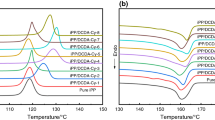
Abstract
In this work, isotactic polypropylene (iPP) was modified with an organic phosphate salt nucleating agent LPN-9081 and CaCO3. The synergistic effect of LNP-9801 and CaCO3 in iPP was investigated using differential scanning calorimetry (DSC), scanning electronic microscope (SEM), and mechanical property tests. The DSC results showed that the peak crystallization temperature (Tc) of modified iPP was increased by about 10 °C when compared to that of pure iPP, this indicates that LNP-9801 and CaCO3 increased the crystallization rate of iPP. The mechanical property tests demonstrated significant improvements in bending modulus and impact strength of modified iPP (impact strength increased by 33.9%, flexural modulus increased by 100.1%), which achieved an optimal balance of stiffness and toughness. Furthermore, the SEM results indicated that LPN-9081 contributed to the good dispersion of CaCO3 in the iPP matrix. All of the above results show that organic phosphate salt nucleating agent LPN-9081 and CaCO3 had good synergistic effects in iPP.
Graphical abstract




Similar content being viewed by others
References
Jiang Q, Jia H, Wang J (2012) Effects of nucleating agents on crystallization behavior and mechanical properties of high-fluid polypropylene. Iran Polym J 21:201–209
Tsioptsias C, Leontiadis K, Tzimpilis E (2020) Polypropylene nanocomposite fibers: a review of current trends and new developments. J Plast Film Sheeting 37:283–311
Horváth F, Bodrogi D, Hilt B (2022) Organogelators with dual β- and α-nucleating ability in isotactic polypropylene. J Therm Anal Calorim 147:9451–9468
Liu L, Yang W, Chen X (2023) Ethylene comonomer-directed epitaxial nucleation and growth of β-nucleated isotactic polypropylene. Macromolecules 56:1965–1972
Bazan P, Salasińska K, Kuciel S (2021) Flame retardant polypropylene reinforced with natural additives. Ind Crops Prod 164:113356–113368
Ichim M, Stelea L, Filip I (2022) Thermal and mechanical characterization of coir fiber–reinforced polypropylene biocomposites. Crystals 12:1249–12665
Tang W, Xu J, Fan Q (2022) Rheological behavior and mechanical properties of ultra-high-filled wood fiber/polypropylene composites using waste wood sawdust and recycled polypropylene as raw materials. Constr Build Mater 351:128977–128989
Castillo LA, Barbosa SE (2020) Comparative analysis of crystallization behavior induced by different mineral fillers in polypropylene nanocomposites. Nanomater Nanotechnol 10:184798042092275
Girones J, Vo LTT, Haudin J-M (2017) Crystallization of polypropylene in the presence of biomass-based fillers of different compositions. Polymer 127:220–231
Vakili MH, Ebadi-Dehaghani H, Haghshenas-Fard M (2011) Crystallization and thermal conductivity of CaCO3 nanoparticle filled polypropylene. J Macromol Sci Part B 50:1637–1645
Gao S, Li B, Bai P (2011) Effect of polysiloxane and silane-modified SiO2 on a novel intumescent flame retardant polypropylene system. Polym Adv Technol 22:2609–2616
Hu N, Tang E, Chang D (2021) Modification of CaCO3 nanoparticle by styrene-acrylic polymer emulsion spraying and its application in polypropylene material. Powder Technol 394:83–91
Gu Y, Liu Y, Fan Z (2023) Influence of β-nucleating compound agents on the mechanical properties and crystallization behavior of polypropylene random copolymer. J Wuhan Univ Technol Mater Sci Ed 38:237–243
Zhang Y, Liu H, Zhang L (2013) Influence of β nucleation agent on the dispersion of nano-CaCO3 in isotactic polypropylene matrix. J Appl Polym Sci 128:3382–3389
Thenepalli T, Jun AY, Han C (2015) A strategy of precipitated calcium carbonate (CaCO3) fillers for enhancing the mechanical properties of polypropylene polymers. Korean J Chem Eng 32:1009–1022
