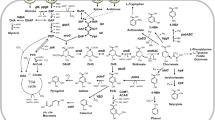
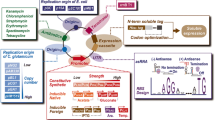
Abstract
Corynebacterium glutamicum (C. glutamicum), an important industrial workhorse, is capable of efficiently producing a variety of value-added chemicals and fuels beyond amino acids. C. glutamicum has a broad natural substrate spectrum and can simultaneously utilize various carbon sources in blends. The substrate spectrum of C. glutamicum has been further extended by detailed knowledge of carbon core metabolism and well-established genetic tools and engineering strategies. At present, many pathways have been successfully engineered in C. glutamicum for access to alternative renewable sources to produce natural or non-natural products, making C. glutamicum a promising and favorable microbial cell factory. In this review, we mainly focus on synthetic biology and metabolic engineering strategies for developing synthetic strains that grow on renewable sources to produce the target products. At the same time, we also explore the promotion and future challenges of existing synthetic biology platforms for industrial platform microorganism metabolic engineering efforts.



Similar content being viewed by others
References
Anusree M, Wendisch VF, Nampoothiri KM (2016) Co-expression of endoglucanase and beta-glucosidase in Corynebacterium glutamicum DM1729 towards direct lysine fermentation from cellulose. Bioresour Technol 213:239–244. https://doi.org/10.1016/j.biortech.2016.03.019
Atsumi S, Hanai T, Liao JC (2008) Non-fermentative pathways for synthesis of branched-chain higher alcohols as biofuels. Nature 451(7174):86–89. https://doi.org/10.1038/nature06450
Baritugo KA, Kim HT, David Y, Choi JI, Hong SH, Jeong KJ, Choi JH, Joo JC, Park SJ (2018) Metabolic engineering of Corynebacterium glutamicum for fermentative production of chemicals in biorefinery. Appl Microbiol Biotechnol 102(9):3915–3937. https://doi.org/10.1007/s00253-018-8896-6
Baumgart M, Unthan S, Rückert C, Sivalingam J, Grünberger A, Kalinowski J, Bott M, Noack S, Frunzke J (2013) Construction of a prophage-free variant of Corynebacterium glutamicum ATCC 13032 for use as a platform strain for basic research and industrial biotechnology. Appl Environ Microbiol 79(19):6006–6015. https://doi.org/10.1128/AEM.01634-13
Baumgart M, Unthan S, Kloss R, Radek A, Polen T, Tenhaef N, Muller MF, Kuberl A, Siebert D, Bruhl N, Marin K, Hans S, Kramer R, Bott M, Kalinowski J, Wiechert W, Seibold G, Frunzke J, Ruckert C, Wendisch VF, Noack S (2018) Corynebacterium glutamicum chassis C1*: building and testing a novel platform host for synthetic biology and industrial biotechnology. ACS Synth Biol 7(1):132–144. https://doi.org/10.1021/acssynbio.7b00261
Becker J, Wittmann C (2012) Bio-based production of chemicals, materials and fuels—Corynebacterium glutamicum as versatile cell factory. Curr Opin Biotechnol 23(4):631–640. https://doi.org/10.1016/j.copbio.2011.11.012
Becker J, Giesselmann G, Hoffmann SL, Wittmann C (2018) Corynebacterium glutamicum for sustainable bioproduction: from metabolic physiology to systems metabolic engineering. Adv Biochem Eng Biotechnol 162:217–263. https://doi.org/10.1007/10_2016_21
Beckers V, Poblete-Castro I, Tomasch J, Wittmann C (2016) Integrated analysis of gene expression and metabolic fluxes in PHA-producing Pseudomonas putida grown on glycerol. Microb Cell Factories 15:73. https://doi.org/10.1186/s12934-016-0470-2
Bhokisham N, Pakhchanian H, Quan D, Tschirhart T, Tsao CY, Payne GF, Bentley WE (2016) Modular construction of multi-subunit protein complexes using engineered tags and microbial transglutaminase. Metab Eng 38:1–9. https://doi.org/10.1016/j.ymben.2016.05.004
Binder S, Schendzielorz G, Stäbler N, Krumbach K, Hoffmann K, Bott M, Eggeling L (2012) A high-throughput approach to identify genomic variants of bacterial metabolite producers at the single-cell level. Genome Biol 13(5):R40. https://doi.org/10.1186/gb-2012-13-5-r40
Binder S, Siedler S, Marienhagen J, Bott M, Eggeling L (2013) Recombineering in Corynebacterium glutamicum combined with optical nanosensors: a general strategy for fast producer strain generation. Nucleic Acids Res 41(12):6360–6369. https://doi.org/10.1093/nar/gkt312
Boyle PM, Silver PA (2012) Parts plus pipes: synthetic biology approaches to metabolic engineering. Metab Eng 14(3):223–232. https://doi.org/10.1016/j.ymben.2011.10.003
Brusseler C, Radek A, Tenhaef N, Krumbach K, Noack S, Marienhagen J (2018) The myo-inositol/proton symporter IolT1 contributes to d-xylose uptake in Corynebacterium glutamicum. Bioresour Technol 249:953–961. https://doi.org/10.1016/j.biortech.2017.10.098
