Abstract
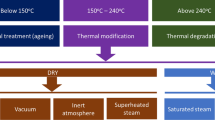
This work investigated the relationship between freeze-drying and supercritical drying of cellulosic fibers with different moisture contents based on pore and crystallinity measurements. The results showed that freeze-drying reduced the specific surface area and pore volume of cellulosic fibers by 20–160-fold, compared to supercritical drying. At the same time, freeze-dried fibers had a higher crystallinity index and crystal size than supercritically dried fibers. Freeze-drying also significantly changed the nitrogen adsorption–desorption isotherm and pore size distribution of cellulosic fibers. However, most importantly, freeze-dried fibers had linear positive correlations with supercritically dried fibers in terms of pore parameters and crystallinity, indicating that freeze-dried samples retained the trends and qualitative relationships of supercritically dried fibers. Therefore, freeze-drying can be used as the pretreatment procedure for pore and crystallinity measurements of cellulosic fibers from poplar and eucalyptus when comparing the effects of thermal drying. This work also deepens the understanding of the drying of cellulosic fibers.







Similar content being viewed by others
References
Alvi SB, Jogdand A, Rengan AK (2021) Cellulose nanosystems from synthesis to applications. Springer, Cham, pp 1–33
Amidon TE, Liu S (2009) Water-based woody biorefinery. Biotechnol Adv 27(5):542–550. https://doi.org/10.1016/j.biotechadv.2009.04.012
Baudron V, Gurikov P, Smirnova I, Whitehouse S (2019) Porous Starch Materials via Supercritical- and Freeze-Drying. Gels 5(1):12. https://doi.org/10.3390/gels5010012
Bhuiyan MTR, Hirai N, Sobue N (2000) Changes of crystallinity in wood cellulose by heat treatment under dried and moist conditions. J Wood Sci 46(6):431–436. https://doi.org/10.1007/bf00765800
Cabeza LF, de Gracia A, Fernández AI, Farid MM (2017) Supercritical CO2 as heat transfer fluid: a review. Appl Therm Eng 125:799–810. https://doi.org/10.1016/j.applthermaleng.2017.07.049
Chen Y, Wan J, Huang M, Ma Y, Wang Y, Lv H, Yang J (2011) Influence of drying temperature and duration on fiber properties of unbleached wheat straw pulp. Carbohyd Polym 85(4):759–764. https://doi.org/10.1016/j.carbpol.2011.03.041
Chunilall V, Bush T, Larsson PT, Iversen T, Kindness A (2010) A CP/MAS 13C-NMR study of cellulose fibril aggregation in eucalyptus dissolving pulps during drying and the correlation between aggregate dimensions and chemical reactivity. Holzforschung 64(6):693–698. https://doi.org/10.1515/hf.2010.097
Diniz JF, Gil MH, Castro J (2004) Hornification—its origin and interpretation in wood pulps. Wood Sci Technol 37(6):489–494
Endo T, Aung EM, Fujii S, Hosomi S, Kimizu M, Ninomiya K, Takahashi K (2017) Investigation of accessibility and reactivity of cellulose pretreated by ionic liquid at high loading. Carbohyd Polym 176:365–373. https://doi.org/10.1016/j.carbpol.2017.08.105
Esteghlalian AR, Bilodeau M, Mansfield SD, Saddler JN (2001) Do enzymatic hydrolyzability and Simons’ stain reflect the changes in the accessibility of lignocellulosic substrates to cellulase enzymes? Biotechnol Progr 17(6):1049–1054. https://doi.org/10.1021/bp0101177
Fahlén J, Salmén L (2005) Pore and matrix distribution in the fiber wall revealed by atomic force microscopy and image analysis. Biomacromolecules 6(1):433–438. https://doi.org/10.1021/bm040068x
