Abstract
Purpose
To quantitatively evaluate the minimally required scanning time of 3′-deoxy-3′-[18F]fluorothymidine ([18F]FLT) positron emission tomography (PET) dynamic acquisition for accurate kinetic assessment of the proliferation in breast cancer tumors.
Procedures
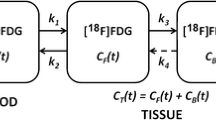
Within a therapeutic intervention trial, 26 breast tumors of 8 breast cancer patients were analyzed from 30-min dynamic [18F]FLT-PET acquisitions. PET/CT was acquired on a Gemini TF 64 system (Philips Healthcare) and reconstructed into 26 frames (8 × 15 s, 6 × 30 s, 5 × 1 min, 5 × 2 min, and 2 × 5 min). Maximum activity concentrations (Bq/ml) of volume of interests over tumors and plasma in descending aorta were obtained over time frames. Kinetic parameters were estimated using in-house developed software with the two-tissue three-compartment irreversible model (2TCM) (K1, k2, k3, and Ki; k4 = 0) and Patlak model (Ki) based on different acquisition durations (Td) (10, 12, 14, 16, 20, 25, and 30 min, separately). Different linear regression onset time (T0) points (1, 2, 3, 4, and 5 min) were applied in Patlak analysis. Ki of the 30-min data set was taken as the gold standard for comparison. Pearson product-moment correlation coefficient (R) of 0.9 was chosen as a limit for the correlation.
Results
The correlation of kinetic parameters between the gold standard and the abbreviated dynamic data series increased with longer Td from 10 to 30 min. k2 and k3 using 2TCM and Ki using Patlak model revealed poor correlations for dynamic PET with Td ≤ 14 min (k2: R = 0.84, 0.85, 0.86; k3: R = 0.67, 0.67, 0.67; Ki: R = 0.72, 0.78, 0.87 at Td = 10, 12, and 14 min, respectively). Excellent correlations were shown for all kinetic parameters when Td ≥ 16 min regardless of the kinetic model and T0 value (R > 0.9).
Conclusions
This study indicates that a 16-min dynamic PET acquisition appears to be sufficient to provide accurate [18F]FLT kinetics to quantitatively assess the proliferation in breast cancer lesions.






Similar content being viewed by others
References
American Cancer Society. Breast cancer facts & figures 2017–2018 (2017) Atlanta: American Cancer Society, Inc. https://www.cancer.org/content/dam/cancer-org/research/cancer-facts-and-statistics/breast-cancer-facts-and-figures/breast-cancer-facts-and-figures-2017-2018.pdf
Escalona S, Blasco JA, Reza MM, Andradas E, Gómez N (2010) A systematic review of FDG-PET in breast cancer. Med Oncol 27:114–129
Rosen EL, Eubank WB, Mankoff DA (2007) FDG PET, PET/CT, and breast cancer imaging. Radiographics 27:S215–S229
Bensch F, van Kruchten M, Lamberts LE, Schröder CP, Hospers GAP, Brouwers AH, van Vugt MATM, de Vries EGE (2013) Molecular imaging for monitoring treatment response in breast cancer patients. Eur J Pharmacol 717:2–11
Oude Munnink TH, Nagengast WB, Brouwers AH et al (2009) Molecular imaging of breast cancer. Breast 18:S66–S73
Lavayssiere R, Cabee AE, Filmont JE (2009) Positron emission tomography (PET) and breast cancer in clinical practice. Eur J Radiol 69:50–58
Smith IC, Welch AE, Hutcheon AW, Miller ID, Payne S, Chilcott F, Waikar S, Whitaker T, Ah-See AK, Eremin O, Heys SD, Gilbert FJ, Sharp PF (2000) Positron emission tomography using [(18)F]-fluorodeoxy-D-glucose to predict the pathologic response of breast cancer to primary chemotherapy. J Clin Oncol 18:1676–1688