Buschke N, Schroder H, Wittmann C (2011) Metabolic engineering of Corynebacterium glutamicum for production of 1,5-diaminopentane from hemicellulose. Biotechnol J 6(3):306–317. https://doi.org/10.1002/biot.201000304
Buschke N, Schafer R, Becker J, Wittmann C (2013) Metabolic engineering of industrial platform microorganisms for biorefinery applications—optimization of substrate spectrum and process robustness by rational and evolutive strategies. Bioresour Technol 135:544–554. https://doi.org/10.1016/j.biortech.2012.11.047
Carlson ED, Gan R, Hodgman CE, Jewett MC (2012) Cell-free protein synthesis: applications come of age. Biotechnol Adv 30(5):1185–1194. https://doi.org/10.1016/j.biotechadv.2011.09.016
Chen Z, Liu D (2016) Toward glycerol biorefinery: metabolic engineering for the production of biofuels and chemicals from glycerol. Biotechnol Biofuels 9:205. https://doi.org/10.1186/s13068-016-0625-8
Chen T, Zhu N, Xia H (2014) Aerobic production of succinate from arabinose by metabolically engineered Corynebacterium glutamicum. Bioresour Technol 151:411–414. https://doi.org/10.1016/j.biortech.2013.10.017
Chen X, Zhang C, Zou R, Stephanopoulos G, Too HP (2017a) In vitro metabolic engineering of amorpha-4,11-diene biosynthesis at enhanced rate and specific yield of production. ACS Synth Biol 6(9):1691–1700. https://doi.org/10.1021/acssynbio.6b00377
Chen Z, Huang J, Wu Y, Wu W, Zhang Y, Liu D (2017b) Metabolic engineering of Corynebacterium glutamicum for the production of 3-hydroxypropionic acid from glucose and xylose. Metab Eng 39:151–158. https://doi.org/10.1016/j.ymben.2016.11.009
Cheng KK, Lee BS, Masuda T, Ito T, Ikeda K, Hirayama A, Deng L, Dong J, Shimizu K, Soga T, Tomita M, Palsson BO, Robert M (2014) Global metabolic network reorganization by adaptive mutations allows fast growth of Escherichia coli on glycerol. Nat Commun 5:3233. https://doi.org/10.1038/ncomms4233
Chistoserdova L (2011) Modularity of methylotrophy, revisited. Environ Microbiol 13(10):2603–2622. https://doi.org/10.1111/j.1462-2920.2011.02464.x
Cho JS, Choi KR, Prabowo CPS, Shin JH, Yang D, Jang J, Lee SY (2017) CRISPR/Cas9-coupled recombineering for metabolic engineering of Corynebacterium glutamicum. Metab Eng 42:157–167. https://doi.org/10.1016/j.ymben.2017.06.010
Choi JW, Yim SS, Jeong KJ (2017) Development of a potential protein display platform in Corynebacterium glutamicum using mycolic acid layer protein, NCgl1337, as an anchoring motif. Biotechnol J 13(2):1700509
Cleto S, Jensen JV, Wendisch VF, Lu TK (2016) Corynebacterium glutamicum metabolic engineering with CRISPR interference (CRISPRi). ACS Synth Biol 5(5):375–385. https://doi.org/10.1021/acssynbio.5b00216
Delebecque CJ, Lindner AB, Silver PA, Aldaye FA (2011) Organization of intracellular reactions with rationally designed RNA assemblies. Science 333(6041):470–474. https://doi.org/10.1126/science.1206938
Dhar KS, Wendisch VF, Nampoothiri KM (2016) Engineering of Corynebacterium glutamicum for xylitol production from lignocellulosic pentose sugars. J Biotechnol 230:63–71. https://doi.org/10.1016/j.jbiotec.2016.05.011
Dudley QM, Karim AS, Jewett MC (2015) Cell-free metabolic engineering: biomanufacturing beyond the cell. Biotechnol J 10(1):69–82. https://doi.org/10.1002/biot.201400330
Eberhardt D, Jensen J, Wendisch V (2014) L-citrulline production by metabolically engineered Corynebacterium glutamicum from glucose and alternative carbon sources. AMB Express 4(1):85. https://doi.org/10.1186/s13568-014-0085-0
Fernandez-Castane A, Feher T, Carbonell P, Pauthenier C, Faulon JL (2014) Computer-aided design for metabolic engineering. J Biotechnol 192(Pt B):302–313. https://doi.org/10.1016/j.jbiotec.2014.03.029
Fu J, Yang YR, Johnson-Buck A, Liu M, Liu Y, Walter NG, Woodbury NW, Yan H (2014) Multi-enzyme complexes on DNA scaffolds capable of substrate channelling with an artificial swinging arm. Nat Nanotechnol 9(7):531–536. https://doi.org/10.1038/nnano.2014.100
Gao C, Yang X, Wang H, Rivero CP, Li C, Cui Z, Qi Q, Lin CSK (2016) Robust succinic acid production from crude glycerol using engineered Yarrowia lipolytica. Biotechnol Biofuels 9(1):179. https://doi.org/10.1186/s13068-016-0597-8
Georgi T, Rittmann D, Wendisch VF (2005) Lysine and glutamate production by Corynebacterium glutamicum on glucose, fructose and sucrose: roles of malic enzyme and fructose-1,6-bisphosphatase. Metab Eng 7(4):291–301. https://doi.org/10.1016/j.ymben.2005.05.001
Gopinath V, Murali A, Dhar KS, Nampoothiri KM (2012) Corynebacterium glutamicum as a potent biocatalyst for the bioconversion of pentose sugars to value-added products. Appl Microbiol Biotechnol 93(1):95–106. https://doi.org/10.1007/s00253-011-3686-4
Gunji Y, Yasueda H (2006) Enhancement of L-lysine production in methylotroph Methylophilus methylotrophus by introducing a mutant LysE exporter. J Biotechnol 127(1):1–13. https://doi.org/10.1016/j.jbiotec.2006.06.003
