Abstract
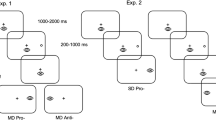
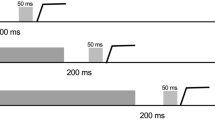
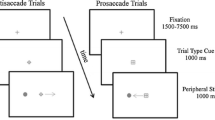
Executive function supports the rapid alternation between tasks for online reconfiguration of attentional and motor goals. The oculomotor literature has found that a prosaccade (i.e., saccade to veridical target location) preceded by an antisaccade (i.e., saccade mirror symmetrical to a target) elicits an increase in reaction time (RT), whereas the converse switch does not. This switch-cost has been attributed to the antisaccade task’s requirement of inhibiting a prosaccade (i.e., response suppression) and transforming a target’s coordinate (i.e., vector inversion)—executive processes thought to contribute to a task-set inertia that proactively interferes with the planning of a subsequent prosaccade. It is, however, unclear whether response suppression and vector inversion contribute to a task-set inertia or whether the phenomenon relates to a unitary component (e.g., response suppression). Here, the same stimulus-driven (SD) prosaccades (i.e., respond at target onset) as used in previous work were used with minimally delayed (MD) prosaccades (i.e., respond at target offset) and arranged in an AABB paradigm (i.e., A = SD prosaccade, B = MD prosaccade). MD prosaccades provide the same response suppression as antisaccades without the need for vector inversion. RTs for SD task-switch trials were longer and more variable than their task-repeat counterparts, whereas values for MD task-switch and task-repeat trials did not reliably differ. Moreover, SD task-repeat and task-switch movement times and amplitudes did not vary and thus demonstrate that a switch-cost is unrelated to a speed accuracy trade-off. Accordingly, results suggest the executive demands of response suppression is sufficient to engender the persistent activation of a non-standard task-set that selectively delays the planning of a subsequent SD prosaccade.



Similar content being viewed by others
Notes
Barton and Manoach’s group (e.g., Barton et al. 2006) have reported that an antisaccade delays the RT of any subsequent pro- or antisaccade. Notably, however, Barton and Manoach’s group employed a cued saccade paradigm wherein two targets were concurrently presented left and right of a central fixation and remained visible throughout a response. In contrast, our group as well as Chan and DeSouza (2013), have employed a classic antisaccade paradigm (Hallett 1978) wherein a single target is exogenously presented and therefore requires the sensorimotor transformation of a target’s coordinates to mirror-symmetrical space (i.e., vector inversion) (see Munoz and Everling 2004). This difference in methodology has been discussed in depth elsewhere (Weiler and Heath 2012a, b, 2014b) and we believe that it provides a parsimonious account for the difference in experimental findings.
For simplicity we henceforth use saccade – instead of prosaccade – to refer to a SR compatible response.
Correlations computed separately for SD and MD trials showed that foreperiod durations and RTs were not reliably related (ps > 0.40).
The text boxes in the main panels of Fig. 2 present skewness (g1) for each experimental condition and demonstrate a positive skew for each. We therefore employed median RT and RT IQR values in our ANOVA model. Notably, however, analysis of mean RT also revealed a task by task-transition interaction, F(1,18) = 7.76, p = 0. 012, ηp2 = 0.30. RTs for SD task-repeat and task-switch trials differed (t(18) = 3.48, p = 0.003, d = 0.80), whereas MD task-switch and task-repeat did not (t(18) = − 0.31, p = 0 .075, d = − 0.07).
