Abstract
Background
The Ca2+/calmodulin-dependent protein kinase kinases (CaMKKs) are serine/threonine-directed protein kinases that are activated following increases in intracellular calcium, playing a critical role in neuronal signaling. Inner-ear-trauma-induced calcium overload in sensory hair cells has been well documented in the pathogenesis of traumatic noise-induced hair cell death and hearing loss, but there are no established pharmaceutical therapies available due to a lack of specific therapeutic targets. In this study, we investigated the activation of CaMKKβ in the inner ear after traumatic noise exposure and assessed the prevention of noise-induced hearing loss (NIHL) with RNA silencing.
Results
Treatment with short hairpin RNA of CaMKKβ (shCaMKKβ) via adeno-associated virus transduction significantly knocked down CaMKKβ expression in the inner ear. Knockdown of CaMKKβ significantly attenuated noise-induced hair cell loss and hearing loss (NIHL). Additionally, pretreatment with naked CaMKKβ small interfering RNA (siCaMKKβ) attenuated noise-induced losses of inner hair cell synapses and OHCs and NIHL. Furthermore, traumatic noise exposure activates CaMKKβ in OHCs as demonstrated by immunolabeling for p-CaMKI. CaMKKβ mRNA assessed by fluorescence in-situ hybridization and immunolabeling for CaMKKβ in OHCs also increased after the exposure. Finally, pretreatment with siCaMKKβ diminished noise-induced activation of AMPKα in OHCs.
Conclusions
These findings demonstrate that traumatic-noise-induced OHC loss and hearing loss occur primarily via activation of CaMKKβ. Targeting CaMKKβ is a key strategy for prevention of noise-induced hearing loss. Furthermore, our data suggest that noise-induced activation of AMPKα in OHCs occurs via the CaMKKβ pathway.



taken from the apical, middle, basal, and hook turns of whole mount surface preparations immunolabeled with myosin VIIa and then stained with DAB to illustrate OHCs and IHCs. OHC 1, 2, 3, and IHC indicate three rows of outer hair cells and one row of inner hair cells. Counts of OHC loss are shown in Fig. 4A. These images are representative of six mice for each group. Scale bar = 10 µm

taken from the lower basal cochlear turn. Scale bar = 20 µm. C Noise-induced auditory threshold shifts are significantly reduced in AAV-shCaMKKβ mice compared to AAV-shCtrl mice calculated as baseline ABR thresholds subtracted from thresholds measured 14 d after noise exposure for each individual mouse. Data are presented as individual mice ± SD. D Noise-diminished DPOAE amplitudes are significantly reversed by shCaMKKβ application measured 14 d after noise exposure. Data are presented as means ± SD. The detailed statistical values are listed in Table S2; *indicates the p value of the comparison between shCtrl + noise exposure vs shCaMKKβ + noise exposure, *p < 0.05, **p < 0.01, ***p < 0.001, ****p < 0.0001

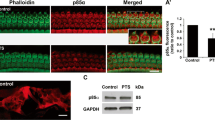
taken from the basal turn at 4 mm from the apex and are representative of 5–7 mice for each group. Scale bar = 10 µm. F Counts of OHC loss along the entire length of the cochlear duct show that pretreatment with siCaMKKβ significantly reduced noise-induced OHC loss. Data are presented as means + SD. Detailed statistical values are listed in Table S4. The number of animals in each group is indicated in the labels, *indicates the p value for the comparison between siCtrl + PTSN vs siCaMKKβ + PTSN. *p < 0.05, **p < 0.01, ***p < 0.001





Similar content being viewed by others
Availability of data and materials
All data generated or analyzed during this study are included in this published article and its supplementary information files.
