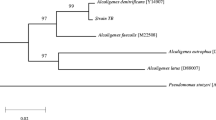
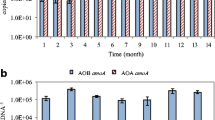
Abstract
Coexistence of an autotrophic ammonia-oxidizing bacterium (Nitrosomonas sp. RA) and heterotrophic bacteria was consistently observed when cultured in an inorganic medium without any external supply of organic carbon. The present study was undertaken to understand the association between autotrophs and the associated heterotrophs for which a system containing active autotrophs and heterotrophs controlled by Hg2+ addition was developed. The study revealed interdependence of heterotrophs and Nitrosomonas sp. RA for growth under iron-limited condition. Growth of Nitrosomonas sp. RA was supported by siderophores produced by the associated heterotroph, Pusillimonas sp., thereby complementing its high iron requirement while the organics (such as pyruvate) excreted by Nitrosomonas sp. RA during its autotrophic growth supported the survival of heterotrophs in the inorganic medium. The study thus sheds light on the nature of the mutual interactions between heterotrophs and autotrophs that play a role in the ammonia-oxidizing system involved in wastewater treatment.






Similar content being viewed by others
References
Ahn YH (2006) Sustainable nitrogen elimination biotechnologies: a review. Process Biochem 41:1709–1721
Andrews SC, Robinson AK, Rodriguez-Quinones F (2003) Bacterial iron homeostasis. FEMS Microbiol Rev 27:215–237
Anthone GE, Barrett DM (2003) Modified method for the determination of pyruvic acid with dinitrophenylhydrazine in the assessment of onion pungency. J Sci Food Agric 83:1210–1213
Arnow LE (1937) Colorimetric determination of the components of 3,4-dihydroxyphenylalanine-tyrosine mixtures. J Biol Chem 118:531–537
Byers BR, Arceneaux JE (1998) Microbial iron transport: iron acquisition by pathogenic microorganisms. Met Ions Biol Syst 35:37–66
Cao B, Ma T, Ren Y, Ren Y, Li G, Li P, Guo X et al (2011) Complete genome sequence of Pusillimonas sp. T7-7, a cold tolerant diesel oil-degrading bacterium isolated from the Bohai Sea in China. J Bacteriol 193:4021–4022
Chain P, Lamerdin J, Larimer F, Regala W, Lao V, Land M, Hauser L et al (2003) Complete genome sequence of the ammonia-oxidizing bacterium and obligate chemolithoautotroph Nitrosomonas europaea. J Bacteriol 185:2759–2773
Clark C, Schmidt EL (1966) Effect of mixed culture on Nitrosomonas europaea simulated by uptake and utilization of pyruvate. J Bacteriol 91:367–373
Depkat-Jakob PS, Hilgarth M, Horn MA, Drake HL (2010) Effect of earthworm feeding guilds on ingested dissimilatory nitrate reducers and denitrifiers in the alimentary canal of the earthworm. Appl Env Microbiol 76:6205–6214
Eisenman L (1999) The home winemakers manual. pp 151–152. Available online: http://www.geocities.com/lumeisenman/
Geetha R, Desai A, Archana G (2009) Effect of the expression of Escherichia coli fhuA gene in Rhizobium sp. IC3123 and ST1 in planta: its role in increased nodule occupancy and function in pigeon pea. Appl Soil Ecol 43:185–190
Gibson F, Magrath DI (1969) Isolation characterization of a hydroxamic acid (aerobactin) formed by Aerobacter aerogenes. Biochim Biophys Acta 192:175–184
Heylen K, Lebbe L, De Vos P (2008) Acidovorax caeni sp. nov., a denitrifying species with genetically diverse isolates from activated sludge. Int J Syst Evol Microbiol 58:73–77
Hockenbury MR, Grady CP Jr (1977) Inhibition of nitrification-effects of selected organic compounds. Water Env Fed 49:768–777
Holger R, Anja F, Peter S, Manfred R, Erko S (1999) Bacillus silvestris sp. nov., a new member of the genus Bacillus that contains lysine in its cell wall. Int J Syst Bacteriol 49:95–802
Hyman MR, Arp DJ (1992) 14C2H2 and 14CO2-labeling studies of the de novo synthesis of polypeptides by Nitrosomonas europaea during recovery from acetylene and light inactivation of ammonia monooxygenase. J Biol Chem 267:1534–1545
Jadhav RS, Desai AJ (1992) Isolation and characterization of siderophore from cowpea Rhizobium (peanut isolate). Curr Microbiol 24:137–141
Jones RD, Hood MA (1980) Interaction between an ammonium-oxidizer, Nitrosomonas sp., and two heterotrophic bacteria, Nocardia atlantica and Pseudomonas sp.: a note. Microb Ecol 6:271–275
Joshi F, Chaudhari A, Joglekar P, Archana G, Desai A (2008) Effect of expression of Bradyrhizobium japonicum 61A152 fegA gene in Mesorhizobium sp., on its competitive survival and nodule occupancy on Arachis hypogeal. Appl Soil Ecol 40:338–347
