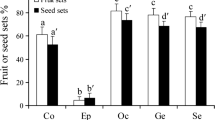
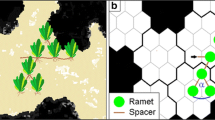
Abstract
Clonal plants can employ two distinct reproductive strategies, clonal and sexual reproduction. Bamboos are monocarpic clonal plants that flower after a long-term clonal reproduction, and then die after seed production. To explore the effect of spatial structure formed through clonal reproduction on the evolution of such long flowering intervals, we developed a model for population growth of clonal plants on a regular lattice. We applied the pair-approximation that enables us to obtain a closed form of the dynamics. We derived the condition that an infinitely long flowering interval (a pure clonal strategy) or annual flowering strategy became an evolutionarily stable strategy. We demonstrate that localized dispersal broadens the region in which finite flowering interval evolves. Our finding highlights the importance to take into account the spatial structure on the studies of life history strategy in clonal plants.




Similar content being viewed by others
References
Janzen, D.H.: Why bamboos wait so long to flower. Ann. Rev. Ecol. Syst. 7, 347–391 (1976)
Nakashizuka, T.: Regeneration of beech (Fagus crenata) after the simultaneous death of undergrowing dwarf bamboo (Sasa kurilensis). Ecol. Res. 3, 21–35 (1988)
Isagi, Y., Shimada, K., Kushima, H., Tanaka, N., Nagao, A., Ishikawa, T., Onodera, H., Watanabe, S.: Clonal structure and flowering traits of a bamboo [Phyllostachys pubescens (Mazel) Ohwi] stand grown from a simultaneous flowering as revealed by AFLP analysis. Mol. Ecol. 13, 2017–2021 (2004)
Hisamoto, Y., Kobayashi, M.: Flowering habit of two bamboo species, Phyllostachys meyeri and Shibataea chinensis, analyzed with flowering gene expression. Plant Species Biol. 28, 109–117 (2013)
Campbell, J.J.N.: Bamboo flowering patterns: a global view with special reference to East Asia. J. Am. Bamboo Soc. 6, 17–35 (1985)
Gadgil, M., Prasad, S.N.: Ecological determinants of life history evolution of two Indian bamboo species. Biotropica 16, 161–172 (1984)
Keeley, J.E., Bond, W.J.: Mast flowering and semelparity in bamboos: the bamboo fire cycle hypothesis. Am. Nat. 154, 383–391 (1999)
Makita, A.: The significance of the mode of clonal growth in the life history of bamboos. Plant Species Biol. 13, 85–92 (1998)
McClure, F.A.: The bamboos: a fresh perspective. Harvard University Press, Cambridge (1966)(Republished from Smithsonian Institution Press, Washington DC and London. 1993)
Taihui, W.: Some ideas about the origin of bamboos. J. Bamboo Res. 2(1), 1–10 (1983)
Tachiki, Y., Makita, A., Suyama, Y., Satake, A.: A spatially explicit model for flowering time in bamboos: long rhizomes drive the evolution of delayed flowering. J. Ecol. 103, 585–593 (2015)
Matsuda, H., Ogita, N., Sasaki, A., Satō, K.: Statistical mechanics of population the lattice lotka-volterra model. Prog. Theor. Phys. 88(6), 1035–1049 (1992)
Harada, Y., Iwasa, Y.: Lattice population dynamics for plants with dispersing seeds and vegetative propagation. Res. Popul. Ecol. 36(2), 237–249 (1994)
Satō, K., Matsuda, H., Sasaki, A.: Pathogen invasion and host extinction in lattice structured populations. J. Math. Biol. 32(3), 251–268 (1994)
Harada, Y., Ezoe, H., Iwasa, Y., Matsuda, H., Sato, K.: Population persistence and spatially limited social interaction. Theor. Popul. Biol. 48(1), 65–91 (1995)
Nakamaru, M., Matsuda, H., Iwasa, Y.: The evolution of cooperation in a lattice-structured population. J. Theor. Biol. 184(1), 65–81 (1997)
Satake, A., Bjørnstad, O.N.: Spatial dynamics of specialist seed predators on synchronized and intermittent seed production of host plants. Am. Nat. 163(4), 591–605 (2004)
Maynard Smith, J., Price, G.R.: The logic of animal conflict. Nature 246, 15–18 (1973)