Abbreviations
- AAV:
-
Adeno-associated virus
- ABR:
-
Auditory brainstem response
- AMPKα:
-
AMP-dependent protein kinase α subunit
- CaMKKβ:
-
Ca2+/calmodulin-dependent protein kinase kinase-β
- DPOAE:
-
Distortion product otoacoustic emissions
- eGFP:
-
Enhanced green fluorescent protein
- FISH:
-
Fluorescent in-situ hybridization
- HCs:
-
Hair cells
- IHCs:
-
Inner hair cells
- IP:
-
Intraperitoneal injection
- KO:
-
Knockout mice
- LKB1:
-
Liver kinase B1
- MET:
-
Mechanoelectrical transducer channels
- NIHL:
-
Noise-induced hearing loss
- OHCs:
-
Outer hair cells
- PBS:
-
Phosphate-buffered saline
- PBS-T:
-
PBS with 0.1% Tween 20
- PTS:
-
Permanent threshold shift
- PTSN:
-
PTS-noise
- ROS:
-
Reactive oxygen species
- RWM:
-
Round window membrane
- SDS-PAGE:
-
Sodium dodecyl sulfate polyacrylamide gel electrophoresis
- SPL:
-
Sound pressure level
- siRNA:
-
Small interfering RNA silencing
- shRNA:
-
Short hairpin RNA silencing
- siControl:
-
Scrambled siRNA
- TTS:
-
Temporary threshold shift
- TDT:
-
Tucker Davis Technology
References
Anderson KA, Ribar TJ, Lin F, Noeldner PK, Green MF, Muehlbauer MJ, Witters LA, Kemp BE, Means AR (2008) Hypothalamic CaMKK2 contributes to the regulation of energy balance. Cell Metab 7:377–388
Askew C, Chien WW (2020) Adeno-associated virus gene replacement for recessive inner ear dysfunction: Progress and challenges. Hear Res 394:107947
Bankoti K, Generotti C, Hwa T, Wang L, O’Malley BW Jr, Li D (2021) Advances and challenges in adeno-associated viral inner-ear gene therapy for sensorineural hearing loss. Molecular therapy Methods & clinical development 21:209–236
Carling D (2017) AMPK signalling in health and disease. Curr Opin Cell Biol 45:31–37
Ceriani F, Mammano F (2012) Calcium signaling in the cochlea - Molecular mechanisms and physiopathological implications. Cell Commun Signal 10:20
Chakraborty C, Sharma AR, Sharma G, Doss CGP, Lee SS (2017) Therapeutic miRNA and siRNA: moving from bench to clinic as next generation medicine. Mol Ther Nucleic Acids 8:132–143
Chen FQ, Zheng HW, Hill K, Sha SH (2012) Traumatic noise activates rho-family GTPases through transient cellular energy depletion. J Neurosci 32:12421–12430
Chen J, Hill K, Sha SH (2016) Inhibitors of histone deacetylases attenuate noise-induced hearing loss. J Assoc Res Otolaryngol 17:289–302
Crane R, Conley SM, Al-Ubaidi MR, Naash MI (2021) Gene therapy to the retina and the cochlea. Front Neurosci 15:652215
Dong JY, Fan PD, Frizzell RA (1996) Quantitative analysis of the packaging capacity of recombinant adeno-associated virus. Hum Gene Ther 7:2101–2112
Dumont RA, Lins U, Filoteo AG, Penniston JT, Kachar B, Gillespie PG (2001) Plasma membrane Ca2+-ATPase isoform 2a is the PMCA of hair bundles. J Neurosci 21:5066–5078
Esterberg R, Hailey DW, Coffin AB, Raible DW, Rubel EW (2013) Disruption of intracellular calcium regulation is integral to aminoglycoside-induced hair cell death. J Neurosci 33:7513–7525
Esterberg R, Hailey DW, Rubel EW, Raible DW (2014) ER-mitochondrial calcium flow underlies vulnerability of mechanosensory hair cells to damage. J Neurosci 34:9703–9719
Fang QJ, Wu F, Chai R, Sha SH (2019) Cochlear surface preparation in the adult mouse. J Vis Exp. https://doi.org/10.3791/60299
Fetoni AR, Paciello F, Rolesi R, Paludetti G, Troiani D (2019) Targeting dysregulation of redox homeostasis in noise-induced hearing loss: Oxidative stress and ROS signaling. Free Radic Biol Med 135:46–59
Fettiplace R (2017) Hair cell transduction, tuning, and synaptic transmission in the mammalian cochlea. Compr Physiol 7:1197–1227
Fridberger A, Flock A, Ulfendahl M, Flock B (1998) Acoustic overstimulation increases outer hair cell Ca2+ concentrations and causes dynamic contractions of the hearing organ. Proc Natl Acad Sci U S A 95:7127–7132
Grotemeier A, Alers S, Pfisterer SG, Paasch F, Daubrawa M, Dieterle A, Viollet B, Wesselborg S, Proikas-Cezanne T, Stork B (2010) AMPK-independent induction of autophagy by cytosolic Ca2+ increase. Cell Signal 22:914–925
