Abstract
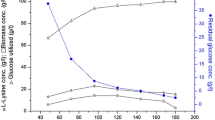
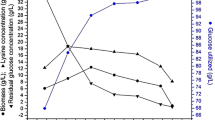
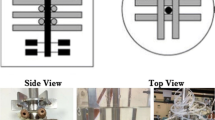
Biofilm-immobilized fermentation is a novel strategy that has been utilized in L-lysine fermentation. In this study, we describe a strategy for designing bioreactors for immobilized fermentation. We have constructed steel structures in which the carriers can be sewn, forming several star-like structures with different angles, and changing the ventilation robot to the aeration tray. In a 10-L bioreactor, this structure with 12 angles assisted the immobilized system to remedy the gap between free-cell and immobilized fermentation in the conversion rate. In a 50-L bioreactor, this enlarged structure with 16 angles illustrated a 4.61% higher conversion rate than the free-cell fermentation (67.75%) and increased the production by 28.56%. This successful case is the first step towards to industrial production of biofilm-based immobilized fermentation.
Key points
• The designed steel structure is useful for L-lysine immobilized fermentation in a 10-L bioreactor.
• The conversion rate of immobilized fermentation increased from 13.99 to 60.07% and is 1.03% higher than that of the free-cell fermentation.
• The conversion rate of the redesigned 50-L bioreactor is higher than that of free-cell fermentation.





Similar content being viewed by others
Data availability
The authors promise the availability of supporting data.
Change history
26 June 2023
A Correction to this paper has been published: https://doi.org/10.1007/s00253-023-12645-7
References
Berenjian A, Chan NL-C, Mahanama R, Talbot A, Regtop H, Kavanagh J, Dehghani F (2013) Effect of biofilm formation by Bacillus subtilis natto on menaquinone-7 biosynthesis. Mol Biotechnol 54:371–378
Chen Z, Liu G, Zhang J, Bao J (2019) A preliminary study on L-lysine fermentation from lignocellulose feedstock and techno-economic evaluation. Bioresour Technol 271:196–201
Ding S, Zhang D, Sha Y, Wang F, Liang C, Chen T, Sun W, Zhuang W, Yu B, Liu D, Li M, Ying H, Chen Y (2021) Efficient preparation of phytase from genetically modified Pichia pastoris in immobilized fermentation biofilms adsorbed on surface-modified cotton fibres. Process Biochem 111(2):69–78
Eggeling L, Bott M (2005) Handbook of Corynebacterium glutamicum. CRC Press LLC, Boca Raton
Feng S, Srinivasan S, Liu Y (2012) Redox potential-driven repeated batch ethanol fermentation under very-high-gravity conditions. Process Biochem 47(3):523–527
Fu T, Pan L, Shang Q, Yu G (2021) Fermentation of alginate and its derivatives by different enterotypes of human gut microbiota: towards personalized nutrition using enterotype-specific dietary fibers. Int J Biol Macromol 183:1649–1659
Howe EE, Jansen GR, Gilfillan EW (1965) Amino acid supplementation of cereal grains as related to the world food supply. Am J Clin Nutr 16:315–320
Israilides CJ, Weir ANC, Bull AT (1989) Effect of antibiotics on lysine production in free and immobilized cells of Bacillus subtilis. Appl Microbiol Biot 32:134–136
Jin C, Bao J (2021) Lysine production by dry biorefining of wheat straw and cofermentation of Corynrbacterium glutamincum. J Agric Food Chem 69(6):1900–1906
Kinoshita S, Nakayama K, Kitada S (1958) L-Lysine production using microbial auxotroph (preliminary reporter). J Gen Appl Microbiol 4:128–129
Krishnan MS, Taylor F, Davision BH, Nghiem NP (2000) Economic analysis of fuel ethanol production from corn starch using fluidized-bed bioreactors. Bioresource Technol 75(2):99–105
Liang C, Ding S, Sun W, Liu L, Zhao W, Zhang D, Ying H, Liu D, Yong C (2020) Biofilm-based fermentation: a novel immobilisation strategy for Saccharomyces cerevisiae cell cycle progression during ethanol production. Appl Microbiol Biot 104(17):7495–7505
Liu Q, Zhao N, Zou Y, Ying H, Chen Y (2020) Feasibility of ethanol production from expired rice by surface immobilization technology in a new type of packed bed pilot reactor. Renew Energ 149:321–328
McWhirter JR, Huttrt JC (1989) Improved oxygen mass transfer modelling for diffused/subsurface aeration system. AICHE J 35(9):1527–1534
Najafpour G, Younesi H, Ismail KSK (2004) Ethanol fermentation in an immobilized cell reactor using Saccharomyces cerevisiae. Bioresour Technol 92(3):251–260
Neuner A, Wagner I, Sieker T, Ulber R, Schneider K, Peifer S, Heinzle E (2013) Production of L-lysine on different silage juices using genetically engineered Corynebacterium glutamicum. J Biotechnol 163(2):217–224