References
Allport DA, Styles EA, Hsieh S (1994) Shifting intentional set: exploring the dynamic control of tasks. In: Umiltà C, Moscovitch M (eds) Attention and performance 15: conscious and nonconscious information processing. The MIT Press, Cambridge, pp 421–452
Barton JJ, Raoof M, Jameel O, Manoach DS (2006) Task-switching with antisaccades versus no-go trials: a comparison of inter-trial effects. Exp Brain Res 172:114–119
Becker W (1989) The neurobiology of saccadic eye movements. Metrics Rev Oculomot Res 3:13–67
Becker W, Fuchs AF (1969) Further properties of the human saccadic system: eye movements and correction saccades with and without visual fixation points. Vis Res 9:1247–1258
Brainard DH (1997) The psychophysics toolbox. Spat Vis 10:433–436
Bulmer MG (1979) Principles of statistics. Dover Publications, New York
Chan JL, DeSouza JF (2013) The effects of attentional load on saccadic task switching. Exp Brain Res 227:301–309
Cherkasova MV, Manoach DS, Intriligator JM, Barton JJ (2002) Antisaccades and task-switching: interactions in controlled processing. Exp Brain Res 144:528–537
Cornelissen FW, Peters EM, Palmer J (2002) The eyelink toolbox: eye tracking with matlab and the psychophysics toolbox. Behav Res Methods Instrum Comput 34:613–617
Dafoe JM, Armstrong IT, Munoz DP (2007) The influence of stimulus direction and eccentricity on pro- and anti-saccades in humans. Exp Brain Res 179:563–570
Derrfuss J, Brass M, Neumann J, von Cramon DY (2005) Involvement of the inferior frontal junction in cognitive control: meta-analyses of switching and Stroop studies. Hum Brain Mapp 25:22–34
Diamond A (2013) Executive functions. Annu Rev Psychol 64:135–168
Edelman JA, Goldberg ME (2001) Dependence of saccade-related activity in the primate superior colliculus on visual target presence. J Neurophysiol 86:676–691
Evans LH, Herron JE, Wilding EL (2015) Direct real-time neural evidence for task-set inertia. Psychol Sci 26:284–290
Everling S, Dorris MC, Klein RM, Munoz DP (1999) Role of primate superior colliculus in preparation and execution of anti-saccades and pro-saccades. J Neurosci 19:2740–2754
Everling S, Johnston K (2013) Control of the superior colliculus by the lateral prefrontal cortex. Philos Trans R Soc Lond B Biol Sci 368:20130068
Fitts PM (1954) The information capacity of the human motor system in controlling the amplitude of movement. J Exp Psychol Gen 47:381–391
Gillen C, Heath M (2014) Target frequency influences antisaccade endpoint bias: evidence for perceptual averaging. Vis Res 105:151–158
Hallett PE (1978) Primary and secondary saccades to goals defined by instructions. Vis Res 8:1279–1296
Harris CM (1995) Does saccadic undershoot minimize saccadic flight-time? A Monte-Carlo study. Vis Res 35:691–701
Heath M, Gillen C, Samani A (2016) Alternating between pro-and antisaccades: switch-costs manifest via decoupling the spatial relations between stimulus and response. Exp Brain Res 234:853–865
Kleiner M, Brainard D, Pelli D, Ingling A, Murray R, Broussard C (2007) What’s new in Psychtoolbox-3. Perception 36:1–16
Lakens D (2017) Equivalence tests: a practical primer for t-tests, correlations, and meta-analyses. Soc Psychol Personal Sci 8:355–362
Lakens D, Scheel AM, Isager P (2018) Equivalence testing for psychological research: a tutorial. Adv Methods Pract Psychol Sci 1:259–269
Li L, Wang M, Zhao QJ, Fogelson N (2012) Neural mechanisms underlying the cost of task switching: an ERP study. PLoS One 7:e42233
Manoach DS, Thakkar KN, Cain MS, Polli FE, Edelman JA, Fischl B, Barton JJ (2007) Neural activity is modulated by trial history: a functional magnetic resonance imaging study of the effects of a previous antisaccade. J Neurosci 27:1791–1798
Monsell S (2003) Task switching. Trends Cogn Sci 7:134–140
Munoz DP, Everling S (2004) Look away: the anti-saccade task and the voluntary control of eye movement. Nat Rev Neurosci 5:218–228