Heider SA, Wolf N, Hofemeier A, Peters-Wendisch P, Wendisch VF (2014a) Optimization of the IPP precursor supply for the production of lycopene, decaprenoxanthin and astaxanthin by Corynebacterium glutamicum. Front Bioeng Biotechnol 2:28. https://doi.org/10.3389/fbioe.2014.00028
Heider SAE, Peters-Wendisch P, Netzer R, Stafnes M, Brautaset T, Wendisch VF (2014b) Production and glucosylation of C 50 and C 40 carotenoids by metabolically engineered Corynebacterium glutamicum. Appl Microbiol Biotechnol 98(3):1223–1235. https://doi.org/10.1007/s00253-013-5359-y
Henke NA, Peters-Wendisch P, Wendisch VF (2017) Carotenoid production by Corynebacterium: the workhorse of industrial amino acid production as host for production of a broad spectrum of C40 and C50 carotenoids. Intech, Rijeka, pp 159–173. https://doi.org/10.5772/67631
Hirakawa H, Haga T, Nagamune T (2012) Artificial protein complexes for biocatalysis. Top Catal 55(16–18):1124–1137. https://doi.org/10.1007/s11244-012-9900-5
Hirakawa H, Kakitani A, Nagamune T (2013) Introduction of selective intersubunit disulfide bonds into self-assembly protein scaffold to enhance an artificial multienzyme complex’s activity. Biotechnol Bioeng 110(7):1858–1864. https://doi.org/10.1002/bit.24861
Huang Y, Li L, Xie S, Zhao N, Han S, Lin Y, Zheng S (2017) Recombineering using RecET in Corynebacterium glutamicum ATCC14067 via a self-excisable cassette. Sci Rep 7(1):7916. https://doi.org/10.1038/s41598-017-08352-9
Hughes RA, Ellington AD (2017) Synthetic DNA synthesis and assembly: putting the synthetic in synthetic biology. Cold Spring Harb Perspect Biol 9(1). https://doi.org/10.1101/cshperspect.a023812
Irla M, Nærdal I, Brautaset T, Wendisch VF (2017) Methanol-based γ-aminobutyric acid (GABA) production by genetically engineered Bacillus methanolicus strains. Ind Crop Prod 106:12–20. https://doi.org/10.1016/j.indcrop.2016.11.050
Jensen J, Wendisch V (2013) Ornithine cyclodeaminase-based proline production by Corynebacterium glutamicum. Microb Cell Factories 12:63. https://doi.org/10.1186/1475-2859-12-63
Jiang Y, Qian F, Yang J, Liu Y, Dong F, Xu C, Sun B, Chen B, Xu X, Li Y, Wang R, Yang S (2017) CRISPR-Cpf1 assisted genome editing of Corynebacterium glutamicum. Nat Commun 8:15179. https://doi.org/10.1038/ncomms15179
Jo S, Yoon J, Lee SM, Um Y, Han SO, Woo HM (2017) Modular pathway engineering of Corynebacterium glutamicum to improve xylose utilization and succinate production. J Biotechnol 258:69–78. https://doi.org/10.1016/j.jbiotec.2017.01.015
Jorge JM, Nguyen AQ, Perez-Garcia F, Kind S, Wendisch VF (2017a) Improved fermentative production of gamma-aminobutyric acid via the putrescine route: systems metabolic engineering for production from glucose, amino sugars, and xylose. Biotechnol Bioeng 114(4):862–873. https://doi.org/10.1002/bit.26211
Jorge JMP, Perez-Garcia F, Wendisch VF (2017b) A new metabolic route for the fermentative production of 5-aminovalerate from glucose and alternative carbon sources. Bioresour Technol 245(Pt B):1701–1709. https://doi.org/10.1016/j.biortech.2017.04.108
Juhas M, Ajioka JW (2017) High molecular weight DNA assembly in vivo for synthetic biology applications. Crit Rev Biotechnol 37(3):277–286. https://doi.org/10.3109/07388551.2016.1141394
Jullesson D, David F, Pfleger B, Nielsen J (2015) Impact of synthetic biology and metabolic engineering on industrial production of fine chemicals. Biotechnol Adv 33(7):1395–1402. https://doi.org/10.1016/j.biotechadv.2015.02.011
Kalinowski J, Bathe B, Bartels D, Bischoff N, Bott M, Burkovski A, Dusch N, Eggeling L, Eikmanns BJ, Gaigalat L, Goesmann A, Hartmann M, Huthmacher K, Krämer R, Linke B, McHardy AC, Meyer F, Möckel B, Pfefferle W, Pühler A, Rey DA, Rückert C, Rupp O, Sahm H, Wendisch VF, Wiegräbe I, Tauch A (2003) The complete Corynebacterium glutamicum ATCC 13032 genome sequence and its impact on the production of l-aspartate-derived amino acids and vitamins. J Biotechnol 104(1–3):5–25. https://doi.org/10.1016/s0168-1656(03)00154-8
Kallscheuer N, Vogt M, Kappelmann J, Krumbach K, Noack S, Bott M, Marienhagen J (2016a) Identification of the phd gene cluster responsible for phenylpropanoid utilization in Corynebacterium glutamicum. Appl Microbiol Biotechnol 100(4):1871–1881. https://doi.org/10.1007/s00253-015-7165-1
Kallscheuer N, Vogt M, Stenzel A, Gatgens J, Bott M, Marienhagen J (2016b) Construction of a Corynebacterium glutamicum platform strain for the production of stilbenes and (2S)-flavanones. Metab Eng 38:47–55. https://doi.org/10.1016/j.ymben.2016.06.003
Kallscheuer N, Vogt M, Marienhagen J (2017) A novel synthetic pathway enables microbial production of polyphenols independent from the endogenous aromatic amino acid metabolism. ACS Synth Biol 6(3):410–415. https://doi.org/10.1021/acssynbio.6b00291