Nishimura T, Vertès AA, Shinoda Y, Inui M, Yukawa H (2007) Anaerobic growth of Corynebacterium glutamicun using nitrate as a terminal electron acceptor. Appl Microbiol Biotechnol 75:889–897
Osuga J, Mori A, Kato J (1984) Acetic acid production by immobilized Acetobacter aceti cells entrapped in k-Carrageenan gel. J Ferment Bioeng 65(2):139–149
Pfefferle W, Moeckel B, Bathe B, Marx A (2003) Biotechnological manufacture of L-lysine. Adv Biochem Eng Biotechnol 79:59–112
Razak MA, Viswanath B (2015) Comparative studies for the biotechnological production of L-Lysine by immobilized cells of wild-type Corynebacterium glutamicun ATCC 13032 and mutant MH 20–22 B. 3 Biotech 5:765–774
Razak MA, Viswanath B (2015) Optimization of fermentation upstream parameters and immobilization of Corynebacterium glutamicum MH 20–22 B cells to enhance the production of L-lysine. 3 Biotech 5:531–540
Ren P, Chen T, Liu N, Sun W, Hu G, Yu Y, Yu B, Ouyang P, Liu D, Chen Y (2020) Efficient biofilm-based fermentation strategies by eDNA formation for l-proline production with Corynebacterium glutamicum. ACS Omega 5:33314–33322
Sakai M, Futamata H, Kanazawa S (2003) Effects of High Concentrations of inorganic salts on swarming ability in fluorescent Pseudomonas strains. Biosci Biotech Bioch 67(7):1479–1484
Takeno S, Ohnishi J, Komatsu T, Masaki T, Sen K, Ikeda M (2007) Anaerobic growth and potential for amino acid production by nitrate respiration in Corynebacterium glutamicun. Appl Microbiol Biotechnol 75:1173–1182
Warnecke T, Gill RT (2005) Organic acid toxicity, tolerance, and production in Escherichia coli biorefining application. Microb Cell Fact 4(1):25
Wieschalka S, Blombach B, Bott M, Eikmanns BJ (2013) Bio-based production of organic acids with Corynebacterium glutamicum. Microb Biotechnol 6:87–102
Xu JZ, Yu HB, Liu LM, Zhang WG (2019) Metabolic engineering of glucose uptake systems in Corynebacterium glutamicum for improving the efficiency of l-lysine production. J Ind Microbiol Biotechnol 46:937–949
Xu JZ, Ruan HZ, Yu HB, Liu LM, Zhang WG (2020) Metabolic engineering of carbohydrate metabolism systems in Corynebacterium glutamicum for improving the efficiency of L-lysine production from mixed sugar. Microb Cell Fact 19:39
Yang L, Zheng C, Chen Y, Shi X, Ying Z, Ying H (2019) Nitric oxide increases biofilm formation in Saccharomyces cerevisiae by activating the transcriptional factor Mac1p and thereby regulating the transmembrane protein Ctr1. Biotechnol Biofuels 12(1):30
Funding
This work was supported by the National Key R&D Program of China (Grant No. 2021YFC2101100); the National Natural Science Foundation of China (22178176): the National Key Research and Development Program of China (Grant No. 2018YFB1501705); the key program of the National Natural Science Foundation of China (Grant No. 21636003); the Outstanding Youth Foundation of China (Grant No. SBK2017010373); the Program for Changjiang Scholars and Innovative Research Team in University (IRT_14R28); Jiangsu National Synergetic Innovation Center for Advanced Materials (SICAM); the Priority Academic Program Development of Jiangsu Higher Education Institutions (PAPD); Key Research and Development Program of Nanjing Jiangbei New Area (ZDYF20200220); the National Natural Science Foundation of China (Grant No.: 22178172).
Author information
Authors and Affiliations
Contributions
GM and WZ contributed equally to this work and should be considered as co-first authors. GM, QL, CZ, and YZ performed the fermentation experiments. WZ and YZ drafted and revised the manuscript. WS, NZ, and DL participated in the fermentation experiments and SEM assay. YC and WS participated in the design of the study and revised the manuscript. HY conceived of the study and participated in its design. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Ethics approval and consent to participate
No participate.
Consent for publication
The authors have consented for publication.
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Meng, G., Zhao, W., Liu, Q. et al. Strategy for the design of a bioreactor for L-lysine immobilized fermentation using Corynebacterium glutamicum. Appl Microbiol Biotechnol 106, 5449–5458 (2022). https://doi.org/10.1007/s00253-022-12103-w
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-022-12103-w