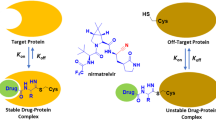
Abstract
Histone deacetylase 6 (HDAC6) plays an important role in cancer treatment, and the development of selective HDAC6 inhibitors (sHDAC6is) attracts more and more attention in recent years. In this research, a series of ACY-1215 analogs based on diphenylpyrimidine scaffold were designed and synthesized. Among these, the most potent compound 7-((4, 6-diphenylpyrimidin-2-yl)amino)-N-hydroxyheptanamide (11a) inhibited HDAC6 with IC50 of 3.8 nM and showed 26~fold selectivity over HDAC1, better than those of ACY-1215. In cellular assay, these diphenylpyrimidines exhibited promising antiproliferative activities against different tumor cell lines. Altogether, this work highlighted the therapeutic potential of diphenylpyrimidine-inspired sHDAC6 inhibitors and provides a valuable lead compound for the discovery of novel antitumor agents.







Similar content being viewed by others
References
Minucci S, Pelicci PG. Histone deacetylase inhibitors and the promise of epigenetic (and more) treatments for cancer. Nat Rev Cancer. 2006;6:38–51. https://doi.org/10.1038/nrc1779
Liang T, Xie Z, Dang B, Wang J, Zhang T, Luan X, et al. Discovery of indole-piperazine derivatives as selective histone deacetylase 6 inhibitors with neurite outgrowth-promoting activities and neuroprotective activities. Bioorg Med Chem Lett. 2023;81:129148. https://doi.org/10.1016/j.bmcl.2023.129148
Falkenberg KJ, Johnstone RW. Histone deacetylases and their inhibitors in cancer, neurological diseases and immune disorders. Nat Rev Drug Discov. 2014;13:673–91. https://doi.org/10.1038/nrd4360
Bolden JE, Peart MJ, Johnstone RW. Anticancer activities of histone deacetylase inhibitors. Nat Rev Drug Discov. 2006;5:769–84. https://doi.org/10.1038/nrd2133
Gregoretti IV, Lee YM, Goodson HV. Molecular evolution of the histone deacetylase family: functional implications of phylogenetic analysis. J Mol Biol. 2004;338:17–31. https://doi.org/10.1016/j.jmb.2004.02.006
Kalin JH, Bergman JA. Development and therapeutic implications of selective histone deacetylase 6 inhibitors. J Med Chem. 2013;56:6297–313. https://doi.org/10.1021/jm4001659
LoPresti P. HDAC6 in diseases of cognition and of neurons. Cells. 2020;10. https://doi.org/10.3390/cells10010012
Govindarajan N, Rao P, Burkhardt S, Sananbenesi F, Schluter OM, Bradke F, et al. Reducing HDAC6 ameliorates cognitive deficits in a mouse model for Alzheimer’s disease. EMBO Mol Med. 2013;5:52–63. https://doi.org/10.1002/emmm.201201923
Witt O, Deubzer HE, Milde T, Oehme I. HDAC family: What are the cancer relevant targets. Cancer Lett. 2009;277:8–21. https://doi.org/10.1016/j.canlet.2008.08.016
Yang PH, Zhang L, Zhang YJ, Zhang J, Xu WF. HDAC6: physiological function and its selective inhibitors for cancer treatment. Drug Discov Ther. 2013;7:233–42. https://doi.org/10.5582/ddt.2013.v7.6.233
Zhao Y, Liang T, Hou X, Fang H. Recent Development of Novel HDAC6 Isoform-selective Inhibitors. Curr Med Chem. 2021;28:4133–51. https://doi.org/10.2174/0929867327666201111142653
Chen X, Chen X, Steimbach RR, Wu T, Li H, Dan W, et al. Novel 2, 5-diketopiperazine derivatives as potent selective histone deacetylase 6 inhibitors: Rational design, synthesis and antiproliferative activity. Eur J Med Chem. 2020;187:111950. https://doi.org/10.1016/j.ejmech.2019.111950
