Food system. Plants 12:3361. https://doi.org/10.3390/plants12193361
Akram NA, Shafiq F, Ashraf M, Iqbal M, Ahmad P (2021) Advances in salt tolerance of some major fiber crops through classical and advanced biotechnological tools: a review. J Plant Growth Regul 40:891–905. https://doi.org/10.1007/s00344-020-10158-5
Amjad M, Akhtar S, Yang A, Akhtar J, Jacobsen SE (2015) Antioxidative response of quinoa exposed to iso-osmotic, ionic and non-ionic salt stress. J Agron Crop Sci 201:452–460. https://doi.org/10.1111/jac.12140
Bates LS, Waldren RP, Teare I (1973) Rapid determination of free proline for water-stress studies. Plant Soil Environ 39:205–207. https://doi.org/10.1007/BF00018060
Bertero D, Medan D, Hall A (1996) Changes in apical morphology during floral initiation and reproductive development in quinoa (Chenopodium quinoa Willd.). Ann Bot 78:317–324. https://academic.oup.com/aob/article/78/3/317/2587489
Beyrami H, Rahimian MH, Salehi M, Yazdani-Biouki R (2020) Effect of different levels of irrigation water salinity on quinoa (Chenopodium quinoa) yield and yield components in spring planting. J Crop Prod 12:111–120
Bose J, Rodrigo-Moreno A, Shabala S (2014) ROS homeostasis in halophytes in the context of salinity stress tolerance. J Exp Bot 65:1241–1257. https://doi.org/10.1093/jxb/ert430
Bottomley PJ, Angle JS, Weaver R (2020) Methods of soil analysis, Part 2: microbiological and biochemical properties, vol 12. Wiley, New York
Chance, B. and Maehly, A.C. (1955) Assay of catalases and peroxidases. Methods Enzymol 2:764-817. https://doi.org/10.1016/S0076-6879(55)02300-8
Flowers TJ, Colmer TD (2015) Plant salt tolerance: adaptations in halophytes. Ann Bot 115:327–331. https://doi.org/10.1093/aob/mcu267
Giannopolitis CN, Ries SK (1977) Superoxide dismutases: I. Occurrence in higher plants. Plant Physiol 59:309–314. https://doi.org/10.1104/pp.59.2.309
Haider G, Ghafoor A (1992) Manual of salinity research methods. International Water Logging and Salinity Research Institute, Lahore
Hariadi Y, Marandon K, Tian Y, Jacobsen SE, Shabala S (2011) Ionic and osmotic relations in quinoa (Chenopodium quinoa Willd.) plants grown at various salinity levels. J Exp Bot 62:185–193. https://doi.org/10.1093/jxb/erq257
Hasanuzzaman M, Nahar K, Alam M, Bhowmik PC, Hossain M, Rahman MM, Fujita M (2014) Potential use of halophytes to remediate saline soils. Biomed Res Int. https://doi.org/10.1155/2014/589341
Hunt R (1978) Plant Growth Analysis. Edward Arnold, London, 26–38
Hussain MI, Farooq M, Muscolo A, Rehman A (2020) Crop diversification and saline water irrigation as potential strategies to save freshwater resources and reclamation of marginal soils—review. Environ Sci Pollut Res 27:28695–28729. https://doi.org/10.1007/s11356-020-09111-6
Iqbal H, Yaning C, Waqas M, Rehman H, Shareef M, Iqbal S (2018) Hydrogen peroxide application improves quinoa performance by affecting physiological and biochemical mechanisms under water-deficit conditions. J Agron Crop Sci 204:541–553. https://doi.org/10.1111/jac.12284
Iqbal S, Basra SMA, Afzal I, Wahid A, Saddiq MS, Hafeez MB, Jacobsen SE (2019) Yield potential and salt tolerance of quinoa on salt-degraded soils of Pakistan. J Agron Crop Sci 205:13–21. https://doi.org/10.1111/jac.12290
Ismail H, Maksimović JD, Maksimović V, Shabala L, Živanović BD, Tian Y, Shabala S (2015) Rutin, a flavonoid with antioxidant activity, improves plant salinity tolerance by regulating K+ retention and Na+ exclusion from leaf mesophyll in quinoa and broad beans. Funct Plant Biol 43:75–86. https://doi.org/10.1071/FP15312