Doot RK, Dunnwald LK, Schubert EK, Muzi M, Peterson LM, Kinahan PE, Kurland BF, Mankoff DA (2007) Dynamic and static approaches to quantifying 18F-FDG uptake for measuring cancer response to therapy, including the effect of granulocyte CSF. J Nucl Med 48:920–925
Been LB, Suurmeijer AJ, Cobben DC et al (2004) [18F]FLT-PET in oncology: current status and opportunities. Eur J Nucl Med Mol Imaging 31:1659–1672
Pio BS, Park CK, Pietras R, Hsueh WA, Satyamurthy N, Pegram MD, Czernin J, Phelps ME, Silverman DHS (2006) Usefulness of 3′-[F-18]fluoro-3′-deoxythymidine with positron emission tomography in predicting breast cancer response to therapy. Mol Imaging Biol 8:36–42
Chalkidou A, Landau DB, Odell EW, Cornelius VR, O’Doherty MJ, Marsden PK (2012) Correlation between Ki-67 immunohistochemistry and 18F-Fluorothymidine uptake in patients with cancer: a systematic review and meta-analysis. Eur J Cancer 48:3499–3513
Kenny L, Coombes RC, Vigushin DM, al-Nahhas A, Shousha S, Aboagye EO (2007) Imaging early changes in proliferation at 1 week post chemotherapy: a pilot study in breast cancer patients with 3′-deoxy-3′-[18F]fluorothymidine positron emission tomography. Eur J Nucl Med Mol Imaging 34:1339–1347
Contractor KB, Kenny LM, Stebbing J, Rosso L, Ahmad R, Jacob J, Challapalli A, Turkheimer F, al-Nahhas A, Sharma R, Coombes RC, Aboagye EO (2011) [18F]-3'Deoxy-3′-fluorothymidine positron emission tomography and breast cancer response to docetaxel. Clin Cancer Res 17:7664–7672
Bollineni VR, Kramer GM, Jansma EP, Liu Y, Oyen WJG (2016) A systematic review on [18F]FLT-PET uptake as a measure of treatment response in cancer patients. Eur J Cancer 55:81–97
Kenny LM, Vigushin DM, Al-Nahhas A et al (2005) Quantification of cellular proliferation in tumor and normal tissues of patients with breast cancer by [18F]fluorothymidine-positron emission tomography imaging: evaluation of analytical methods. Cancer Res 65:10104–10112
Watabe H, Ikoma Y, Kimura Y, Naganawa M, Shidahara M (2006) PET kinetic analysis—compartmental model. Ann Nucl Med 20:583–588
Muzi M, Vesselle H, Grierson JR, Mankoff DA, Schmidt RA, Peterson L, Wells JM, Krohn KA (2005) Kinetic analysis of 3′-deoxy-3′-fluorothymidine PET studies: validation studies in patients with lung cancer. J Nucl Med 46:274–282
Dimitrakopoulou-Strauss A, Pan L, Strauss LG (2012) Quantitative approaches of dynamic FDG-PET and PET/CT studies (dPET/CT) for the evaluation of oncological patients. Cancer Imaging 12:283–289
Patlak CS, Blasberg RG, Fenstermacher JD (1983) Graphical evaluation of blood-to-brain transfer constants from multiple-time uptake data. J Cereb Blood Flow Metab 3:1–7
Gramer BM, Muenzel D, Leber V, von Thaden AK, Feussner H, Schneider A, Vembar M, Soni N, Rummeny EJ, Huber AM (2012) Impact of iterative reconstruction on CNR and SNR in dynamic myocardial perfusion imaging in an animal model. Eur Radiol 22:2654–2661
Gaeta CM, Vercher-Conejero JL, Sher AC, Kohan A, Rubbert C, Avril N (2013) Recurrent and metastatic breast cancer PET, PET/CT, PET/MRI: FDG and new biomarkers. Q J Nucl Med Mol Imaging 57:352–366
Manohar K, Mittal BR, Senthil R, Kashyap R, Bhattacharya A, Singh G (2012) Clinical utility of F-18 FDG PET/CT in recurrent breast carcinoma. Nucl Med Commun 33:591–596
Raijmakers P, Temmerman OPP, Saridin CP, Heyligers IC, Becking AG, van Lingen A, Lammertsma AA (2014) Quantification of 18F-fluoride kinetics: evaluation of simplified methods. J Nucl Med 55:1122–1127