Al-Samhan M, Al-Attar F (2022) Comparative analysis of the mechanical, thermal and barrier properties of polypropylene incorporated with CaCO3 and nano CaCO3. Surf Interfaces 31:102055–102061
Younis AA, El-Wakil AA (2021) New composites from waste polypropylene/eggshell characterized by high flame retardant and mechanical properties. Fibers Polym 22:3456–3468
García-López D, Merino JC, Pastor JM (2003) Influence of the CaCO3 nanoparticles on the molecular orientation of the polypropylene matrix. J Appl Polym Sci 88:947–952
Li C-q, Liang C, Chen Z-m (2021) Surface modification of calcium carbonate: a review of theories, methods and applications. J Central South Univ 28:2589–2611
Lv X, Kang M, Yuan L (2020) Quantitative evaluation of fillers dispersion state in CaCO3/polypropylene composites through visualization and fractal analysis. Polym Compos 41:1605–1613
Zhu YD, Allen GC, Jones PG (2014) Dispersion characterisation of CaCO3 particles in PP/CaCO3 composites. Compos A Appl Sci Manuf 60:38–43
Ersoy O, Köse H (2020) Comparison of the effect of reactive and nonreactive treatments on the dispersion characteristics of a calcium carbonate (calcite) filler in a polypropylene matrix composite. Polym Compos 41:3483–3490
Hernández Y, Lozano T, Morales-Cepeda AB (2019) Stearic acid as interface modifier and lubricant agent of the system: polypropylene/calcium carbonate nanoparticles. Polym Eng Sci 59:E279–E285
Jing X, Gong W, Feng Z (2017) Novel comb-like copolymer dispersant for polypropylene/CaCO3 composites and Its influence on dispersion, crystallization, mechanical, and thermal properties. Polym-Plast Technol Eng 57:986–996
Al-Samhan M, Al-Attar F, Al-Fadhli J (2021) The influence of nano CaCO3 on nucleation and interface of PP nano composite: matrix processability and impact resistance. Polymers 13:1389–1398
Jing Y, Nai X, Dang L (2018) Reinforcing polypropylene with calcium carbonate of different morphologies and polymorphs. Sci Eng Compos Mater 25:745–751
Jiang X, Fan Y, Li F (2012) Preparation and properties of dynamically cured polypropylene (PP)/maleic anhydride–grafted polypropylene (MAH-g-PP)/calcium carbonate (CaCO3)/epoxy composites. J Thermoplast Compos Mater 26:1192–1205
Yoshimoto S, Ueda T, Yamanaka K (2001) Epitaxial act of sodium 2,2′-methylene-bis-(4,6-di-t-butylphenylene)phosphate on isotactic polypropylene. Polymer 42:9627–9631
Zhang Y-F, Xin Z (2006) Effects of substituted aromatic heterocyclic phosphate salts on properties, crystallization, and melting behaviors of isotactic polypropylene. J Appl Polym Sci 100:4868–4874
Li J, Liang Z, Gao C (2021) The application of organic phosphate nucleating agents in polypropylene with different molecular weights. Crystals 11:1543
Long L, He W, Zhang M (2015) Nucleation effects of sodium and ammonium salts of 2,2′-methylene-bis-(4,6-di-t-butylphenylene)phosphate in isotactic polypropylene. Polym Eng Sci 55:22–28
Thio YS, Argon AS, Cohen RE (2002) Toughening of isotactic polypropylene with CaCO3 particles. Polymer 43:3661–3674
Peng Y, Musah M, Via B (2021) Calcium carbonate particles filled homopolymer polypropylene at different loading levels: mechanical properties characterization and materials failure analysis. J Compos Sci 5:302
Zhang YF, Zhou PZ, Li Y (2019) The influences of α/β compound nucleating agents based on octamethylenedicarboxylic dibenzoylhydrazide on crystallization and melting behavior of isotactic polypropylene. Polym Adv Technol 30:1777–1788
Sarturato ACP, Dos Anjos EGR, Marini J (2023) Polypropylene/talc/graphene nanoplates (GNP) hybrid composites: effect of GNP content on the thermal, rheological, mechanical, and electrical properties. J Appl Polym Sci 140:e53657