Hardie DG (2003) Minireview: the AMP-activated protein kinase cascade: the key sensor of cellular energy status. Endocrinology 144:5179–5183
He Z-H, Pan S, Zheng H-W, Fang Q-J, Hill K, Sha S-H (2021) Treatment with calcineurin inhibitor fk506 attenuates noise-induced hearing loss. Front Cell Develop Biol. https://doi.org/10.3389/fcell.2021.648461
Henderson D, Bielefeld EC, Harris KC, Hu BH (2006) The role of oxidative stress in noise-induced hearing loss. Ear Hear 27:1–19
Hill K, Yuan H, Wang X, Sha SH (2016) Noise-induced loss of hair cells and cochlear synaptopathy are mediated by the activation of AMPK. J Neurosci 36:7497–7510
Ho MK, Li X, Wang J, Ohmen JD, Friedman RA (2014) FVB/NJ mice demonstrate a youthful sensitivity to noise-induced hearing loss and provide a useful genetic model for the study of neural hearing loss. Audiol Neurotol Extra 4:1–11
Hurley RL, Anderson KA, Franzone JM, Kemp BE, Means AR, Witters LA (2005) The Ca2+/calmodulin-dependent protein kinase kinases are AMP-activated protein kinase kinases. J Biol Chem 280:29060–29066
Isgrig K, McDougald DS, Zhu J, Wang HJ, Bennett J, Chien WW (2019) AAV2.7m8 is a powerful viral vector for inner ear gene therapy. Nat Commun 10:427
Jeon SM (2016) Regulation and function of AMPK in physiology and diseases. Exp Mol Med 48:e245
Kersigo J, Pan N, Lederman JD, Chatterjee S, Abel T, Pavlinkova G, Silos-Santiago I, Fritzsch B (2018) A RNAscope whole mount approach that can be combined with immunofluorescence to quantify differential distribution of mRNA. Cell Tissue Res 374:251–262
Kimura Y, Corcoran EE, Eto K, Gengyo-Ando K, Muramatsu MA, Kobayashi R, Freedman JH, Mitani S, Hagiwara M, Means AR, Tokumitsu H (2002) A CaMK cascade activates CRE-mediated transcription in neurons of Caenorhabditis elegans. EMBO Rep 3:962–966
Kopke R, Slade MD, Jackson R, Hammill T, Fausti S, Lonsbury-Martin B, Sanderson A, Dreisbach L, Rabinowitz P, Torre P 3rd, Balough B (2015) Efficacy and safety of N-acetylcysteine in prevention of noise induced hearing loss: a randomized clinical trial. Hear Res 323:40–50
Kramer S, Dreisbach L, Lockwood J, Baldwin K, Kopke R, Scranton S, O’Leary M (2006) Efficacy of the antioxidant N-acetylcysteine (NAC) in protecting ears exposed to loud music. J Am Acad Audiol 17:265–278
Kujawa SG, Liberman MC (2009) Adding insult to injury: cochlear nerve degeneration after “temporary” noise-induced hearing loss. J Neurosci 29:14077–14085
Landegger LD, Pan B, Askew C, Wassmer SJ, Gluck SD, Galvin A, Taylor R, Forge A, Stankovic KM, Holt JR, Vandenberghe LH (2017) A synthetic AAV vector enables safe and efficient gene transfer to the mammalian inner ear. Nat Biotechnol 35:280–284
Le Prell CG, Hammill TL, Murphy WJ (2019) Noise-induced hearing loss and its prevention: Integration of data from animal models and human clinical trials. J Acoust Soc Am 146:4051
Lin CY, Wu JL, Shih TS, Tsai PJ, Sun YM, Ma MC, Guo YL (2010) N-Acetyl-cysteine against noise-induced temporary threshold shift in male workers. Hear Res 269:42–47
Mammano F, Frolenkov GI, Lagostena L, Belyantseva IA, Kurc M, Dodane V, Colavita A, Kachar B (1999) ATP-Induced Ca(2+) release in cochlear outer hair cells: localization of an inositol triphosphate-gated Ca(2+) store to the base of the sensory hair bundle. J Neurosci 19:6918–6929
Marcelo KL, Means AR, York B (2016) The Ca(2+)/Calmodulin/CaMKK2 Axis: Nature’s Metabolic CaMshaft. Trends Endocrinol Metab 27:706–718
Means AR (2000) Regulatory cascades involving calmodulin-dependent protein kinases. Mol Endocrinol 14:4–13
Muller M, von Hunerbein K, Hoidis S, Smolders JW (2005) A physiological place-frequency map of the cochlea in the CBA/J mouse. Hear Res 202:63–73
Najar MA, Rex DAB, Modi PK, Agarwal N, Dagamajalu S, Karthikkeyan G, Vijayakumar M, Chatterjee A, Sankar U, Prasad TSK (2021) A complete map of the Calcium/calmodulin-dependent protein kinase kinase 2 (CAMKK2) signaling pathway. J Cell Commun Signal 15:283–290
Nyberg S, Abbott NJ, Shi X, Steyger PS, Dabdoub A (2019) Delivery of therapeutics to the inner ear: The challenge of the blood-labyrinth barrier. Sci Transl Med. https://doi.org/10.1126/scitranslmed.aao0935