Chen X, Zhao S, Li H, Wang X, Geng A, Cui H, et al. Design, synthesis and biological evaluation of novel isoindolinone derivatives as potent histone deacetylase inhibitors. Eur J Med Chem. 2019;168:110–22. https://doi.org/10.1016/j.ejmech.2019.02.032
Chen X, Gong G, Chen X, Song R, Duan M, Qiao R, et al. Design, synthesis and biological evaluation of novel benzoylimidazole derivatives as raf and histone deacetylases dual inhibitors. Chem Pharm Bull. 2019;67:1116–22. https://doi.org/10.1248/cpb.c19-00425
Gong G, Qi J, Lv Y, Dong S, Cao C, Li D, et al. Discovery of 1,3-disubstituted 2,5-diketopiperazine derivatives as potent class I HDACs inhibitors. Chem Pharm Bull. 2020;68:466–72. https://doi.org/10.1248/cpb.c20-00056
Xing L, Gong G, Chen X, Chen X. Discovery of indole-piperazine hybrid structures as potent selective class I histone deacetylases inhibitors. Chem Pharm Bull. 2023;71:206–12. https://doi.org/10.1248/cpb.c22-00635
Wong JC, Hong R, Schreiber SL. Structural biasing elements for in-cell histone deacetylase paralog selectivity. J Am Chem Soc. 2003;125:5586–7. https://doi.org/10.1021/ja0341440
Haggarty SJ, Koeller KM, Wong JC, Grozinger CM, Schreiber SL. Domain-selective small-molecule inhibitor of histone deacetylase 6 (HDAC6)-mediated tubulin deacetylation. Proc Natl Acad Sci USA. 2003;100:4389–94. https://doi.org/10.1073/pnas.0430973100
Santo L, Hideshima T, Kung AL, Tseng JC, Tamang D, Yang M, et al. Preclinical activity, pharmacodynamic, and pharmacokinetic properties of a selective HDAC6 inhibitor, ACY-1215, in combination with bortezomib in multiple myeloma. Blood. 2012;119:2579–89. https://doi.org/10.1182/blood-2011-10-387365
Huang P, Almeciga-Pinto I, Jarpe M, van Duzer JH, Mazitschek R, Yang M, et al. Selective HDAC inhibition by ACY-241 enhances the activity of paclitaxel in solid tumor models. Oncotarget. 2017;8:2694–707. https://doi.org/10.18632/oncotarget.13738
Pathak V, Maurya HK, Sharma S, Srivastava KK, Gupta A. Synthesis and biological evaluation of substituted 4,6-diarylpyrimidines and 3,5-diphenyl-4,5-dihydro-1H-pyrazoles as anti-tubercular agents. Bioorg Med Chem Lett. 2014;24:2892–6. https://doi.org/10.1016/j.bmcl.2014.04.094
Kanagarajan V, Thanusu J, Gopalakrishnan M. Synthesis and in vitro microbiological evaluation of an array of biolabile 2-morpholino-N-(4,6-diarylpyrimidin-2-yl)acetamides. Eur J Med Chem. 2010;45:1583–9. https://doi.org/10.1016/j.ejmech.2009.12.068
Lee YH, Park J, Ahn S, Lee Y, Lee J, Shin SY, et al. Design, synthesis, and biological evaluation of polyphenols with 4,6-diphenylpyrimidin-2-amine derivatives for inhibition of Aurora kinase A. Daru. 2019;27:265–81. https://doi.org/10.1007/s40199-019-00272-5
Val C, Rodriguez-Garcia C, Prieto-Diaz R, Crespo A, Azuaje J, Carbajales C, et al. Optimization of 2-Amino-4,6-diarylpyrimidine-5-carbonitriles as Potent and Selective A(1) Antagonists. J Med Chem. 2022;65:2091–106. https://doi.org/10.1021/acs.jmedchem.1c01636
Acknowledgements
This work was supported by the National Natural Science Foundation of China (81903464) and the Natural Science Basic Research Plan in Shaanxi Province of China (2020JQ-284).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Duan, H., Wang, J., Gong, G. et al. Design, synthesis and antiproliferative activity of ACY-1215 analogs as potent selective histone deacetylases 6 inhibitors. Med Chem Res 32, 2432–2441 (2023). https://doi.org/10.1007/s00044-023-03150-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00044-023-03150-7