Lv Z, Hu C, Xue J (2008) Effect of zeolite 5A on the crystalline behavior of polypropylene (PP) in PP/β-nucleating agent system. Polym Compos 29:1291–1296
Ding Q, Fu H, Hua C (2020) Effect of β-nucleating agent on crystallization of wollastonite-filled recycled polypropylene composites. J Therm Anal Calorim 144:713–719
Grząbka-Zasadzińska A, Klapiszewski Ł, Jesionowski T (2020) Functional MgO–lignin hybrids and their application as fillers for polypropylene composites. Molecules 25:864
Aguilar H, Yazdani-Pedram M, Toro P (2014) Synergic effect of two inorganic fillers on the mechanical and thermal properties of hybrid polypropylene composites. J Chil Chem Soc 59:2468–2473
Chen H, Wang M, Lin Y (2007) Morphology and mechanical property of binary and ternary polypropylene nanocomposites with nanoclay and CaCO3 particles. J Appl Polym Sci 106:3409–3416
Chen M, Wan C, Shou W (2008) Effects of interfacial adhesion on properties of polypropylene/Wollastonite composites. J Appl Polym Sci 107:1718–1723
Yao J, Hu H, Sun Z (2021) Synchronously strengthen and toughen polypropylene using tartaric acid-modified nano-CaCO3. Nanomaterials 11:2493–2507
Jiang X, Zhao S, Meng X (2018) Effect of the metal phenylphosphonates on the nonisothermal crystallization and performance of isotactic polypropylene. J Polym Sci Part B: Polym Phys 57:161–173
Momen O, Mehrabi-Mazidi M, Jahangiri N (2015) Isotactic polypropylene (PP) modified by ABS and CaCO3 nanoparticles: effect of composition and compatibilization on the phase morphology, mechanical properties and fracture behavior. Polym Bull 72:2757–2782
Fan T, Zhao Q, Guo W (2021) Effects of inorganic particles on the crystallization, mechanical properties and cellular structure of foamed PP composites in the IMD/MIM process. RSC Adv 11:36651–36662
Liang JZ, Wu CB (2012) Fractal characterization of impact fracture surface of polypropylene nanocomposites. Adv Polym Technol 31:71–81
Weon JI, Gam KT, Boo WJ (2006) Impact-toughening mechanisms of calcium carbonate-reinforced polypropylene nanocomposite. J Appl Polym Sci 99:3070–3076
Liang JZ, Tang CY, Li RKY (1998) Mechanical properties of polypropylene/CaCO3 composites. Met Mater 4:616–619
Yao ZT, Chen T, Li HY (2013) Mechanical and thermal properties of polypropylene (PP) composites filled with modified shell waste. J Hazard Mater 262:212–217
Bartczak Z, Argon AS, Cohen RE (1999) Toughness mechanism in semi-crystalline polymer blends: II. High-density polyethylene toughened with calcium carbonate filler particles. Polymer 40:2347–2365
Kiss A, Fekete E, Pukanszky B (2007) Aggregation of CaCO3 particles in PP composites: effect of surface coating. Compos Sci Technol 67:1574–1583
Zebarjad SM, Tahani M, Sajjadi SA (2004) Influence of filler particles on deformation and fracture mechanism of isotactic polypropylene. J Mater Process Technol 155–156:1459–1464
Acknowledgements
This work was financially supported by Natural Science Foundation of Changsha (No. kq2202184) and Hunan Provincial Science & Technology Department (No. 2021GK5007).
Funding
Natural Science Foundation of Changsha, kq2202184, Yue-Fei Zhang, Hunan Provincial Science and Technology Department, 2021GK5007, Yue-Fei Zhang
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing financial interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Dai, X., Li, Z., Wu, X. et al. The synergistic effect of an organic phosphate salt nucleating agent and CaCO3 in isotactic polypropylene. Polym. Bull. 81, 7157–7169 (2024). https://doi.org/10.1007/s00289-023-05051-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00289-023-05051-6