Foston M, Ragauskas AJ (2010) Changes in the structure of the cellulose fiber wall during dilute acid pretreatment in populus studied by 1H and 2H NMR. Energ Fuel 24(10):5677–5685. https://doi.org/10.1021/ef100882t
French AD (2014) Idealized powder diffraction patterns for cellulose polymorphs. Cellulose 21(2):885–896. https://doi.org/10.1007/s10570-013-0030-4
Gama FM, Teixeira JA, Mota M (1994) Cellulose morphology and enzymatic reactivity: a modified solute exclusion technique. Biotechnol Bioeng 43(5):381–387. https://doi.org/10.1002/bit.260430506
Gane PAC, Ridgway CJ, Lehtinen E, Valiullin R, Furó I, Schoelkopf J, Paulapuro H, Daicic J (2004) Comparison of NMR cryoporometry, mercury intrusion porosimetry, and DSC thermoporosimetry in characterizing pore size distributions of compressed finely ground calcium carbonate structures. Ind Eng Chem Res 43(24):7920–7927. https://doi.org/10.1021/ie049448p
Haggkvist M, Li T, Odberg L (1998) Effects of drying and pressing on the pore structure in the cellulose fibre wall studied by 1H and 2H NMR relaxation. Cellulose 5(1):33–49
Hong J, Ye X, Zhang YHP (2007) Quantitative determination of cellulose accessibility to cellulase based on adsorption of a nonhydrolytic fusion protein containing CBM and GFP with Its applications. Langmuir 23(25):12535–12540. https://doi.org/10.1021/la7025686
Huang R, Su R, Qi W, He Z (2010) Understanding the key factors for enzymatic conversion of pretreated lignocellulose by partial least square analysis. Biotechnol Progr 26(2):384–392. https://doi.org/10.1002/btpr.324
Huang R, Su R, Qi W, He Z (2010) Understanding the key factors for enzymatic conversion of pretreated lignocellulose by partial least square analysis. Biotechnol Progr 26(2):384–392. https://doi.org/10.1002/btpr.324
Jayme G (1943) Über die Reaktionsfähigkeit von Zellstoffen. (on the reactivity of cellulose) (In German). Cellulosechemie 21:73–86
Junior CS, Milagres AMF, Ferraz A, Carvalho W (2013) The effects of lignin removal and drying on the porosity and enzymatic hydrolysis of sugarcane bagasse. Cellulose 20(6):3165–3177. https://doi.org/10.1007/s10570-013-0032-2
Kang K, Hwang K, Park J, Lee J, Kim J, Lee J (2018) Critical point drying: an effective drying method for direct measurement of the surface area of a pretreated cellulosic biomass. Polym Basel 10(6):676. https://doi.org/10.3390/polym10060676
Kimura M, Hatakeyama T, Nakano J (1974) DSC study on recrystallization of amorphous cellulose with water. J Appl Polym Sci 18(10):3069–3076. https://doi.org/10.1002/app.1974.070181015
Kimura M, Qi Z, Fukuzumi H, Kuga S, Isogai A (2014) Mesoporous structures in never-dried softwood cellulose fibers investigated by nitrogen adsorption. Cellulose 21(5):3193–3201. https://doi.org/10.1007/s10570-014-0342-z
Kimura M, Qi Z, Isogai A (2016) Analysis of mesopore structures in wood cell walls and pulp fibers by nitrogen adsorption method. Nord Pulp Pap Res J 31(2):198–204. https://doi.org/10.3183/npprj-2016-31-02-p198-204
Köhnke T, Lund K, Brelid H, Westman G (2010) Kraft pulp hornification: a closer look at the preventive effect gained by glucuronoxylan adsorption. Carbohyd Polym 81(2):226–233. https://doi.org/10.1016/j.carbpol.2010.02.023
Laivins GV, Scallan AM (1993) The mechanism of hornification of wood pulps. Prod Papermak 2:1235–1260
Larrondo L, St-Amour S, Monasterios C (1995) The porous structure of paper coatings-a comparison of mercury porosimetry and stain-inhibition methods of measurement. TAPPI PRESS, 79