Kang MK, Lee J, Um Y, Lee TS, Bott M, Park SJ, Woo HM (2014) Synthetic biology platform of CoryneBrick vectors for gene expression in Corynebacterium glutamicum and its application to xylose utilization. Appl Microbiol Biotechnol 98(13):5991–6002. https://doi.org/10.1007/s00253-014-5714-7
Kato N, Yurimoto H, Thauer RK (2006) The physiological role of the ribulose monophosphate pathway in bacteria and archaea. Biosci Biotechnol Biochem 70(1):10–21. https://doi.org/10.1271/bbb.70.10
Kawaguchi H, Vertes AA, Okino S, Inui M, Yukawa H (2006) Engineering of a xylose metabolic pathway in Corynebacterium glutamicum. Appl Environ Microbiol 72(5):3418–3428. https://doi.org/10.1128/AEM.72.5.3418-3428.2006
Kawaguchi H, Sasaki M, Vertes AA, Inui M, Yukawa H (2008) Engineering of an L-arabinose metabolic pathway in Corynebacterium glutamicum. Appl Microbiol Biotechnol 77(5):1053–1062. https://doi.org/10.1007/s00253-007-1244-x
Kawaguchi H, Sasaki M, Vertes AA, Inui M, Yukawa H (2009) Identification and functional analysis of the gene cluster for L-arabinose utilization in Corynebacterium glutamicum. Appl Environ Microbiol 75(11):3419–3429. https://doi.org/10.1128/AEM.02912-08
Keasling JD (2012) Synthetic biology and the development of tools for metabolic engineering. Metab Eng 14(3):189–195. https://doi.org/10.1016/j.ymben.2012.01.004
Kiefer P, Heinzle E, Wittmann C (2002) Influence of glucose, fructose and sucrose as carbon sources on kinetics and stoichiometry of lysine production by Corynebacterium glutamicum. J Ind Microbiol Biotechnol 28(6):338–343. https://doi.org/10.1038/sj/jim/7000252
Kim J, Hirasawa T, Sato Y, Nagahisa K, Furusawa C, Shimizu H (2009) Effect of odhA overexpression and odhA antisense RNA expression on Tween-40-triggered glutamate production by Corynebacterium glutamicum. Appl Microbiol Biotechnol 81(6):1097–1106. https://doi.org/10.1007/s00253-008-1743-4
Kim SJ, Hyeon JE, Jeon SD, Choi GW, Han SO (2014) Bi-functional cellulases complexes displayed on the cell surface of Corynebacterium glutamicum increase hydrolysis of lignocelluloses at elevated temperature. Enzym Microb Technol 66:67–73. https://doi.org/10.1016/j.enzmictec.2014.08.010
Kinoshita S, Udaka S, Shimono M (1957) Studies on the amino acid fermentation. Part 1. Production of L-glutamic acid by various microorganisms. J Gen Appl Microbiol 50(6):331–343. https://doi.org/10.2323/jgam.3.193
Kotrbova-Kozak A, Kotrba P, Inui M, Sajdok J, Yukawa H (2007) Transcriptionally regulated adhA gene encodes alcohol dehydrogenase required for ethanol and n-propanol utilization in Corynebacterium glutamicum R. Appl Microbiol Biotechnol 76(6):1347–1356. https://doi.org/10.1007/s00253-007-1094-6
Krutsakorn B, Honda K, Ye X, Imagawa T, Bei X, Okano K, Ohtake H (2013) In vitro production of n-butanol from glucose. Metab Eng 20:84–91. https://doi.org/10.1016/j.ymben.2013.09.006
Kurosawa K, Radek A, Plassmeier JK, Sinskey AJ (2015) Improved glycerol utilization by a triacylglycerol-producing Rhodococcus opacus strain for renewable fuels. Bioresour Technol 8:31. https://doi.org/10.1186/s13068-015-0209-z
Lange J, Muller F, Takors R, Blombach B (2018) Harnessing novel chromosomal integration loci to utilize an organosolv-derived hemicellulose fraction for isobutanol production with engineered Corynebacterium glutamicum. Microb Biotechnol 11(1):257–263. https://doi.org/10.1111/1751-7915.12879
Lee SY, Kim HU (2015) Systems strategies for developing industrial microbial strains. Nat Biotechnol 33(10):1061–1072. https://doi.org/10.1038/nbt.3365
Lee SY, Le TH, Chang ST, Park JS, Kim YH, Min J (2010) Utilization of phenol and naphthalene affects synthesis of various amino acids in Corynebacterium glutamicum. Curr Microbiol 61(6):596–600. https://doi.org/10.1007/s00284-010-9658-6
Lee J, Saddler JN, Um Y, Woo HM (2016) Adaptive evolution and metabolic engineering of a cellobiose- and xylose- negative Corynebacterium glutamicum that co-utilizes cellobiose and xylose. Microb Cell Factories 15(1):20. https://doi.org/10.1186/s12934-016-0420-z
Lessmeier L, Wendisch VF (2015) Identification of two mutations increasing the methanol tolerance of Corynebacterium glutamicum. BMC Microbiol 15:216. https://doi.org/10.1186/s12866-015-0558-6
Lessmeier L, Pfeifenschneider J, Carnicer M, Heux S, Portais JC, Wendisch VF (2015) Production of carbon-13-labeled cadaverine by engineered Corynebacterium glutamicum using carbon-13-labeled methanol as co-substrate. Appl Microbiol Biotechnol 99(23):10163–10176. https://doi.org/10.1007/s00253-015-6906-5