Ohinata Y, Yamasoba T, Schacht J, Miller JM (2000) Glutathione limits noise-induced hearing loss. Hear Res 146:28–34
Ohlemiller KK (2019) Mouse methods and models for studies in hearing. J Acoust Soc Am 146:3668
Oishi N, Chen FQ, Zheng HW, Sha SH (2013) Intra-tympanic delivery of short interfering RNA into the adult mouse cochlea. Hear Res 296:36–41
Orrenius S, Zhivotovsky B, Nicotera P (2003) Regulation of cell death: the calcium-apoptosis link. Nat Rev Mol Cell Biol 4:552–565
Pfisterer SG, Mauthe M, Codogno P, Proikas-Cezanne T (2011) Ca2+/calmodulin-dependent kinase (CaMK) signaling via CaMKI and AMP-activated protein kinase contributes to the regulation of WIPI-1 at the onset of autophagy. Mol Pharmacol 80:1066–1075
Racioppi L, Nelson ER, Huang W, Mukherjee D, Lawrence SA, Lento W, Masci AM, Jiao Y, Park S, York B, Liu Y, Baek AE, Drewry DH, Zuercher WJ, Bertani FR, Businaro L, Geradts J, Hall A, Means AR, Chao N, Chang CY, McDonnell DP (2019) CaMKK2 in myeloid cells is a key regulator of the immune-suppressive microenvironment in breast cancer. Nat Commun 10:2450
Racioppi L, Noeldner PK, Lin F, Arvai S, Means AR (2012) Calcium/calmodulin-dependent protein kinase kinase 2 regulates macrophage-mediated inflammatory responses. J Biol Chem 287:11579–11591
Ryan AF, Bennett TM, Woolf NK, Axelsson A (1994) Protection from noise-induced hearing loss by prior exposure to a nontraumatic stimulus: role of the middle ear muscles. Hear Res 72:23–28
Salehi P, Nelson CN, Chen Y, Lei D, Crish SD, Nelson J, Zuo H, Bao J (2018) Detection of single mRNAs in individual cells of the auditory system. Hear Res 367:88–96
Selbert MA, Anderson KA, Huang QH, Goldstein EG, Means AR, Edelman AM (1995) Phosphorylation and activation of Ca(2+)-calmodulin-dependent protein kinase IV by Ca(2+)-calmodulin-dependent protein kinase Ia kinase. Phosphorylation of threonine 196 is essential for activation. J Biol Chem 270:17616–17621
Sha SH, Schacht J (2017) Emerging therapeutic interventions against noise-induced hearing loss. Expert Opin Investig Drugs 26:85–96
Shende P, Patel C (2019) siRNA: an alternative treatment for diabetes and associated conditions. J Drug Target 27:174–182
Shuster B, Casserly R, Lipford E, Olszewski R, Milon B, Viechweg S, Davidson K, Enoch J, McMurray M, Rutherford MA, Ohlemiller KK, Hoa M, Depireux DA, Mong JA, Hertzano R (2021) Estradiol protects against noise-induced hearing loss and modulates auditory physiology in female mice. Int J Mol Sci. https://doi.org/10.3390/ijms222212208
Skelding KA, Rostas JA (2012) The role of molecular regulation and targeting in regulating calcium/calmodulin stimulated protein kinases. Adv Exp Med Biol 740:703–730
Spicer SS, Thomopoulos GN, Schulte BA (1999) Novel membranous structures in apical and basal compartments of inner hair cells. J Comp Neurol 409:424–437
Sun L, Li J, Xiao X (2000) Overcoming adeno-associated virus vector size limitation through viral DNA heterodimerization. Nat Med 6:599–602
Tanaka M, Asaoka M, Yanagawa Y, Hirashima N (2011) Long-term gene-silencing effects of siRNA introduced by single-cell electroporation into postmitotic CNS neurons. Neurochem Res 36:1482–1489
Taylor CR, Levenson RM (2006) Quantification of immunohistochemistry–issues concerning methods, utility and semiquantitative assessment II. Histopathology 49:411–424
Ulfendahl M, Scarfone E, Flock A, Le Calvez S, Conradi P (2000) Perilymphatic fluid compartments and intercellular spaces of the inner ear and the organ of Corti. Neuroimage 12:307–313
Viberg A, Canlon B (2004) The guide to plotting a cochleogram. Hear Res 197:1–10
Walker RA (2006) Quantification of immunohistochemistry–issues concerning methods, utility and semiquantitative assessment I. Histopathology 49:406–410
Wan G, Gomez-Casati ME, Gigliello AR, Liberman MC, Corfas G (2014) Neurotrophin-3 regulates ribbon synapse density in the cochlea and induces synapse regeneration after acoustic trauma. Elife. https://doi.org/10.7554/eLife.03564
