Abstract
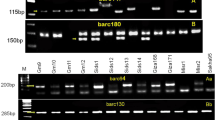
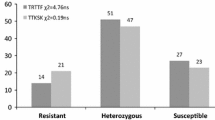
The entire spectrum of cultivated turmeric germplasm available today is highly susceptible to rhizome rot disease caused by necrotrophic oomycete Pythium aphanidermatum. Conventional host resistance breeding approaches have not been very successful in the crop owing to its lack of genetic variability, obligatory asexual nature and stigmatic incompatibility. The present study sought to develop easy, rapid and reliable molecular markers to identify new turmeric germplasm that could be used as source of resistance to P. aphanidermatum. Bulk segregant analyses using pooled DNA from resistant and susceptible genotypes with 40 inter simple sequence markers resulted in the isolation of a putative resistance specific marker ClRSM. The resistance specific fragment was converted into a sequence tagged marker ClSTS, which could successfully amplify a 720 bp, fragment in resistance turmeric genotypes but not in the susceptible plants. Southern blotting confirmed it as a single copy locus found associated with the resistant genotypes. Further, the ClSTS marker precisely identified 10 resistant and five susceptible genotypes among the 15 turmeric germplasm with unknown disease response collected from different agroclimatic regions. Inoculation assessment of the 15 individuals with a virulent strain of P. aphanidermatum corroborated with the STS marker results. Hence, the identified STS marker can be use as an efficient tool for early and rapid identification of new turmeric cultivars resistant to rhizome rot through marker-assisted selection.





Similar content being viewed by others
Abbreviations
- ClRSM:
-
Curcuma longa resistance specific marker
- ISSR:
-
Inter simple sequence repeats
- STS:
-
Sequence tagged sites
References
Agrawal M, Shrivastava N, Padh H (2008) Advances in molecular marker techniques and their applications in plant sciences. Plant Cell Rep 27:617–631
Duke JA (2002) CRC Handbook of Medicinal Spices. CRC Press LLC, Boca Raton, p 33431
Dake G (1995) Diseases of ginger (Zingiber officinale Rosc.) and their management. J Sp Arom Crops 4:40–48
Danilova TV, Karlov GI (2006) Application of inters simple sequence repeat (ISSR) polymorphism for detection of sex-specific molecular markers in hop (Humulus lupulus L.). Euphytica 151:15–21
Dhamayanthi KPM, Sasikumar B, Remashree AB (2003) Reproductive biology and incompatibility studies in ginger (Zingiber officinale Rosc.). Phytomorphology 53:123–131
Doyle JJ, Doyle JL (1990) Isolation of plant genomic DNA from fresh tissue. Focus 12(1):13–15
Gupta SK, Charpe A, Prabhu KV, Haque QMR (2006) Identification and validation of molecular markers linked to the leaf rust resistance gene Lr19 in wheat. Theor Appl Genet 113(6):1027–1036
Jain S, Shrivastava S, Nayak S, Sumbhate S (2007) Recent trends in Curcuma longa L. Pharmacognosy Rev 1(1):119–128
Jeong YM, Mun JH, Lee I, Woo JC, Hong CB, Kim SG (2006) Distinct Roles of the First Introns on the Expression of Arabidopsis Profilin Gene Family Members. Plant Physiol 140:196–209
Kar B, Nanda S, Nayak PK, Nayak S, Joshi RK (2013) Molecular characterization and functional analysis of CzR1, a coiled coil-nucleotide-binding-site-leucine rich repeat R-gene from Curcuma zedoaria L that confers resistance to Pythium aphanidermatum. Physiol Mol Plant Pathol 83:59–68
Kavitha PG, Thomas G (2007) Evaluation of Zingiberaceae for resistance to ginger soft rot caused by Pythium aphanidermatum (Edson) Fitzp. Plant Gent Resorc News Lett 152:1–4
Kavitha PG, Thomas G (2008) Population genetic structure of the clonal plant Zingiber zerumbet (L.) Smith (Zingiberaceae), a wild relative of cultivated ginger, and its response to Pythium aphanidermatum. Euphytica 160:89–100
Lee J, Do JW, Yoon JB (2011) Development of STS markers linked to the major QTLs for resistance to the pepper anthracnose caused by Colletotrichum acutatum and C. capsici. Hort Envron Biotechnol 52(6):596–601
Michelmore R, Paran I, Keselli V (1991) Identification of markers linked to disease resistance genes by bulk segregant analysis: a rapid method to detect markers in specific genomic regions by using segregating populations. Proc Natl Acad Sci USA 88:9828–9832
Moon H, Nicholson JS (2007) AFLP and SCAR Markers Linked to TomatoSpotted Wilt Virus Resistance in Tobacco. Crop Sci 47:1887–1894
Nair KPP (2013) The agronomy and economy of Turmeric and Ginger: The Invaluable Medicinal Spice Crops. Newnes Publishers. Pp 140–143.