Li P, Modica J, Howarth A, Vargasl E, Moghadam P, Snurr R, Mrksich M, Hupp J, Farha O (2016a) Toward design rules for enzyme immobilization in hierarchical mesoporous metal-organic frameworks. Chem 1(1):154–169. https://doi.org/10.1016/j.chempr.2016.05.001
Li Q, Wu H, Li Z, Ye Q (2016b) Enhanced succinate production from glycerol by engineered Escherichia coli strains. Bioresour Technol 218:217–223. https://doi.org/10.1016/j.biortech.2016.06.090
Li P, Chen Q, Wang TC, Vermeulen NA, Mehdi BL, Dohnalkova A, Browning ND, Shen D, Anderson R, Gómez-Gualdrón DA, Cetin FM, Jagiello J, Asiri AM, Stoddart JF, Farha OK (2018a) Hierarchically engineered mesoporous metal-organic frameworks toward cell-free immobilized enzyme systems. Chem 4(5):1022–1034. https://doi.org/10.1016/j.chempr.2018.03.001
Li R, Wijma HJ, Song L, Cui Y, Otzen M, Tian Y, Du J, Li T, Niu D, Chen Y, Feng J, Han J, Chen H, Tao Y, Janssen DB, Wu B (2018b) Computational redesign of enzymes for regio- and enantioselective hydroamination. Nat Chem Biol 14:664–670. https://doi.org/10.1038/s41589-018-0053-0
Lindner SN, Seibold GM, Henrich A, Kramer R, Wendisch VF (2011) Phosphotransferase system-independent glucose utilization in Corynebacterium glutamicum by inositol permeases and glucokinases. Appl Environ Microbiol 77(11):3571–3581. https://doi.org/10.1128/AEM.02713-10
Litsanov B, Brocker M, Bott M (2013) Glycerol as a substrate for aerobic succinate production in minimal medium with Corynebacterium glutamicum. Microb Biotechnol 6(2):189–195. https://doi.org/10.1111/j.1751-7915.2012.00347.x
Liu X, Zhang W, Zhao Z, Dai X, Yang Y, Bai Z (2017) Protein secretion in Corynebacterium glutamicum. Crit Rev Biotechnol 37(4):541–551. https://doi.org/10.1080/07388551.2016.1206059
Lv Y, Wu Z, Han S, Lin Y, Zheng S (2011) Genome sequence of Corynebacterium glutamicum S9114, a strain for industrial production of glutamate. J Bacteriol 193(21):6096–6097. https://doi.org/10.1128/JB.06074-11
Lv Y, Liao J, Wu Z, Han S, Lin Y, Zheng S (2012) Genome sequence of Corynebacterium glutamicum ATCC 14067, which provides insight into amino acid biosynthesis in coryneform bacteria. J Bacteriol 194(3):742–743. https://doi.org/10.1128/JB.06514-11
Matano C, Uhde A, Youn JW, Maeda T, Clermont L, Marin K, Krämer R, Wendisch VF, Seibold GM (2014) Engineering of Corynebacterium glutamicum for growth and l-lysine and lycopene production from N-acetyl-glucosamine. Appl Microbiol Biotechnol 98(12):5633–5643. https://doi.org/10.1007/s00253-014-5676-9
Matano C, Kolkenbrock S, Hamer SN, Sgobba E, Moerschbacher BM, Wendisch VF (2016) Corynebacterium glutamicum possesses beta-N-acetylglucosaminidase. BMC Microbiol 16(1):177. https://doi.org/10.1186/s12866-016-0795-3
Meiswinkel TM, Gopinath V, Lindner SN, Nampoothiri KM, Wendisch VF (2013a) Accelerated pentose utilization by Corynebacterium glutamicum for accelerated production of lysine, glutamate, ornithine and putrescine. Microb Biotechnol 6(2):131–140. https://doi.org/10.1111/1751-7915.12001
Meiswinkel TM, Rittmann D, Lindner SN, Wendisch VF (2013b) Crude glycerol-based production of amino acids and putrescine by Corynebacterium glutamicum. Bioresour Technol 145:254–258. https://doi.org/10.1016/j.biortech.2013.02.053
Meyer F, Keller P, Hartl J, Groninger OG, Kiefer P, Vorholt JA (2018) Methanol-essential growth of Escherichia coli. Nat Commun 9(1):1508. https://doi.org/10.1038/s41467-018-03937-y
Muller JE, Heggeset TM, Wendisch VF, Vorholt JA, Brautaset T (2015a) Methylotrophy in the thermophilic Bacillus methanolicus, basic insights and application for commodity production from methanol. Appl Microbiol Biotechnol 99(2):535–551. https://doi.org/10.1007/s00253-014-6224-3
Muller JE, Meyer F, Litsanov B, Kiefer P, Potthoff E, Heux S, Quax WJ, Wendisch VF, Brautaset T, Portais JC, Vorholt JA (2015b) Engineering Escherichia coli for methanol conversion. Metab Eng 28:190–201. https://doi.org/10.1016/j.ymben.2014.12.008
Nguyen TA, Kim KR, Han SJ, Cho HY, Kim JW, Park SM, Park JC, Sim SJ (2010) Pretreatment of rice straw with ammonia and ionic liquid for lignocellulose conversion to fermentable sugars. Bioresour Technol 101(19):7432–7438. https://doi.org/10.1016/j.biortech.2010.04.053
Nguyen AQ, Schneider J, Reddy GK, Wendisch VF (2015) Fermentative production of the diamine putrescine: system metabolic engineering of Corynebacterium glutamicum. Metabolites 5(2):211–231. https://doi.org/10.3390/metabo5020211
Okai N, Masuda T, Takeshima Y, Tanaka K, Yoshida KI, Miyamoto M, Ogino C, Kondo A (2017) Biotransformation of ferulic acid to protocatechuic acid by Corynebacterium glutamicum ATCC 21420 engineered to express vanillate O-demethylase. AMB Express 7(1):130. https://doi.org/10.1186/s13568-017-0427-9