Nee DE, Wager TD, Jonides J (2007) Interference resolution: insights from a meta-analysis of neuroimaging tasks. Cogn Affect Behav Neurosci 7:1–17
Pierrot-Deseilligny C, Rivaud S, Gaymard B, Müri R, Vermersch AI (1995) Cortical control of saccades. Ann Neurol 37:557–567
Pouget P, Logan GD, Palmeri TJ, Boucher L, Paré M, Schall JD (2011) Neural basis of adaptive response time adjustment during saccade countermanding. J Neurosci 31:12604–12612
Schall JD (2002) The neural selection and control of saccades by the frontal eye field. Philos Trans R Soc Lond B Biol Sci 357:1073–1082
Stuphorn V, Schall JD (2006) Executive control of countermanding saccades by the supplementary eye field. Nat Neurosci 9:925–931
Trappenberg TP, Dorris MC, Munoz DP, Klein RM (2001) A model of saccade initiation based on the competitive integration of exogenous and endogenous signals in the superior colliculus. J Cogn Neurosci 13:256–271
Webb B, Humphreys D, Heath M (2018) Oculomotor executive dysfunction during the early and later stages of sport-related concussion recovery. J Neurotrauma 35:1874–1881
Weiler J, Heath M (2012a) The prior-antisaccade effect influences the planning and online control of prosaccades. Exp Brain Res 216:545–552
Weiler J, Heath M (2012b) Task-switching in oculomotor control: Unidirectional switch-cost when alternating between pro- and antisaccades. Neurosci Lett 530:150–154
Weiler J, Heath M (2014a) Oculomotor task switching: alternating from a nonstandard to a standard response yields the unidirectional prosaccade switch-cost. J Neurophysiol 112:2176–2184
Weiler J, Heath M (2014b) Repetitive antisaccade execution does not increase the unidirectional prosaccade switch-cost. Acta Psychol (Amst) 146:67–72
Weiler J, Mitchell T, Heath M (2014) Response suppression delays the planning of subsequent stimulus-driven saccades. PLoS One 9:e86408
Weiler J, Hassall CD, Krigolson OE, Heath M (2015) The unidirectional prosaccade switch-cost: Electroencephalographic evidence of task-set inertia in oculomotor control. Behav Brain Res 278:323–329
Wenban-Smith MG, Findlay JM (1991) Express saccades: is there a separate population in humans? Exp Brain Res 87:218–222
Westwood DA, Heath M, Roy EA (2003) No evidence for accurate visuomotor memory: systematic and variable error in memory-guided reaching. J Mot Behav 35:127–133
White JM, Sparks DL, Stanford TR (1994) Saccades to remembered target locations: an analysis of systematic and variable errors. Vis Res 34:79–92
Wolohan FDA, Knox PC (2014) Oculomotor inhibitory control in express saccade makers. Exp Brain Res 232:3949–3963
Wurtz RH, Albano JE (1980) Visual-motor function of the primate superior colliculus. Annu Rev Neurosci 3:189–226
Wylie G, Allport A (2000) Task switching and the measurement of “switch costs”. Psychol Res 63:212–233
Wylie GR, Sumowski JF, Murray M (2011) Are there control processes, and (if so) can they be studied? Psychol Res 75:535–543
Yeung N, Nystrom LE, Aronson JA, Cohen JD (2006) Between-task competition and cognitive control in task switching. J Neurosci 26:1429–1438
Zambarbieri D, Beltrami G, Versino M (1995) Saccade latency toward auditory targets depends on the relative position of the sound source with respect to the eyes. Vis Res 35:3305–3312
Acknowledgements
This work was supported by a Discovery Grant from the Natural Sciences and Engineering Research Council (NSERC) of Canada, and Faculty Scholar and Major Academic Development Fund Awards from the University of Western Ontario. The authors declare no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Tari, B., Fadel, M.A. & Heath, M. Response suppression produces a switch-cost for spatially compatible saccades. Exp Brain Res 237, 1195–1203 (2019). https://doi.org/10.1007/s00221-019-05497-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00221-019-05497-z