Veronese M, Rizzo G, Aboagye EO, Bertoldo A (2014) Parametric imaging of F-fluoro-3-deoxy-3-L-fluorothymidine PET data to investigate tumour heterogeneity. Eur J Nucl Med Mol Imaging 41:1781–1792
Krak NC, van der Hoeven JJ, Hoekstra OS et al (2003) Measuring [(18)F]FDG uptake in breast cancer during chemotherapy: comparison of analytical methods. Eur J Nucl Med Mol Imaging 30:674–681
Dunnwald LK, Gralow JR, Ellis GK, Livingston RB, Linden HM, Specht JM, Doot RK, Lawton TJ, Barlow WE, Kurland BF, Schubert EK, Mankoff DA (2008) Tumor metabolism and blood flow changes by positron emission tomography: relation to survival in patients treated with neoadjuvant chemotherapy for locally advanced breast cancer. J Clin Oncol 26:4449–4457
Dunnwald LK, Doot RK, Specht JM, Gralow JR, Ellis GK, Livingston RB, Linden HM, Gadi VK, Kurland BF, Schubert EK, Muzi M, Mankoff DA (2011) PET tumor metabolism in locally advanced breast cancer patients undergoing neoadjuvant chemotherapy: value of static versus kinetic measures of fluorodeoxyglucose uptake. Clin Cancer Res 17:2400–2409
Visser EP, Philippens ME, Kienhorst L et al (2008) Comparison of tumor volumes derived from glucose metabolic rate maps and SUV maps in dynamic 18F-FDG PET. J Nucl Med 49:892–898
Freedman NM, Sundaram SK, Kurdziel K, Carrasquillo J, Whatley M, Carson J, Sellers D, Libutti S, Yang J, Bacharach S (2003) Comparison of SUV and Patlak slope for monitoring of cancer therapy using serial PET scans. Eur J Nucl Med Mol Imaging 30:46–53
Cheebsumon P, Velasquez LM, Hoekstra CJ, Hayes W, Kloet RW, Hoetjes NJ, Smit EF, Hoekstra OS, Lammertsma AA, Boellaard R (2011) Measuring response to therapy using FDG PET: semi-quantitative and full kinetic analysis. Eur J Nucl Med Mol Imaging 38:832–842
Mankoff DA, Dunnwald LK, Gralow JR, Ellis GK, Charlop A, Lawton TJ, Schubert EK, Tseng J, Livingston RB (2002) Blood flow and metabolism in locally advanced breast cancer: relationship to response to therapy. J Nucl Med 43:500–509
Desilva A, Wuest M, Wang M, Hummel J, Mossman K, Wuest F, Hitt MM (2012) Comparative functional evaluation of immunocompetent mouse breast cancer models established from PyMT-tumors using small animal PET with [18F]FDG and [18F]FLT. Am J Nucl Med Mol Imaging 2:88–98
Shields AF, Briston DA, Chandupatla S, Douglas KA, Lawhorn-Crews J, Collins JM, Mangner TJ, Heilbrun LK, Muzik O (2005) A simplified analysis of [18F]3′-deoxy-3′-fluorothymidine metabolism and retention. Eur J Nucl Med Mol Imaging 32:1269–1275
Kramer GM, Frings V, Heijtel D, Smit EF, Hoekstra OS, Boellaard R, QuIC-ConCePT Consortium (2017) Parametric method performance for dynamic 3′-deoxy-3'-18F-fluorothymidine PET/CT in epidermal growth factor receptor-mutated non-small cell lung carcinoma patients before and during therapy. J Nucl Med 58:920–925
Acknowledgements
The authors acknowledge the help from Preethi Subramanian M.S. for the [18F]FLT data management.
Funding
This project was financially supported by NCI grant U01CA076576, Ohio Third Frontier ODSA TECH09-028, and the Wright Center of Innovation Development Fund.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Zhang, J., Liu, X., Knopp, M.I. et al. How Long of a Dynamic 3′-Deoxy-3′-[18F]fluorothymidine ([18F]FLT) PET Acquisition Is Needed for Robust Kinetic Analysis in Breast Cancer?. Mol Imaging Biol 21, 382–390 (2019). https://doi.org/10.1007/s11307-018-1231-x
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11307-018-1231-x