Perez-Garcia F, Max Risse J, Friehs K, Wendisch VF (2017a) Fermentative production of L-pipecolic acid from glucose and alternative carbon sources. Biotechnol J 12(7). https://doi.org/10.1002/biot.201600646
Perez-Garcia F, Ziert C, Risse JM, Wendisch VF (2017b) Improved fermentative production of the compatible solute ectoine by Corynebacterium glutamicum from glucose and alternative carbon sources. J Biotechnol 258:59–68. https://doi.org/10.1016/j.jbiotec.2017.04.039
Peters RJ, Marguet M, Marais S, Fraaije MW, van Hest JC, Lecommandoux S (2014) Cascade reactions in multicompartmentalized polymersomes. Angew Chem 53(1):146–150. https://doi.org/10.1002/anie.201308141
Pfeifenschneider J, Brautaset T, Wendisch VF (2017) Methanol as carbon substrate in the bio-economy: metabolic engineering of aerobic methylotrophic bacteria for production of value-added chemicals. Biofuels Bioprod Biorefin 11(4):719–731. https://doi.org/10.1002/bbb.1773
Qi H, Du Y, Zhou X, Zheng W, Zhang L, Wen J, Liu L (2017) Engineering a new metabolic pathway for itaconate production in Pichia stipitis from xylose. Biochem Eng J 126:101–108. https://doi.org/10.1016/j.bej.2017.06.011
Radek A, Krumbach K, Gatgens J, Wendisch VF, Wiechert W, Bott M, Noack S, Marienhagen J (2014) Engineering of Corynebacterium glutamicum for minimized carbon loss during utilization of D-xylose containing substrates. J Biotechnol 192(Pt A):156–160. https://doi.org/10.1016/j.jbiotec.2014.09.026
Radek A, Muller MF, Gatgens J, Eggeling L, Krumbach K, Marienhagen J, Noack S (2016) Formation of xylitol and xylitol-5-phosphate and its impact on growth of d-xylose-utilizing Corynebacterium glutamicum strains. J Biotechnol 231:160–166. https://doi.org/10.1016/j.jbiotec.2016.06.009
Radek A, Tenhaef N, Muller MF, Brusseler C, Wiechert W, Marienhagen J, Polen T, Noack S (2017) Miniaturized and automated adaptive laboratory evolution: evolving Corynebacterium glutamicum towards an improved d-xylose utilization. Bioresour Technol 245(Pt B):1377–1385. https://doi.org/10.1016/j.biortech.2017.05.055
Ravasi P, Peiru S, Gramajo H, Menzella HG (2012) Design and testing of a synthetic biology framework for genetic engineering of Corynebacterium glutamicum. Microb Cell Factories 11(1):147–147. https://doi.org/10.1186/1475-2859-11-147
Rittmann D, Lindner SN, Wendisch VF (2008) Engineering of a glycerol utilization pathway for amino acid production by Corynebacterium glutamicum. Appl Environ Microbiol 74(20):6216–6222. https://doi.org/10.1128/AEM.00963-08
Rizk M, Antranikian G, Elleuche S (2012) End-to-end gene fusions and their impact on the production of multifunctional biomass degrading enzymes. Biochem Biophys Res Commun 428(1):1–5. https://doi.org/10.1016/j.bbrc.2012.09.142
Rollin JA, Martin del Campo J, Myung S, Sun F, You C, Bakovic A, Castro R, Chandrayan SK, Wu C-H, Adams MWW, Senger RS, Zhang Y-HP (2015) High-yield hydrogen production from biomass by in vitro metabolic engineering: mixed sugars coutilization and kinetic modeling. Proc Natl Acad Sci U S A 112(16):4964–4969. https://doi.org/10.1073/pnas.1417719112
Saha BC (2003) Hemicellulose bioconversion. J Ind Microbiol Biotechnol 30(5):279–291. https://doi.org/10.1007/s10295-003-0049-x
Sasaki M, Jojima T, Kawaguchi H, Inui M, Yukawa H (2009) Engineering of pentose transport in Corynebacterium glutamicum to improve simultaneous utilization of mixed sugars. Appl Microbiol Biotechnol 85(1):105–115. https://doi.org/10.1007/s00253-009-2065-x
Schneider J, Wendisch VF (2010) Putrescine production by engineered Corynebacterium glutamicum. Appl Microbiol Biotechnol 88(4):859–868. https://doi.org/10.1007/s00253-010-2778-x
Schneider J, Niermann K, Wendisch VF (2011) Production of the amino acids l-glutamate, l-lysine, l-ornithine and l-arginine from arabinose by recombinant Corynebacterium glutamicum. J Biotechnol 154(2–3):191–198. https://doi.org/10.1016/j.jbiotec.2010.07.009
Schneider J, Eberhardt D, Wendisch VF (2012) Improving putrescine production by Corynebacterium glutamicum by fine-tuning ornithine transcarbamoylase activity using a plasmid addiction system. Appl Microbiol Biotechnol 95(1):169–178. https://doi.org/10.1007/s00253-012-3956-9
Schrader J, Schilling M, Holtmann D, Sell D, Filho MV, Marx A, Vorholt JA (2009) Methanol-based industrial biotechnology: current status and future perspectives of methylotrophic bacteria. Trends Biotechnol 27(2):107–115. https://doi.org/10.1016/j.tibtech.2008.10.009
Segler MHS, Preuss M, Waller MP (2018) Planning chemical syntheses with deep neural networks and symbolic AI. Nature 555(7698):604–610. https://doi.org/10.1038/nature25978
Seibold G, Auchter M, Berens S, Kalinowski J, Eikmanns BJ (2006) Utilization of soluble starch by a recombinant Corynebacterium glutamicum strain: growth and lysine production. J Biotechnol 124(2):381–391. https://doi.org/10.1016/j.jbiotec.2005.12.027