Kindaichi T, Ito T, Okabe S (2004) Ecophysiological interaction between nitrifying bacteria and heterotrophic bacteria in autotrophic nitrifying biofilms as determined by microautoradiography-fluorescence in situ hybridization. Appl Env Microbiol 70:1641–1650
Klotz MG, Arp DJ, Chain PSG, El-Sheikh AF, Hauser L, Hommes NG, Larimer FW et al (2006) The complete genome sequence of the marine, chemolithoautotrophic, ammonia-oxidizing bacterium Nitrosococcus oceani ATCC19707. Appl Environ Microbiol 72:6299–6315
Kowalchuk GA, Stephen JR (2001) Ammonia-oxidizing bacteria: a model for molecular microbial ecology. Annu Rev Microbiol 55:485–529
Meiklejohn J (1953) Iron and the nitrifying bacteria. J Gen Microbiol 8:58–65
Nogueira R, Elenter D, Brito ALF, Melo LF, Wagner M, Morgenroth E (2005) Evaluating heterotrophic growth in a nitrifying biofilm reactor using fluorescence in situ hybridization and mathematical modeling. Water Sci Technol 52:135–141
Norton JM, Klotz MG, Stein LY, Arp DJ, Bottomley PJ, Chain PSG, Hauser LJ et al (2008) Complete genome sequence of nitrosospira multiformis, an ammonia-oxidizing bacterium from the soil environment. Appl Environ Microbiol 74:3559–3572
Ohashi A, Viray De Silva DG, Mobarry B, Manem JA, Stahl DA, Rittmann BE (1995) Influence of substrate C/N ratio on the structure of multi-species biofilms consisting of nitrifiers and heterotrophs. Water Sci Technol 32:75–84
Okabe S, Hirata K, Ozawa Y, Watanabe Y (1996) Spatial microbial distributions of nitrifiers and heterotrophs in mixed population biofilms. Botechnol Bioeng 50:24–35
Patel KD, Chudasama CJ, Ingle SS (2011) Molecular characterization of Bacillus thuringiensis isolated from diverse habitats of India. J Basic Microbiol 51:1–9
Postle K (1993) TonB protein and energy transduction between membranes. J Bioenerg Biomemb 25:591–601
Powell PE, Szaniszlo PJ, Reid CPP (1983) Confirmation of occurrence of hydroxamate siderophores in soil by a novel Escherichia coli bioassay. Appl Environ Microbiol 46:1080–1083
Rittmann BE, Regan JM, Stahl DA (1994) Nitrification as a source of soluble organic substrate in biological treatment. Water Sci Technol 30:1–8
Scheiner D (1976) Determination of ammonia and Kjeldahl nitrogen by indophenol method. Water Res 10:31–36
Schmidt TM, Delong TEF, Pace NR (1991) Analysis of a marine picoplankton community by 16S rRNA gene cloning and sequencing. J Bacteriol 173:4371–4378
Schwyn B, Neilands JB (1987) Universal chemical assay for the detection and determination of siderophores. Anal Biochem 160:47–56
Scott TA, Melvin EH (1953) Quantification of total sugars by anthrone method. Anal Chem 25:1656
Stein LY, Arp DJ, Berube PM, Chain PSG, Hauser L, Jetten MSM, Klotz MG et al (2007) Whole-genome analysis of the ammonia-oxidizing bacterium, Nitrosomonas eutropha C91: implications for niche adaptation. Env Microbiol 9:2993–3007
Tomar M (1999) Quality assessment of water and wastewater. CRS Press, India, pp 184–199
Upadhyay AK, Petasis DT, Arcerio DM, Hooper AB, Hendrich MP (2003) Spectroscopic characterization and assignment of reduction potentials in the tetraheme cytochrome C554 from Nitrosomonas europaea. J Am Chem Soc 125:1738–1747
van Eck G-R (1996) Physiological and chemical tests for drinking water. NEN 1056, IV-2. Nederlands Normalisatie Instituut, Rijswijk, The Netherlands
Vega RR, Corsini D, Le Tourneau D (1970) Nonvolatile organic acids produced by Sclerotinia sclerotiorum in synthetic liquid media. Mycologia 62:332–338
Wei X, Vajrala N, Hauser L, Sayavedra-Soto LA, Arp DJ (2006) Iron nutrition and physiological responses to iron stress in Nitrosomonas europaea. Arch Microbiol 186:107–118
Acknowledgments
This study was funded by University Grants Commission, New Delhi UGC Order No. F. No. 37-375/2009(SR) and GNFC, Bharuch, India. We thank Dr. Pranav Vyas for reviewing the work and his insightful discussion.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Friedrich Widdel.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Keluskar, R., Nerurkar, A. & Desai, A. Mutualism between autotrophic ammonia-oxidizing bacteria (AOB) and heterotrophs present in an ammonia-oxidizing colony. Arch Microbiol 195, 737–747 (2013). https://doi.org/10.1007/s00203-013-0926-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00203-013-0926-2