Leu S, Zhu JY (2013) Substrate-related factors affecting enzymatic saccharification of lignocelluloses: our recent understanding. Bioenerg Res 6(2):405–415. https://doi.org/10.1007/s12155-012-9276-1
Li Y, Li B, Mo W, Yang W, Wu S (2019) Influence of residual lignin and thermal drying on the ultrastructure of chemical hardwood pulp and its enzymatic hydrolysis properties. Cellulose 26(3):2075–2085. https://doi.org/10.1007/s10570-018-2184-6
Lovikka VA, Khanjani P, Väisänen S, Vuorinen T, Maloney TC (2016) Porosity of wood pulp fibers in the wet and highly open dry state. Micropor Mesopor Mat 234:326–335. https://doi.org/10.1016/j.micromeso.2016.07.032
Maloney TC, Li TQ, Weise U, Paulapuro H (1997) Intra-and inter-fibre pore closure in wet pressing. Appita J 50(4):301–306
Maloney TC, Paulapuro H, Stenius P (1998) Hydration and swelling of pulp fibers measured with differential scanning calorimetry. Nord Pulp Pap Res J 13(1):31–36. https://doi.org/10.3183/npprj-1998-13-01-p031-036
Meng X, Foston M, Leisen J, DeMartini J, Wyman CE, Ragauskas AJ (2013) Determination of porosity of lignocellulosic biomass before and after pretreatment by using Simons’ stain and NMR techniques. Bioresour Technol 144:467–476. https://doi.org/10.1016/j.biortech.2013.06.091
Mo W, Li B (2021) Initial characteristics of the dried poplar fiber as an accessibility indicator for its enzymatic digestibility in the whole hydrolysis process. Wood Sci Technol 55(5):1337–1357. https://doi.org/10.1007/s00226-021-01318-1
Mo W, Li B, Li Y, Li Y, Wu S (2019) Overcoming the drying-induced pore closure of APMP poplar fibers in old newsprint by surfactant treatment to promote enzymatic hydrolysis of the cellulose. Cellulose 26(9):5529–5541. https://doi.org/10.1007/s10570-019-02471-4
Mo W, Ke K, Shen X, Li B (2020) The influence of “thermal drying pretreatment” on enzymatic hydrolysis of cellulose and xylan in poplar fibers with high lignin content. Carbohyd Polym 228:115400. https://doi.org/10.1016/j.carbpol.2019.115400
Mo W, Li B, Chai X (2020) Impact of fiber initial water content on the water retention capacity of poplar APMP fibers during the thermal drying. Wood Sci Technol 54(1):227–235. https://doi.org/10.1007/s00226-019-01148-2
Moon RJ, Martini A, Nairn J, Simonsen J, Youngblood J (2011) Cellulose nanomaterials review: structure, properties and nanocomposites. Chem Soc Rev 4(7):3941–3994. https://doi.org/10.1039/c0cs00108b
Nazhad MM, Ramos LP, Paszner L, Saddler JN (1995) Structural constraints affecting the initial enzymatic hydrolysis of recycled paper. Enzyme Microb Tech 17(1):68–74. https://doi.org/10.1016/0141-0229(94)00057-X
Östlund Å, Köhnke T, Nordstierna L, Nydén M (2010) NMR cryoporometry to study the fiber wall structure and the effect of drying. Cellulose 17(2):321–328. https://doi.org/10.1007/s10570-009-9383-0
Pönni R, Vuorinen T, Kontturi E (2012) Proposed nano-scale coalescence of cellulose in chemical pulp fibers during technical treatments. Bioresources 7(4):6077–6108. https://doi.org/10.15376/biores.7.4.6077-6108
Saito Y, Endo T, Ando D, Nakatsubo F, Yano H (2018) Influence of drying process on reactivity of cellulose and xylan in acetylation of willow (Salix schwerinii E. L. Wolf) kraft pulp monitored by HSQC-NMR spectroscopy. Cellulose 25(11):6319–6331. https://doi.org/10.1007/s10570-018-2034-6
Salmén L, Burgert I (2009) Cell wall features with regard to mechanical performance. A review COST Action E35 2004–2008: wood machining – micromechanics and fracture. Holzforschung 63(2):121–129. https://doi.org/10.1515/HF.2009.011