Nanda S, Kar B, Nayak S, Jha S, Joshi RK (2013) Development of an ISSR based STS marker for sex identification in pointed gourd (Trichosanthes dioica Roxb.). Sci Hort 150:11–15
Joshi RK, Mohanty S, Kar B, Nayak S (2012) Assessment of genetic diversity in Zingiberaceae through nucleotide binding site (NBS) based motif directed profiling. Biochem Genet 50:642–656
Ratnaparkhe MB, Santra DK, Tullu A, Muehlbauer FJ (1998) Inheritance of inter-simple-sequence-repeat polymorphism and linkage with a Fusarium wilt resistance gene in chickpea. Theor Appl Genet 96:348–353
Ravindran PN, Nirmalbabu K, Sivaraman K (2007) Turmeric: the genus Curcuma (Medicinal and Aromatic plants - Industrial profiles). CRC Press, Florida
Reddy MP, Sarla N, Siddiq EA (2002) Inter simple sequence repeat (ISSR) polymorphism and its application in plant breeding. Euphytica 128:9–17
Ruan CJ, Li H, Mopper S (2009) Characterization and identification of ISSR markers associated with resistance to dried-shrink disease in sea buckthorn. Mol Breed 24:255–268
Salgueiro S, Pignocchi C, Parry MAJ (2000) Intron-mediated gusA ex-pression in Tritordeum and wheat resulting from particle bombardment. Plant Mol Biol 42:615–622
Sasikumar B (2005) Genetic resources of Curcuma: diversity, characterization and utilization. Plant Genet Res 3:230–251
Selvan MT, Thomas KG, Manojkumar K, Ginger (Zingiber officinale Rosc.) (2002) In: Singh HP, Sivaraman K, Selvan MT (eds) Indian spices – production and utilization, Coconut Development Board, pp 110–131
Shalini KV, Manjunatha S, Lebrun P, Berger A, Baudouin L, Pirany N, Ranganath RM, Prasad DT (2007) Identification of molecular markers associated with mite resistance in coconut (Cocos nucifera L.). Genome 50:35–42
Sievers F, Wilm A, Dineen D, Gibson TJ, Karplus K, Li W, Lopez R, McWilliam H, Remmert M, Söding J, Thompson JD, Higgins DG (2011) Fast, scalable generation of high-quality protein multiple sequence alignments using Clustal Omega. Mol Syst Biol 7:539
Sigrist MS, Pinheiro JB, Filho JAA, Zucchi MI (2011) Genetic diversity of turmeric germplasm (Curcuma longa; Zingiberaceae) identified by microsatellite markers. Genet Mol Res 10(1):419–428
Singh S, Sidhu JS, Huang N, Vikal Y, Li Z, Brar DS, Dhaliwal HS, Khush GS (2001) Pyramiding three bacterial blight resistance genes (xa5, xa13 and Xa21) using marker-assisted selection into indica rice cultivar PR106. Theor Appl Genet 102(6):1011–1015
Untergrasser A, Cutcutache I, Koressaar T, Ye J, Faircloth BC, Remm M, Rozen SG (2012) Primer3 - new capabilities and interfaces. Nucl Acids Res 40(15):e115
Ushamailini C, Nakkeeran S, Marimuthu T (2008) Development of biomanure for the management of turmeric rhizome rot. Arch Phytopathol Plant Protect 41(5):365–376
Wang W, Hu Y, Sun D, Staehelin C, Xin D, Xe J (2012) Identification and evaluation of two diagnostic markers linked to Fusarium wilt resistance (race 4) in banana (Musa spp.). Mol Biol Rep 39:451–459
Wen WE, Li GQ, He ZH, Yang WY, Xu ML, Xia XC (2008) Development of an STS marker tightly linked to Yr26 against wheat stripe rust using the resistance gene-analog polymorphism (RGAP) technique. Mol Breed 22:507–515
Acknowledgement
BK gratefully acknowledges the financial assistance in the form of senior research fellowship from the Council of Scientific and Industrial Research, Government of India, New Delhi. The authors are thankful to the President, Siksha O Anusandhan University for his guidance and support and grateful to DST-FIST, Govt. of India, for the facilities provided to Centre of Biotechnology, Siksha O Anusandhan University.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(DOCX 19 kb)
Rights and permissions
About this article
Cite this article
Kar, B., Nayak, S. & Joshi, R.K. Development and evaluation of STS diagnostic marker to track turmeric (Curcuma longa L.) resistance against rhizome rot caused by Pythium aphanidermatum . Australasian Plant Pathol. 43, 167–175 (2014). https://doi.org/10.1007/s13313-013-0259-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13313-013-0259-2