Abstract
Purpose
To investigate the feasibility of reducing the acquisition time for continuous dynamic positron emission tomography (PET) while retaining acceptable performance in quantifying kinetic metrics of 2-[18F]-fluoro-2-deoxy-D-glucose ([18F]FDG) in tumors.
Methods
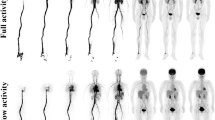
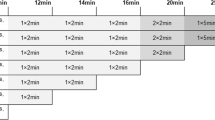
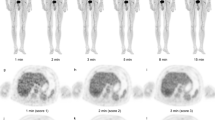
In total, 78 oncological patients underwent total-body dynamic PET imaging for ≥ 60 min, with 8, 20, and 50 patients receiving full activity (3.7 MBq/kg), half activity (1.85 MBq/kg), and ultra-low activity (0.37 MBq/kg) of [18F]FDG, respectively. The dynamic data were divided into 21-, 30-, 45- and ≥ 60-min groups. The kinetic analysis involved model fitting to derive constant rates (VB, K1 to k3, and Ki) for both tumors and normal tissues, using both reversible and irreversible two-tissue-compartment models. One-way ANOVA with repeated measures or the Freidman test compared the kinetic metrics among groups, while the Deming regression assessed the correlation of kinetic metrics among groups.
Results
All kinetic metrics in the 30-min and 45-min groups were statistically comparable to those in the ≥ 60-min group. The relative differences between the 30-min and ≥ 60-min groups ranged from 12.3% ± 15.1% for K1 to 29.8% ± 30.0% for VB, and those between the 45-min and ≥ 60-min groups ranged from 7.5% ± 8.7% for Ki to 24.0% ± 24.3% for VB. However, this comparability was not observed between the 21-min and ≥ 60-min groups. The significance trend of these comparisons remained consistent across different models (reversible or irreversible), administrated activity levels, and partial volume corrections for lesions. Significant correlations in tumor kinetic metrics were identified between the 30-/45-min and ≥ 60-min groups, with Deming regression slopes > 0.813. In addition, the comparability of kinetic metrics between the 30-min and ≥ 60-min groups were established for normal tissues.
Conclusion
The acquisition time for dynamic PET imaging can be reduced to 30 min without compromising the ability to reveal tumor kinetic metrics of [18F]FDG, using the total-body PET/CT system.






Similar content being viewed by others
Data availability
The dataset used and/or analyzed in current study are available from the corresponding author on reasonable request.
Code availability
Not applicable.
References
Strauss LG, Klippel S, Pan L, Schonleben K, Haberkorn U, Dimitrakopoulou-Strauss A. Assessment of quantitative FDG PET data in primary colorectal tumours: which parameters are important with respect to tumour detection? Eur J Nucl Med Mol Imaging. 2007;34(6):868–77.
Dimitrakopoulou-Strauss A, Strauss LG, Heichel T, Wu H, Burger C, Bernd L, et al. The role of quantitative 18F-FDG PET studies for the differentiation of malignant and benign bone lesions. J Nucl Med. 2002;43(4):510–8.
Strauss LG, Dimitrakopoulou-Strauss A, Koczan D, Bernd L, Haberkorn U, Ewerbeck V, et al. 18F-FDG kinetics and gene expression in giant cell tumors. J Nucl Med. 2004;45(9):1528–35.
Cochet A, Pigeonnat S, Khoury B, Vrigneaud JM, Touzery C, Berriolo-Riedinger A, et al. Evaluation of breast Tumor blood flow with dynamic first-pass 18F-FDG PET/CT: comparison with angiogenesis markers and prognostic factors. J Nucl Med. 2012;53(4):512–20.
Dimitrakopoulou-Strauss A, Strauss LG, Schwarzbach M, Burger C, Heichel T, Willeke F, et al. Dynamic PET 18F-FDG studies in patients with primary and recurrent soft-tissue sarcomas: impact on diagnosis and correlation with grading. J Nucl Med. 2001;42(5):713–20.
Rusten E, Rodal J, Revheim ME, Skretting A, Bruland OS, Malinen E. Quantitative dynamic 18FDG-PET and tracer kinetic analysis of soft tissue sarcomas. Acta Oncol. 2013;52(6):1160–7.
Dunnwald LK, Doot RK, Specht JM, Gralow JR, Ellis GK, Livingston RB, et al. PET Tumor metabolism in locally advanced Breast cancer patients undergoing neoadjuvant chemotherapy: value of static versus kinetic measures of fluorodeoxyglucose uptake. Clin Cancer Res. 2011;17(8):2400–9.