Segal L, Creely JJ, Martin AE Jr, Conrad CM (1959) An empirical method for estimating the degree of crystallinity of native cellulose using the X-ray diffractometer. Text Res J 29(10):786–794
Shen X, Sun R (2021) Recent advances in lignocellulose prior-fractionation for biomaterials, biochemicals, and bioenergy. Carbohyd Polym 261:117884. https://doi.org/10.1016/j.carbpol.2021.117884
Sing KSW (1985) Reporting physisorption data for gas/solid systems with special reference to the determination of surface area and porosity (Recommendations 1984). Pure Appl Chem 57(4):603–619. https://doi.org/10.1351/pac198557040603
Sluiter A, Hames B, Ruiz RO, Scarlata C, Sluiter J, Templeton D (2008) Determination of structural carbohydrates and lignin in biomass, National Renewable Energy Laboratory
Stone JE, Scallan AM (1968) A structural model for the cell wall of water-swollen wood pulp fibers based on their accessibility to macromolecules. Cellul Chem Technol 2(3):343–358
Thompson DN, Chen H, Grethlein HE (1992) Comparison of pretreatment methods on the basis of available surface area. Bioresour Technol 39(2):155–163. https://doi.org/10.1016/0960-8524(92)90135-K
Wadehra IL, Manley RSJ (1965) Recrystallization of amorphous cellulose. J Appl Polym Sci 9(7):2627–2630. https://doi.org/10.1002/app.1965.070090722
Wan J, Wang Y, Ma Y, Xiao Q (2009) Influence of pressing and drying on the microstructure of recycled plant fibers. Cell Chem Technol 43(1):71
Xu J, Zong M, Fu S, Li N (2016) Correlation between physicochemical properties and enzymatic digestibility of rice straw pretreated with cholinium ionic liquids. Acs Sustain Chem Eng 4(8):4340–4345. https://doi.org/10.1021/acssuschemeng.6b00860
Zimmermann MV, Borsoi C, Lavoratti A, Zanini M, Zattera AJ, Santana RM (2016) Drying techniques applied to cellulose nanofibers. J Reinf Plast Comp 35(8):628–643. https://doi.org/10.1177/0731684415626286
Acknowledgements
This work was supported by Natural Science Foundation of Guangdong Province [Grant Number 2021A1515011012]; National Natural Science Foundation of China [Grant Number 31470607]; Foundation of State Key Laboratory of Biobased Material and Green Papermaking, Qilu University of Technology, Shandong Academy of Sciences [Grant Number GZKF202033], China.
Author information
Authors and Affiliations
Contributions
WM: Conceptualization; Methodology, Formal analysis and investigation, Writing—original draft preparation. FK: Methodology, Writing—review and editing, Funding acquisition. KC: Funding acquisition, Resources, Supervision. BL: Writing—review and editing, Funding acquisition, Resources, Supervision.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no relevant financial or non-financial interests to disclose.
Human and/or animals participants
This article does not contain any studies with human participants or animals performed by any of the authors.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Data availability
All data needed to evaluate the conclusions in the paper are present in the paper. Additional data related to this paper may be requested from the authors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Mo, W., Kong, F., Chen, K. et al. Relationship between freeze-drying and supercritical drying of cellulosic fibers with different moisture contents based on pore and crystallinity measurements. Wood Sci Technol 56, 867–882 (2022). https://doi.org/10.1007/s00226-022-01387-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00226-022-01387-w