Shen XH, Zhou NY, Liu SJ (2012) Degradation and assimilation of aromatic compounds by Corynebacterium glutamicum: another potential for applications for this bacterium? Appl Microbiol Biotechnol 95(1):77–89. https://doi.org/10.1007/s00253-012-4139-4
Smanski MJ, Zhou H, Claesen J, Shen B, Fischbach MA, Voigt CA (2016) Synthetic biology to access and expand nature’s chemical diversity. Nat Rev Microbiol 14(3):135–149. https://doi.org/10.1038/nrmicro.2015.24
Su H, Lin J, Wang G (2016) Metabolic engineering of Corynebacterium crenatium for enhancing production of higher alcohols. Sci Rep 6:39543. https://doi.org/10.1038/srep39543
Tateno T, Fukuda H, Kondo A (2007) Production of L-lysine from starch by Corynebacterium glutamicum displaying alpha-amylase on its cell surface. Appl Microbiol Biotechnol 74(6):1213–1220. https://doi.org/10.1007/s00253-006-0766-y
Tateno T, Hatada K, Tanaka T, Fukuda H, Kondo A (2009) Development of novel cell surface display in Corynebacterium glutamicum using porin. Appl Microbiol Biotechnol 84(4):733–739. https://doi.org/10.1007/s00253-009-2021-9
Tauch A, Homann I, Mormann S, Rüberg S, Billault A, Bathe B, Brand S, Brockmanngretza O, Rückert C, Schischka N (2002) Strategy to sequence the genome of Corynebacterium glutamicum ATCC 13032: use of a cosmid and a bacterial artificial chromosome library. J Biotechnol 95(1):25–38. https://doi.org/10.1016/S0168-1656(01)00443-6
Teramoto H, Inui M, Yukawa H (2009) Regulation of expression of genes involved in quinate and shikimate utilization in Corynebacterium glutamicum. Appl Environ Microbiol 75(11):3461–3468. https://doi.org/10.1128/AEM.00163-09
Uhde A, Youn JW, Maeda T, Clermont L, Matano C, Krämer R, Wendisch VF, Seibold GM, Marin K (2013) Glucosamine as carbon source for amino acid-producing Corynebacterium glutamicum. Appl Microbiol Biotechnol 97(4):1679–1687. https://doi.org/10.1007/s00253-012-4313-8
van Dongen SF, Nallani M, Cornelissen JJ, Nolte RJ, van Hest JC (2009) A three-enzyme cascade reaction through positional assembly of enzymes in a polymersome nanoreactor. Chemistry 15(5):1107–1114. https://doi.org/10.1002/chem.200802114
Verhoeven MD, Lee M, Kamoen L, van den Broek M, Janssen DB, Daran JG, van Maris AJ, Pronk JT (2017) Mutations in PMR1 stimulate xylose isomerase activity and anaerobic growth on xylose of engineered Saccharomyces cerevisiae by influencing manganese homeostasis. Sci Rep 7:46155. https://doi.org/10.1038/srep46155
Wang Y, Zhang Y, Jiang T, Meng J, Sheng B, Yang C, Gao C, Xu P, Ma C (2015) A novel biocatalyst for efficient production of 2-oxo-carboxylates using glycerol as the cost-effective carbon source. Biotechnol Biofuels 8:186. https://doi.org/10.1186/s13068-015-0368-y
Wang Z, Wu J, Gao MJ, Zhu L, Zhan XB (2017) High production of xanthan gum by a glycerol-tolerant strain Xanthomonas campestris WXLB-006. Prep Biochem Biotechnol 47(5):468–472. https://doi.org/10.1080/10826068.2017.1292288
Wang Y, Liu Y, Liu J, Guo Y, Fan L, Ni X, Zheng X, Wang M, Zheng P, Sun J, Ma Y (2018) MACBETH: multiplex automated Corynebacterium glutamicum base editing method. Metab Eng 47:200–210. https://doi.org/10.1016/j.ymben.2018.02.016
Wendisch VF, Jorge JMP, Perez-Garcia F, Sgobba E (2016) Updates on industrial production of amino acids using Corynebacterium glutamicum. World J Microbiol Biotechnol 32(6):105. https://doi.org/10.1007/s11274-016-2060-1
Wheeldon I, Minteer SD, Banta S, Barton SC, Atanassov P, Sigman M (2016) Substrate channelling as an approach to cascade reactions. Nat Chem 8(4):299–309. https://doi.org/10.1038/nchem.2459
Whitaker WB, Sandoval NR, Bennett RK, Fast AG, Papoutsakis ET (2015) Synthetic methylotrophy: engineering the production of biofuels and chemicals based on the biology of aerobic methanol utilization. Curr Opin Biotechnol 33:165–175. https://doi.org/10.1016/j.copbio.2015.01.007
Whitaker WB, Jones JA, Bennett RK, Gonzalez JE, Vernacchio VR, Collins SM, Palmer MA, Schmidt S, Antoniewicz MR, Koffas MA, Papoutsakis ET (2017) Engineering the biological conversion of methanol to specialty chemicals in Escherichia coli. Metab Eng 39:49–59. https://doi.org/10.1016/j.ymben.2016.10.015
Wisselink HW, Toirkens MJ, del Rosario Franco Berriel M, Winkler AA, van Dijken JP, Pronk JT, van Maris AJ (2007) Engineering of Saccharomyces cerevisiae for efficient anaerobic alcoholic fermentation of L-arabinose. Appl Environ Microbiol 73(15):4881–4891. https://doi.org/10.1128/AEM.00177-07
Witthoff S, Mühlroth A, Marienhagen J, Bott M (2013) C1 metabolism in Corynebacterium glutamicum: an endogenous pathway for oxidation of methanol to carbon dioxide. Appl Environ Microbiol 79(22):6974–6983. https://doi.org/10.1128/AEM.02705-13