Dimitrakopoulou-Strauss A, Strauss LG, Egerer G, Vasamiliette J, Mechtersheimer G, Schmitt T, et al. Impact of dynamic 18F-FDG PET on the early prediction of therapy outcome in patients with high-risk soft-tissue sarcomas after neoadjuvant chemotherapy: a feasibility study. J Nucl Med. 2010;51(4):551–8.
Humbert O, Lasserre M, Bertaut A, Fumoleau P, Coutant C, Brunotte F, et al. Breast Cancer Blood Flow and Metabolism on Dual-Acquisition 18F-FDG PET: correlation with Tumor phenotype and neoadjuvant chemotherapy response. J Nucl Med. 2018;59(7):1035–41.
Dimitrakopoulou-Strauss A, Strauss LG, Burger C, Ruhl A, Irngartinger G, Stremmel W, et al. Prognostic aspects of 18F-FDG PET kinetics in patients with metastatic colorectal carcinoma receiving FOLFOX chemotherapy. J Nucl Med. 2004;45(9):1480–7.
Ilan E, Sandstrom M, Velikyan I, Sundin A, Eriksson B, Lubberink M. Parametric Net Influx Rate images of 68Ga-DOTATOC and 68Ga-DOTATATE: quantitative accuracy and improved image contrast. J Nucl Med. 2017;58(5):744–9.
Pantel AR, Viswanath V, Muzi M, Doot RK, Mankoff DA. Principles of Tracer Kinetic Analysis in Oncology, Part I: principles and overview of Methodology. J Nucl Med. 2022;63(3):342–52.
Fujimura Y, Kimura Y, Simeon FG, Dickstein LP, Pike VW, Innis RB, et al. Biodistribution and Radiation Dosimetry in humans of a new PET Ligand, 18F-PBR06, to Image Translocator protein (18 kDa). J Nucl Med. 2010;51(1):145–9.
Mizrahi R, Rusjan PM, Vitcu I, Ng A, Wilson AA, Houle S, et al. Whole body biodistribution and radiation dosimetry in humans of a new PET ligand, [18F]-FEPPA, to image translocator protein (18 kDa). Mol Imaging Biol. 2013;15(3):353–9.
Zhu W, Li Q, Bai B, Conti PS, Leahy RM. Patlak image estimation from dual time-point list-mode PET data. IEEE Trans Med Imaging. 2014;33(4):913–24.
Karakatsanis NA, Casey ME, Lodge MA, Rahmim A, Zaidi H. Whole-body direct 4D parametric PET imaging employing nested generalized Patlak expectation-maximization reconstruction. Phys Med Biol. 2016;61(15):5456–85.
Strauss LG, Pan L, Cheng C, Haberkorn U, Dimitrakopoulou-Strauss A. Shortened acquisition protocols for the quantitative assessment of the 2-tissue-compartment model using dynamic PET/CT 18F-FDG studies. J Nucl Med. 2011;52(3):379–85.
Wu Y, Feng T, Zhao Y, Xu T, Fu F, Huang Z, et al. Whole-body Parametric Imaging of 18F-FDG PET using uEXPLORER with reduced scanning time. J Nucl Med. 2022;63(4):622–8.
Badawi RD, Shi H, Hu P, Chen S, Xu T, Price PM, et al. First Human Imaging Studies with the EXPLORER Total-Body PET scanner. J Nucl Med. 2019;60(3):299–303.
Alberts I, Hunermund JN, Prenosil G, Mingels C, Bohn KP, Viscione M, et al. Clinical performance of long axial field of view PET/CT: a head-to-head intra-individual comparison of the Biograph Vision quadra with the Biograph Vision PET/CT. Eur J Nucl Med Mol Imaging. 2021;48(8):2395–404.
Liu G, Yu H, Shi D, Hu P, Hu Y, Tan H, et al. Short-time total-body dynamic PET imaging performance in quantifying the kinetic metrics of 18F-FDG in healthy volunteers. Eur J Nucl Med Mol Imaging. 2022;49(8):2493–503.
Boellaard R, Delgado-Bolton R, Oyen WJ, Giammarile F, Tatsch K, Eschner W, et al. FDG PET/CT: EANM procedure guidelines for tumour imaging: version 2.0. Eur J Nucl Med Mol Imaging. 2015;42(2):328–54.