Wang J, Puel JL (2018) Toward Cochlear Therapies. Physiol Rev 98:2477–2522
Wang X, Zhu Y, Long H, Pan S, Xiong H, Fang Q, Hill K, Lai R, Yuan H, Sha SH (2018) Mitochondrial calcium transporters mediate sensitivity to noise-induced losses of hair cells and cochlear synapses. Front Mol Neurosci 11:469
Wang Y, Hirose K, Liberman MC (2002) Dynamics of noise-induced cellular injury and repair in the mouse cochlea. J Assoc Res Otolaryngol 3:248–268
Wang Y, Sun Y, Chang Q, Ahmad S, Zhou B, Kim Y, Li H, Lin X (2013) Early postnatal virus inoculation into the scala media achieved extensive expression of exogenous green fluorescent protein in the inner ear and preserved auditory brainstem response thresholds. J Gene Med 15:123–133
Winder WW, Thomson DM (2007) Cellular energy sensing and signaling by AMP-activated protein kinase. Cell Biochem Biophys 47:332–347
Wu F, Xiong H, Sha S (2020) Noise-induced loss of sensory hair cells is mediated by ROS/AMPKalpha pathway. Redox Biol 29:101406
Xiong H, Long H, Pan S, Lai R, Wang X, Zhu Y, Hill K, Fang Q, Zheng Y, Sha SH (2019) Inhibition of histone methyltransferase G9a attenuates noise-induced cochlear synaptopathy and hearing loss. J Assoc Res Otolaryngol 20:217–232
Yamane H, Nakai Y, Takayama M, Konishi K, Iguchi H, Nakagawa T, Shibata S, Kato A, Sunami K, Kawakatsu C (1995) The emergence of free radicals after acoustic trauma and strial blood flow. Acta Otolaryngol Suppl 519:87–92
Yamashita D, Jiang HY, Schacht J, Miller JM (2004) Delayed production of free radicals following noise exposure. Brain Res 1019:201–209
Yu Q, Wang Y, Chang Q, Wang J, Gong S, Li H, Lin X (2014) Virally expressed connexin26 restores gap junction function in the cochlea of conditional Gjb2 knockout mice. Gene Ther 21:71–80
Yuan H, Wang X, Hill K, Chen J, Lemasters J, Yang SM, Sha SH (2015) Autophagy attenuates noise-induced hearing loss by reducing oxidative stress. Antioxid Redox Signal 22:1308–1324
Zheng HW, Chen J, Sha SH (2014) Receptor-interacting protein kinases modulate noise-induced sensory hair cell death. Cell Death Dis 5:e1262
Acknowledgements
We thank Dr. Jochen Schacht for his valuable comments on the manuscript. We also thank Dr. Yuan Shao in the MUSC Biorepository & Tissue Analysis Shared Resource for technical assistance with in-situ hybridization RNAscope and Andra Talaska for proofreading of the manuscript.
Funding
The research project described was supported by grant R01 DC009222 from the National Institute on Deafness and Other Communication Disorders, National Institutes of Health. All experiments described in this manuscript were conducted in the WR Building at Medical University of South Carolina in renovated space supported by grant C06 RR014516. Animals were housed in MUSC CRI animal facilities supported by grant C06 RR015455 from the Extramural Research Facilities Program of the National Center for Research Resources.
Author information
Authors and Affiliations
Contributions
FW, KH, QF authors have contributed equally to this work. SS designed research; FW, KH, QF, ZH, HZ, XW, and HX performed research; FW, KH, QF, and SS analyzed data; FW, KH, and SS wrote the paper. All authors have reviewed the contents of the manuscript, approve of its contents, and validate the accuracy of the data.
Corresponding author
Ethics declarations
Conflict of interests
The authors declare that they have no competing interests.
Ethics approval and consent to participate
No human data or tissue were involved. All animal research protocols were approved by the Institutional Animal Care & Use Committee at MUSC. Animal care was under the supervision of the Division of Laboratory Animal Resources at MUSC.
Consent for publication
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Wu, F., Hill, K., Fang, Q. et al. Traumatic-noise-induced hair cell death and hearing loss is mediated by activation of CaMKKβ. Cell. Mol. Life Sci. 79, 249 (2022). https://doi.org/10.1007/s00018-022-04268-4
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00018-022-04268-4