Jacobsen SE (2017) The scope for adaptation of quinoa in Northern Latitudes of Europe. J Agron Crop Sci 203:603–613. https://doi.org/10.1111/jac.12228
Koyro HW, Eisa SS (2008) Effect of salinity on composition, viability and germination of seeds of Chenopodium quinoa Willd. Plant Soil 302:79–90. https://doi.org/10.1007/s11104-007-9457-4
Maughan PJ, Turner TB, Coleman CE, Elzinga DB, Jellen EN, Morales JA, Udall JA, Fairbanks DJ, Bonifacio A (2009) Characterization of Salt Overly Sensitive 1 (SOS1) gene homoeologs in quinoa (Chenopodium quinoa Willd.). Genome 52:647–657. https://doi.org/10.1139/G09-041
Mittler R (2002) Oxidative stress, antioxidants and stress tolerance. Trend Plant Sci 7:405–410. https://doi.org/10.1016/S1360-1385(02)02312-9
Munns R, Tester M (2008) Mechanisms of salinity tolerance. Ann Rev Plant Biol 59:651–681. https://doi.org/10.1146/annvrev.arplant.59.032607.092911
Negrao S, Schmöckel S, Tester M (2017) Evaluating physiological responses of plants to salinity stress. Ann Bot 119:1–11. https://doi.org/10.1093/aob/mcw191
Panta S, Flowers T, Lane P, Doyle R, Haros G, Shabala S (2014) Halophyte agriculture: success stories. Environ Exp Bot 107:71–83. https://doi.org/10.1016/j.envexpbot.2014.05.006
Parvez S, Abbas G, Shahid M, Amjad M, Hussain M, Asad SA, Imran M, Naeem MA (2020) Effect of salinity on physiological, biochemical and photostabilizing attributes of two genotypes of quinoa (Chenopodium quinoa Willd.) exposed to arsenic stress. Ecotoxicol Environ Saf 15:109814. https://doi.org/10.1016/j.ecoenv.2019.109814
Qureshi AS, Daba AW (2020) Evaluating growth and yield parameters of five quinoa (Chenopodium quinoa W.) genotypes under different salt stress conditions. J Agric Sci 12:128–140. https://doi.org/10.5539/jas.v12n3p128
Rangani J, Parida AK, Panda A, Kumari A (2016) Coordinated changes in antioxidative enzymes protect the photosynthetic machinery from salinity induced oxidative damage and confer salt tolerance in an extreme halophyte Salvadora persica L. Front Plant Sci 7:50–68. https://doi.org/10.3389/fpls.2016.00050
Rehman S, Abbas G, Shahid M, Saqib M, Farooq AB, Hussain M, Murtaza B, Amjad M, Naeem MA, Farooq A (2019) Effect of salinity on cadmium tolerance, ionic homeostasis and oxidative stress responses in conocarpus exposed to cadmium stress: Implications for phytoremediation. Ecotoxicol Environ Saf 171:146–153. https://doi.org/10.1016/j.ecoenv.2018.12.077
Riaz F, Abbas G, Saqib M, Amjad M, Farooq A, Ahmad S, Naeem MA, Umer M, Khalid MS, Ahmed K, Ahmad N (2020) Comparative effect of salinity on growth, ionic and physiological attributes of two quinoa genotypes. Pak J Agric Sci 57:115–122. https://doi.org/10.21162/PAKJAS/20.9018
Ruiz K, Biondi S, Martínez E, Orsini F, Antognoni F, Jacobsen SE (2016) Quinoa—a model crop for understanding salt-tolerance mechanisms in halophytes. Plant Biosyst 150:357–371. https://doi.org/10.1080/11263504.2015.1027317
Sairam RK, Rao KV, Srivastava G (2002) Differential response of wheat genotypes to long term salinity stress in relation to oxidative stress, antioxidant activity and osmolyte concentration. Plant Sci 163:1037–1046. https://doi.org/10.1016/S0168-9452(02)00278-9
Salinity US (1954) Laboratory staff. “Diagnosis and improvement of saline and alkali soils.” Agric Handb 60:83–100. https://doi.org/10.20710/dojo.71.1_18
Shabala S (2009) Salinity and programmed cell death: unravelling mechanisms for ion specific signalling. J Exp Bot 60:709–712. https://doi.org/10.1093/jxb/erp013
Shabala S, Shabala L, Van Volkenburgh E, Newman I (2005) Effect of divalent cations on ion fluxes and leaf photochemistry in salinized barley leaves. J Exp Bot 56:1369–1378. https://doi.org/10.1093/jxb/eri138