Fahrig, L., Coffin, D.P., Lauenroth, W.K., Shugart, H.H.: The advantage of long-distance clonal spreading in highly disturbed habitats. Evol. Ecol. 8, 172–187 (1994)
Geritz, S.A.H., Kisdi, É., Meszéna, G., Metz, J.A.J.: Evolutionarily singular strategies and the adaptive growth and branching of the evolutionary tree. Evol. Ecol. 12, 35–57 (1998)
Takenaka, Y., Matsuda, H., Iwasa, Y.: Competition and evolutionary stability of plants in a spatially structured habitat. Res. Popul. Ecol. 39(1), 67–75 (1997)
Ikegami, M., Whigham, D.F., Werger, M.J.A.: Effects of local density of clonal plants on their sexual and vegetative propagation strategies in a lattice structure model. Ecol. Model. 234, 51–59 (2012)
Takada, T., Nakajima, H.: The optimal allocation to seed reproduction and vegetative reproduction in perennial plants—an application of the density-dependent transition matrix model. J. Theor. Biol. 182, 179–191 (1996)
Levin, S.A., Muller-Landau, H.C., Nathan, R., Chave, J.: The ecology and evolution of seed dispersal: a theoretical perspective. Ann. Rev. Ecol. Evol. Syst. 34, 575–604 (2003)
Nathan, R.: Long-distance dispersal of plants. Science 313, 786–788 (2006)
Acknowledgments
This work was supported by Grant-in-Aid for JSPS Fellows Number 2542 and JSPS KAKENHI [15H04518] to Y.T. We are grateful to Yuma Sakai, Kazunori Satō and Takenori Takada for useful comments.
Author information
Authors and Affiliations
Corresponding author
Appendix: Derivation of population dynamics by using pair-approximation
Appendix: Derivation of population dynamics by using pair-approximation
By using the definition of local density, Eq. 1 in the main text is written as follows:
Note that we abbreviate the time t from the equation, because the time of every variable \((p_\mathrm{r} ,q_{\mathrm{r}/\mathrm{r}} )\) in the right-hand side of equality is always \(t. {p}'_\mathrm{r}\) means the global density in year \(t+1, p_{\mathrm{r,}t+1} \). We need to derive the dynamics of pair density for both clonal and sexual reproduction. When individuals undergo clonal reproduction \((\hbox {if }t\hbox { mod } \tau _\mathrm{r} \ne \hbox {0})\), the doublet density in the next year \(({p}'_{\mathrm{rr}} =p_{\mathrm{rr,}t+1} )\) is written as follows;
where \(q_{\sigma _0 /\sigma _1 \sigma _2 } \{\sigma _0 ,\sigma _1 ,\sigma _2 =0\hbox { or r}\}\) is the conditional probability that the randomly chosen n.n. site of \(\sigma _1 \) site is \(\sigma _0 \) site, provided that another randomly chosen n.n. site of \(\sigma _1 \)site is \(\sigma _2 \) site. We should proceed further to calculate the dynamics of theses conditional probabilities of a higher order by tracing the dynamics of triplet probabilities, but the dynamics would contain conditional probabilities of higher orders. Hence we adopt pair approximation to construct a closed dynamics of singlet and doublet densities by neglecting correlations beyond pairs (Matsuda et al. [12]; Harada and Iwasa [13]):
Using pair approximation and the definition of conditional probability, Eq. 36 becomes:
The top of Eqs. 36 and 39 constitute a closed dynamical system of two variables, \(p_\mathrm{r} \) and \(q_{\mathrm{r}/\mathrm{r}} \).
When we apply the same method to the case of sexual reproduction, we get the dynamics of local density as follows:
The bottom of Eqs. 36 and 39 also constitute a closed dynamical system of two variables. Hence the population dynamics of bamboo can be expressed as follows:
Hence we can apply standard analysis to a pair of non-linear difference equation.
About this article
Cite this article
Tachiki, Y., Satake, A. Pair-approximation analyses of evolutionarily stable strategies for flowering time in clonal monocarpic plants. Japan J. Indust. Appl. Math. 32, 615–639 (2015). https://doi.org/10.1007/s13160-015-0194-4
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13160-015-0194-4