Witthoff S, Schmitz K, Niedenfuhr S, Noh K, Noack S, Bott M, Marienhagen J (2015) Metabolic engineering of Corynebacterium glutamicum for methanol metabolism. Appl Environ Microbiol 81(6):2215–2225. https://doi.org/10.1128/AEM.03110-14
Woo HM, Park JB (2014) Recent progress in development of synthetic biology platforms and metabolic engineering of Corynebacterium glutamicum. J Biotechnol 180:43–51. https://doi.org/10.1016/j.jbiotec.2014.03.003
Wu G, Yan Q, Jones JA, Tang YJ, Fong SS, Koffas MAG (2016) Metabolic burden: cornerstones in synthetic biology and metabolic engineering applications. Trends Biotechnol 34(8):652–664. https://doi.org/10.1016/j.tibtech.2016.02.010
Xu P, Rizzoni EA, Sul SY, Stephanopoulos G (2017) Improving metabolic pathway efficiency by statistical model-based multivariate regulatory metabolic engineering. ACS Synth Biol 6(1):148–158. https://doi.org/10.1021/acssynbio.6b00187
Yang J, Zhu Y, Men Y, Sun S, Zeng Y, Zhang Y, Sun Y, Ma Y (2016) Pathway construction in Corynebacterium glutamicum and strain engineering to produce rare sugars from glycerol. J Agric Food Chem 64(50):9497–9505. https://doi.org/10.1021/acs.jafc.6b03423
Yao W, Chu C, Deng X, Zhang Y, Liu M, Zheng P, Sun Z (2009) Display of alpha-amylase on the surface of Corynebacterium glutamicum cells by using NCgl1221 as the anchoring protein, and production of glutamate from starch. Arch Microbiol 191(10):751–759. https://doi.org/10.1007/s00203-009-0506-7
Yim SS, An SJ, Kang M, Lee J, Jeong KJ (2013) Isolation of fully synthetic promoters for high-level gene expression in Corynebacterium glutamicum. Biotechnol Bioeng 110(11):2959–2969. https://doi.org/10.1002/bit.24954
Yim SS, Choi JW, Lee SH, Jeong KJ (2016) Modular optimization of a hemicellulose-utilizing pathway in Corynebacterium glutamicum for consolidated bioprocessing of hemicellulosic biomass. ACS Synth Biol 5(4):334–343. https://doi.org/10.1021/acssynbio.5b00228
Yim SS, Choi JW, Lee SH, Jeon EJ, Chung WJ, Jeong KJ (2017) Engineering of Corynebacterium glutamicum for consolidated conversion of hemicellulosic biomass into xylonic acid. Biotechnol J 12(11). https://doi.org/10.1002/biot.201700040
You C, Zhang YH (2013) Self-assembly of synthetic metabolons through synthetic protein scaffolds: one-step purification, co-immobilization, and substrate channeling. ACS Synth Biol 2(2):102–110. https://doi.org/10.1021/sb300068g
Yukawa H, Omumasaba CA, Nonaka H, Kos P, Okai N, Suzuki N, Suda M, Tsuge Y, Watanabe J, Ikeda Y, Vertes AA, Inui M (2007) Comparative analysis of the Corynebacterium glutamicum group and complete genome sequence of strain R. Microbiology 153(Pt 4):1042–1058. https://doi.org/10.1099/mic.0.2006/003657-0
Zhang B, Zhou N, Liu YM, Liu C, Lou CB, Jiang CY, Liu SJ (2015a) Ribosome binding site libraries and pathway modules for shikimic acid synthesis with Corynebacterium glutamicum. Microb Cell Factories 14:71. https://doi.org/10.1186/s12934-015-0254-0
Zhang Y, Bu Y, Liu J (2015b) Production of L-ornithine from sucrose and molasses by recombinant Corynebacterium glutamicum. Folia Microbiol 60(5):393–398. https://doi.org/10.1007/s12223-014-0371-x
Zhao A, Hu X, Wang X (2017) Metabolic engineering of Escherichia coli to produce gamma-aminobutyric acid using xylose. Appl Microbiol Biotechnol 101(9):3587–3603. https://doi.org/10.1007/s00253-017-8162-3
Zhou S, Ding R, Chen J, Du G, Li H, Zhou J (2017) Obtaining a panel of cascade promoter-5'-UTR complexes in Escherichia coli. ACS Synth Biol 6(6):1065–1075. https://doi.org/10.1021/acssynbio.7b00006
Zhu Z, Zhang YP (2017) In vitro metabolic engineering of bioelectricity generation by the complete oxidation of glucose. Metab Eng 39:110–116. https://doi.org/10.1016/j.ymben.2016.11.002
Zhu H, Yi X, Liu Y, Hu H, Wood TK, Zhang X (2013) Production of acetol from glycerol using engineered Escherichia coli. Bioresour Technol 149:238–243. https://doi.org/10.1016/j.biortech.2013.09.062
Zhu Z, Kin Tam T, Sun F, You C, Percival Zhang YH (2014) A high-energy-density sugar biobattery based on a synthetic enzymatic pathway. Nat Commun 5:3026. https://doi.org/10.1038/ncomms4026
Funding
All the authors are thankful for the financial support of the Science and Technology Planning Project of Guangzhou City (No. 201607010307), and the National Natural Science Foundation of China (No. 31671840) to Suiping Zheng.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Rights and permissions
About this article
Cite this article
Zhao, N., Qian, L., Luo, G. et al. Synthetic biology approaches to access renewable carbon source utilization in Corynebacterium glutamicum. Appl Microbiol Biotechnol 102, 9517–9529 (2018). https://doi.org/10.1007/s00253-018-9358-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-018-9358-x