Shabala L, Mackay A, Tian Y, Jacobsen SE, Zhou D, Shabala S (2012) Oxidative stress protection and stomatal patterning as components of salinity tolerance mechanism in quinoa (Chenopodium quinoa). Physiol Plant 146:26–38. https://doi.org/10.1111/j.1399-3054.2012.01599.x
Shabala S, Hariadi Y, Jacobsen SE (2013) Genotypic difference in salinity tolerance in quinoa is determined by differential control of xylem Na+ loading and stomatal density. J Plant Physiol 170:906–914. https://doi.org/10.1016/j.jplph.2013.01.014
Shavrukov Y, Langridge P, Tester M (2009) Salinity tolerance and sodium exclusion in genus Triticum. Breed Sci 59:671–678. https://doi.org/10.1270/jsbbs.59.671
Stanschewski CS, Rey E, Fiene G, Craine EB, Wellman G, Melino VJ, Patiranage DSR, Johansen K, Schmöckel SM, Bertero D, Oakey H (2021) Quinoa phenotyping methodologies: an international consensus. Plants 10:1759. https://doi.org/10.3390/plants10091759
Steel RGD, Torrie JH and Dicky DA (1997) Principles and procedures of statistics : a biometrical approach, 3rd Edn. McGraw Hill, Inc. Book Co., New York, p 352–358
Syed A, Sarwar G, Shah SH, Muhammad S (2021) Soil salinity research in 21st century in Pakistan: its impact on availability of plant nutrients, growth and yield of crops. Commun Soil Sci Plant Anal 52:183–200. https://doi.org/10.1080/00103624.2020.1854294
Szabados L, Savouré A (2010) Proline: a multifunctional amino acid. Trend Plant Sci 15:89–97. https://doi.org/10.1016/j.tplants.2009.11.009
Tahjib-Ul-Arif M, Siddiqui MN, Sohag AA, Sakil MA, Rahman MM, Polash MA, Mostofa MG, Tran LS (2018) Salicylic acid-mediated enhancement of photosynthesis attributes and antioxidant capacity contributes to yield improvement of maize plants under salt stress. J Plant Growth Regul 37:1318–1330
Takagi H, Yamada S (2013) Roles of enzymes in anti-oxidative response system on three species of chenopodiaceous halophytes under NaCl-stress condition. Soil Sci Plant Nutr 59:603–611. https://doi.org/10.1080/00380768.2013.809600
Tavakkoli E, Fatehi F, Coventry S, Rengasamy P, McDonald GK (2011) Additive effects of Na+ and Cl– ions on barley growth under salinity stress. J Exp Bot 62:2189–2203. https://doi.org/10.1093/jxb/erq422
Waqas M, Yaning C, Iqbal H, Shareef M, Rehman H, Yang Y (2017) Paclobutrazol improves salt tolerance in quinoa: beyond the stomatal and biochemical interventions. J Agron Crop Sci 203:315–322. https://doi.org/10.1111/jac.12217
Acknowledgements
The authors are highly thankful to Professor Mark Tester, KAUST, Saudi Arabia for the provision of quinoa accessions and Department of Agronomy, UAF for the provision of quinoa cultivar UAF-Q7.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Author information
Authors and Affiliations
Contributions
IA and GM: Conception and Design of the work, provision of resources, supervision. AZ and AR: Conduct experiment, acquisition, analysis, and interpretation of data, draft the work. WS and ZURF: Data analysis and revision of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that he/she has no conflict of interest.
Additional information
Handling Author: Vijay Pratap Singh.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zafar, A., Murtaza, G., Afzal, I. et al. Comparative Physiological and Biochemical Mechanisms of Salt Tolerance in Four Quinoa Cultivars Under Varying Salinity and Sodicity Levels. J Plant Growth Regul (2024). https://doi.org/10.1007/s00344-024-11315-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00344-024-11315-w