Boudraa A, Zaidi H. Image segmentation techniques in nuclear medicine imaging. In: Zaidi H, editor. Quantitative analysis in Nuclear Medicine Imaging. New York: Springer; 2006. pp. 308–57.
Foster B, Bagci U, Mansoor A, Xu Z, Mollura DJ. A review on segmentation of positron emission tomography images. Comput Biol Med. 2014;50:76–96.
Liu G, Xu H, Hu P, Tan H, Zhang Y, Yu H, et al. Kinetic metrics of 18F-FDG in normal human organs identified by systematic dynamic total-body positron emission tomography. Eur J Nucl Med Mol Imaging. 2021;48(8):2363–72.
Liu G, Hu P, Yu H, Tan H, Zhang Y, Yin H, et al. Ultra-low-activity total-body dynamic PET imaging allows equal performance to full-activity PET imaging for investigating kinetic metrics of 18F-FDG in healthy volunteers. Eur J Nucl Med Mol Imaging. 2021;48(8):2373–83.
Wahl LM, Asselin MC, Nahmias C. Regions of interest in the venous sinuses as input functions for quantitative PET. J Nucl Med. 1999;40(10):1666–75.
Tseng J, Dunnwald LK, Schubert EK, Link JM, Minoshima S, Muzi M, et al. 18F-FDG kinetics in locally advanced Breast cancer: correlation with Tumor blood flow and changes in response to neoadjuvant chemotherapy. J Nucl Med. 2004;45(11):1829–37.
Srinivas SM, Dhurairaj T, Basu S, Bural G, Surti S, Alavi A. A recovery coefficient method for partial volume correction of PET images. Ann Nucl Med. 2009;23(4):341–8.
Torizuka T, Nobezawa S, Momiki S, Kasamatsu N, Kanno T, Yoshikawa E, et al. Short dynamic FDG-PET imaging protocol for patients with Lung cancer. Eur J Nucl Med. 2000;27(10):1538–42.
Dimitrakopoulou-Strauss A, Strauss LG, Egerer G, Vasamiliette J, Schmitt T, Haberkorn U, et al. Prediction of chemotherapy outcome in patients with metastatic soft tissue sarcomas based on dynamic FDG PET (dPET) and a multiparameter analysis. Eur J Nucl Med Mol Imaging. 2010;37(8):1481–9.
Feng T, Zhao Y, Shi H, Li H, Zhang X, Wang G, et al. Total-body quantitative Parametric Imaging of Early Kinetics of 18F-FDG. J Nucl Med. 2021;62(5):738–44.
Wang Z, Wu Y, Li X, Bai Y, Chen H, Ding J, et al. Comparison between a dual-time-window protocol and other simplified protocols for dynamic total-body 18F-FDG PET imaging. EJNMMI Phys. 2022;9(1):63.
Wu Y, Feng T, Shen Y, Fu F, Meng N, Li X, et al. Total-body parametric imaging using the Patlak model: feasibility of reduced scan time. Med Phys. 2022;49(7):4529–39.
Sari H, Eriksson L, Mingels C, Alberts I, Casey ME, Afshar-Oromieh A, et al. Feasibility of using abbreviated scan protocols with population-based input functions for accurate kinetic modeling of [18F]-FDG datasets from a long axial FOV PET scanner. Eur J Nucl Med Mol Imaging. 2023;50(2):257–65.
Funding
This study was funded by the Shanghai Municipal Key Clinical Specialty Project (grant number: SHSLCZDZK03401), the Major Science and Technology Projects for Major New Drug Creation (grant number: 2019ZX09302001), the Shanghai Science and Technology Committee program (grant number: 20DZ2201800), and the Three-year Action Plan of Clinical Skills and Innovation of Shanghai Hospital Development Center (grant number: SHDC2020CR3079B).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that there is not any conflict of interest regarding this study.
Ethics approval
This study was approved by the Ethics Committee of Zhongshan Hospital of Fudan University (approval number: B2019-160).
Consent to participate
Written informed consents were obtained from included subjects for participation of this study.
Consent for publication
The authors affirm that human research participants provided informed consent for publication of the studied data.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Guobing Liu, Yimeng Shi and Xiaoguang Hou contributed equally to this article.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Liu, G., Shi, Y., Hou, X. et al. Dynamic total-body PET/CT imaging with reduced acquisition time shows acceptable performance in quantification of [18F]FDG tumor kinetic metrics. Eur J Nucl Med Mol Imaging 51, 1371–1382 (2024). https://doi.org/10.1007/s00259-023-06526-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00259-023-06526-4