Abstract
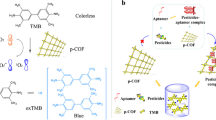
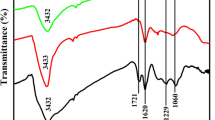
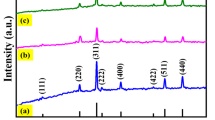
Nanozymes have both the high catalytic activity of natural enzymes and the stability and economy of mimetic enzymes. Research on nanozymes is rapidly emerging, and the continuous development of highly catalytic active nanozymes is of far-reaching significance. This work reports heteroatomic nitrogen (N) and phosphorus (P) double-doped mesoporous carbon structures and metallic Fe coordination generated sponge-like nanozymes (Fe/NPCs) with good peroxidase activity. On this basis, we constructed a highly sensitive colorimetric sensor with cysteine and phenol as simulated analytes using Fe/NPCs nanozymes, and the response limits reached 53.6 nM and 5.4 nM, respectively. Besides, the method has high accuracy in the detection of cysteine and phenol at low concentrations in serum and tap water, which lays a foundation for application in the fields of environmental protection and biosensors.
Graphical abstract







Similar content being viewed by others
References
Gao L, Zhuang J, Nie L, Zhang J, Zhang Y, et al. Intrinsic peroxidase-like activity of ferromagnetic nanoparticles. Nat Nanotechnol. 2007;2:577–83.
Wang H, Wan K, Shi X. Recent advances in nanozyme research. Adv Mater. 2019;31:1805368.
Zhang Z, Zhang X, Liu B, Liu J. Molecular imprinting on inorganic nanozymes for hundred-fold enzyme specificity. J Am Chem Soc. 2017;139:5412–9.
Zhang J, Wu S, Lu X, Wu P, Liu J. Manganese as a catalytic mediator for photo-oxidation and breaking the pH limitation of nanozymes. Nano Lett. 2019;19:3214–20.
Wu JH, Lv WX, Yang QT, et al. Label-free homogeneous electrochemical detection of microRNA based on target-induced anti-shielding against the catalytic activity of two-dimension nanozyme. Biosens Bioelectron. 2021;171:112707.
Zhang H, Liang X, Han L, Li F. “Non-naked” gold with glucose oxidase-like activity: a nanozyme for tandem catalysis. Small. 2018;14:1803256.
Lu N, Zhang M, Ding L, Zheng J, Zeng C, et al. Yolk–shell nanostructured Fe3O4@C magnetic nanoparticles with enhanced peroxidase-like activity for label-free colorimetric detection of H2O2 and glucose. Nanoscale. 2017;9:4508–15.
Wu JH, Yang QT, Li Q, Li HY, Li F. Two-dimensional MnO2 nanozyme-mediated homogeneous electrochemical detection of organophosphate pesticides without the interference of H2O2 and color. Anal Chem. 2021;93:4084–91.
Hu Y, Gao XJ, Zhu Y, Muhammad F, Tan S, Cao W, et al. Nitrogen-doped carbon nanomaterials as highly active and specific peroxidase mimics. Chem Mater. 2018;30:6431–9.
Lin L, Song X, Chen Y, Rong M, Zhao T, et al. Intrinsic peroxidase-like catalytic activity of nitrogen-doped graphene quantum dots and their application in the colorimetric detection of H2O2 and glucose. Anal Chim Acta. 2015;869:89–95.
Fan K, Xi J, Fan L, Wang P, Zhu C, Tang Y, et al. Vivo guiding nitrogen-doped carbon nanozyme for tumor catalytic therapy. Nat Commun. 2018;9:1440–50.
Zhu DQ, Zhang ML, Pu L, Gai PP, Li F. Nitrogen-enriched conjugated polymer enabled metal-free carbon nanozymes with efficient oxidase-like activity. Small. 2021;18:2104993.
Li W, Wang D, Zhang Y, Tao L, Wang T, et al. Defect engineering for fuel-cell electrocatalysts. Adv Mater. 2020;32:1907879.
Liu Z, Ai J, Sun M, Han F, Li Z, Peng Q, et al. Phosphorous-doped graphite layers with outstanding electrocatalytic activities for the oxygen and hydrogen evolution reactions in water electrolysis. Adv Funct Mater. 2020;30:1910741.
Liu XF, Antonietti M. Molten salt activation for synthesis of porous carbon nanostructures and carbon sheets. Carbon. 2014;69:460–6.
Qu K, Zheng Y, Zhang X, Davey K, Dai S, Qiao SZ. Promotion of electrocatalytic hydrogen evolution reaction on nitrogen-doped carbon nanosheets with secondary heteroatoms. ACS Nano. 2017;11:7293–300.
Gao K, Wang B, Tao L, Cunning BV, Zhang Z, et al. Efficient metal-free electrocatalysts from N-doped carbon nanomaterials: mono-doping and co-doping. Adv Mater. 2019;31:1805121.
Zhang CH, Liu WD, Chen CX, Ni PJ, et al. Emerging interstitial/substitutional modification of Pd-based nanomaterials with nonmetallic elements for electrocatalytic applications. Nanoscale. 2022;14:2915–42.
