Abstract
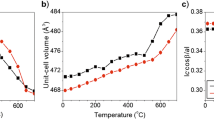
The thermal dehydration process of fibroferrite, FeOH(SO4)·5H2O, a secondary iron-bearing hydrous sulfate, was investigated by in situ high-temperature synchrotron X-ray powder diffraction (HT-XRPD), in situ high-temperature Fourier transform infrared spectroscopy (HT-FTIR) and thermal analysis (TGA-DTA) combined with evolved gas mass spectrometry. The data analysis allowed the determination of the stability fields and the reaction paths for this mineral as well as characterization of its high-temperature products. Five main endothermic peaks are observed in the DTA curve collected from room T up to 800 °C. Mass spectrometry of gases evolved during thermogravimetric analysis confirms that the first four mass loss steps are due to water emission, while the fifth is due to a dehydroxylation process; the final step is due to the decomposition of the remaining sulfate ion. The temperature behavior of the different phases occurring during the heating process was analyzed, and the induced structural changes are discussed. In particular, the crystal structure of a new phase, FeOH(SO4)·4H2O, appearing at about 80 °C due to release of one interstitial H2O molecule, was solved by ab initio real-space and reciprocal-space methods. This study contributes to further understanding of the dehydration mechanism and thermal stability of secondary sulfate minerals.









Similar content being viewed by others
References
Alpers CN, Jambor JL and Nordstrom DK (eds) (2000) Sulfate minerals: crystallography, geochemistry and environmental significance. Mineralogical Society of America, Rev Mineral Geochem 40, Washington, DC, xiv + 608 pp
Altomare C, Cuocci C, Giacovazzo C, Moliterni A, Rizzi R, Corriero N, Falcicchio A (2013) EXPO2013: a kit of tools for phasing crystal structures from powder data. J Appl Cryst 46:1231–1235
Amemija Y (1990) Imaging plate-X-ray area detector based on photostimulable phosphor. Synchrotron Radiat News 3:21–26
Atencio D, Hypolitho R (1993) Minerais e equivalentes sintéticos do sistema Fe2O3–SO3–H2O. Bol IG-USP Ser Cient 24:67–75
Christidis PC, Rentzeperis PJ (1976) The crystal structure of rhombohedral Fe2(SO4)3. Z Kristallogr 144:341–352
Della Ventura G, Ventruti G, Bellatreccia F, Scordari F, Cestelli Guidi M (2013) FTIR transmission spectroscopy of sideronatrite, a sodium-iron hydrous sulfate. Mineral Mag 77:499–507
Fitzpatrick RW, Fritsch E, Self PG (1996) Interpretation of soil features produced by ancient and modern process in degraded landscapes; V. Development of saline sulfidic features in non-tidal seepage areas. Geoderma 69:1–29
Gomez MA, Ventruti G, Celikin M, Assaaoudi H, Putz H, Becze L, Leea KE, Demopoulos GP (2013) The nature of synthetic basic ferric arsenate sulfate (Fe(AsO4)1−x(SO4)x(OH)x) and basic ferric sulfate (FeOHSO4): their crystallographic, molecular and electronic structure with applications in the environment and energy. RSC Adv 37:16840–16849
Larson AC, Von Dreele RB (2000) General structure analysis system (GSAS). Los Alamos National Laboratory Report LAUR 86–748
Meneghini C, Artioli G, Balerna A, Gualtieri AF, Norby P, Mobilio S (2001) Multipurpose imaging-plate camera for in situ powder XRD at the GILDA beamline. J Synchrotron Radiat 8:1162–1166
Norby P (1996) In situ time-resolved synchrotron powder diffraction studies of syntheses and chemical reactions. Mater Sci Forum 147:228–231
Palache C, Berman H, Frondel C (1951) The system of mineralogy of James Dwight Dana and Edward Salisbury Dana, Yale University 1837–1892, vol 2, 7th edn. Wiley, New York
Radica F, Della Ventura G, Bellatreccia F, Cestelli Guidi M (2015) HT-FTIR micro-spectroscopy of cordierite: the CO2 absorbance from in situ and quenched experiments. Phys Chem Miner 43:63–91
Scordari F (1981) Fibroferrite: a mineral with a Fe(OH)(H2O)2SO4 spiral chain and its relationship to Fe(OH)SO4, butlerite and parabutlerite. Tsch Mineral Petrogr 28:17–29
Sun M, Rousse G, Abakumov AM, Van Tendeloo G, Sougrati MT, Courty M, Doublet ML, Tarascon JM (2014) An oxysulfate Fe2O(SO4)2 electrode for sustainable Li-based batteries. J Am Chem Soc 136:12658–12666
Ventruti G, Scordari F, Schingaro E, Gualtieri AF, Meneghini C (2005) The order-disorder character of FeOHSO4 obtained from the thermal decomposition of metahohmannite, Fe +32 [O(SO4)2]·4H2O. Am Mineral 90:679–686
Ventruti G, Scordari F, Della Ventura G, Bellatreccia F, Gualtieri AF, Lausi A (2013) The thermal stability of sideronatrite and its decomposition products in the system Na2O–Fe2O3–SO2–H2O. Phys Chem Miner 40:659–670
Ventruti G, Della Ventura G, Orlando R, Scordari F (2015a) Crystal-structure and vibrational spectroscopy of hohmannite, Fe2[(SO4)2O]8H2O. Mineral Mag 79:11–24
Ventruti G, Della Ventura G, Scordari F, Susta U, Gualtieri AF (2015b) In-situ high-temperature XRD and FTIR investigation of hohmannite, a water-rich Fe-sulfate, and its decomposition products. J Therm Anal Calorim 119(3):1793–1802
Ventruti G, Della Ventura G, Bellatreccia F, Lacalamita M, Schingaro E (2016) Structure refinement, hydrogen bond system and vibrational spectroscopy of the iron sulfate fibroferrite, Fe(OH)SO4·5H2O
Zhang M, Salje EKH, Carpenter MA, Wang JY, Groat LA, Lager GA, Wang L, Beran A, Bismayer U (2007) Temperature dependence of IR absorption of hydrous/hydroxyl species in minerals and synthetic materials. Am Mineral 92:1502–1517
Acknowledgments
This work was supported by the Grants from the Italian Ministry of University and Research (PRIN 2010–2011). We thank two reviewers, S. Krivovichev and H. Effenberger, for their constructive comments, which helped us to improve the clarity of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ventruti, G., Ventura, G.D., Corriero, N. et al. In situ high-temperature X-ray diffraction and spectroscopic study of fibroferrite, FeOH(SO4)·5H2O. Phys Chem Minerals 43, 587–595 (2016). https://doi.org/10.1007/s00269-016-0819-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00269-016-0819-4