Zhang XH, Li QG, Hou DY. Effects of feed solution pH and draw solution concentration on the performance of phenolic compounds removal in forward osmosis process. J Environ Chem Eng. 2017;5:2508–14.
Zhang CS, Wang XJ, Wang LG. Removal of phenolic substances from wastewater by algae. Environ Chem Lett. 2020;18:377–92.
Lu Y, Yan LH, Zhang JF. Biodegradation of phenolic compounds from coking wastewater by immobilized white rot fungus Phanerochaete chrysosporium. J Hazard Mater. 2009;165:1091–7.
Tian HL, Xu XJ, Li BA. Biodegradation of phenolic compounds in high saline wastewater by biofilms adhering on aerated membranes. J Hazard Mater. 2020;392:122463.
Rahmanian N, Jafari SM, Galanakis CM. Recovery and removal of phenolic compounds from olive mill wastewater. J Am Oil Chem Soc. 2014;91:1–18.
Ma XY, Zhang XW, Ye L, et al. Phenolic compounds promote the horizontal transfer of antibiotic resistance genes in activated sludge. Sci Total Environ. 2021;800:149549.
Liu S, Huang BY, Liang L, et al. Nanocapsulation of horseradish peroxidase (HRP) enhances enzymatic performance in removing phenolic compounds. Int J Biol Macromol. 2020;150:814–22.
Alshabib M, Onaizi SA. A review on phenolic wastewater remediation using homogeneous and heterogeneous enzymatic processes: current status and potential challenges. Sep Purif Technol. 2019;219:186–207.
Salehi S, Abdollahi K, Mokhtarani B, et al. Applications of biocatalysts for sustainable oxidation of phenolic pollutants: a review. Sustainability. 2021;13:8620.
Paisio CE, Agostini E, Bertuzzi ML, et al. Lethal and teratogenic effects of phenol on Bufo arenarum embryos. J Hazard Mater. 2009;167:64–8.
Nesvera J, Rucka L, Patek M. Catabolism of phenol and its derivatives in bacteria: genes, their regulation, and use in the biodegradation of toxic pollutants. Adv Appl Microbiol. 2015;93:107–60.
Paterson B, Cowie CE, Jackson PE. Determination of phenols in environmental waters using liquid chromatography with electrochemical detection. J Chromatogr A. 1996;731:95–102.
Fukushi K, Yakushiji Y, Saito K, et al. Simultaneous determination of a pyridine-triphenylborane anti-fouling agent and its estimated degradation products using capillary zone electrophoresis. J Chromatogr A. 2010;1217:2187–90.
Chen K, Zhang ZL, Liu W, et al. A graphene-based electrochemical sensor for rapid determination of phenols in water. Sensors. 2013;13:6204–16.
Xiao X, Wang YH, Tong ZW, et al. Synthesis of pumpkin-like CeO2 microstructures and electrochemical detection for phenol. Inorg Nano-Met Chem. 2019;49:349–53.
Seebunrueng K, Dejchaiwatana CH, Santaladchaiyakitb Y, Srijaranai S. Development of supramolecular solvent based microextraction prior to high performance liquid chromatography for simultaneous determination of phenols in environmental water. RSC Adv. 2017;7:50143–9.
Cesarino I, Moraes FC, Machado SAS, et al. Real-time electrochemical determination of phenolic compounds after benzene oxidation. J Electroanal Chem. 2012;672:34–9.
Zhang Y, Yang KL. Quantitative detection of phenol in wastewater using square wavevoltammetry with pre-concentration. Anal Chim Acta. 2021;1178:338788.
Ma J, Yu Y, Chen CL, Zuo X, et al. Using multifunctional polymeric soft template for synthesizing nitrogen and phosphorus co–doped mesoporous carbon frameworks electrocatalysts for oxygen reduction reaction. ChemistrySelect. 2018;3:9013–20.
Suk Jung W, Popov BN. Improved durability of Pt catalyst supported on N-doped mesoporous graphitized carbon for oxygen reduction reaction in polymer electrolyte membrane fuel cells. Carbon. 2017;122:746–55.
Deng C, Zhong H, Li X, Yao L, Zhang H. A highly efficient electrocatalyst for oxygen reduction reaction: phosphorus and nitrogen co-doped hierarchically ordered porous carbon derived from an iron-functionalized polymer. Nanoscale. 2016;8:1580–7.
Zhang H, Wang T, Wang J, Liu H, Dao TD, et al. Surface-plasmon-enhanced photodriven CO2 reduction catalyzed by metal–organic-framework-derived iron nanoparticles encapsulated by ultrathin carbon layers. Adv Mater. 2016;28:3703–10.
Funding
This work was financially supported by the Natural Science Foundation of Beijing Municipality (no. 2192010 and no. 2182012), the National Natural Science Foundation (no. 11179033), and Capacity Building for Sci-Tech Innovation-Fundamental Scientific Research Funds, Scientific Research Project of Beijing Educational Committee (KM202010028007).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Xue, Y., Zhong, H., Liu, B. et al. Colorimetric sensing strategy for detection of cysteine, phenol cysteine, and phenol based on synergistic doping of multiple heteroatoms into sponge-like Fe/NPC nanozymes. Anal Bioanal Chem 414, 4217–4225 (2022). https://doi.org/10.1007/s00216-022-04074-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00216-022-04074-8